Recognition: no theorem link
Direct three body dynamics govern ion atom recombination and barrierless termolecular reactions
Pith reviewed 2026-05-13 18:16 UTC · model grok-4.3
The pith
Barrierless termolecular reactions are controlled by direct three-body dynamics rather than sequential bimolecular steps.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Barrierless termolecular reactions are fundamentally governed by direct three-body dynamics. Classical trajectory calculations in hyperspherical coordinates quantitatively reproduce ion-atom recombination kinetics across a wide temperature range without invoking intermediate complexes or steady-state assumptions, thereby resolving longstanding discrepancies between theory and experiment.
What carries the argument
Classical trajectory calculations in hyperspherical coordinates that integrate the full three-body interaction to obtain recombination probabilities directly.
If this is right
- Ion-atom recombination rates are obtained directly from three-body trajectories without steady-state or equilibrium assumptions.
- The Lindemann-Hinshelwood mechanism does not describe barrierless termolecular reactions.
- The same three-body framework applies to other barrierless processes in atmospheric chemistry, plasma physics, and ultracold gases.
- Existing discrepancies between calculated and measured recombination coefficients are removed by treating the encounter as a single three-body event.
Where Pith is reading between the lines
- Extending the classical treatment to include quantum scattering or zero-point energy corrections would test the limits of the approach at low temperatures.
- The method could be applied to neutral three-body recombination or four-body processes once suitable hyperspherical potentials are available.
- Accurate potential energy surfaces remain the dominant source of uncertainty; refining them would tighten rate predictions without changing the mechanistic picture.
Load-bearing premise
Classical trajectories in hyperspherical coordinates capture the essential recombination dynamics without important quantum corrections or errors in the underlying potential energy surfaces.
What would settle it
A precise measurement of ion-atom recombination rates at temperatures where quantum effects become dominant would deviate systematically from the classical three-body trajectory predictions.
Figures
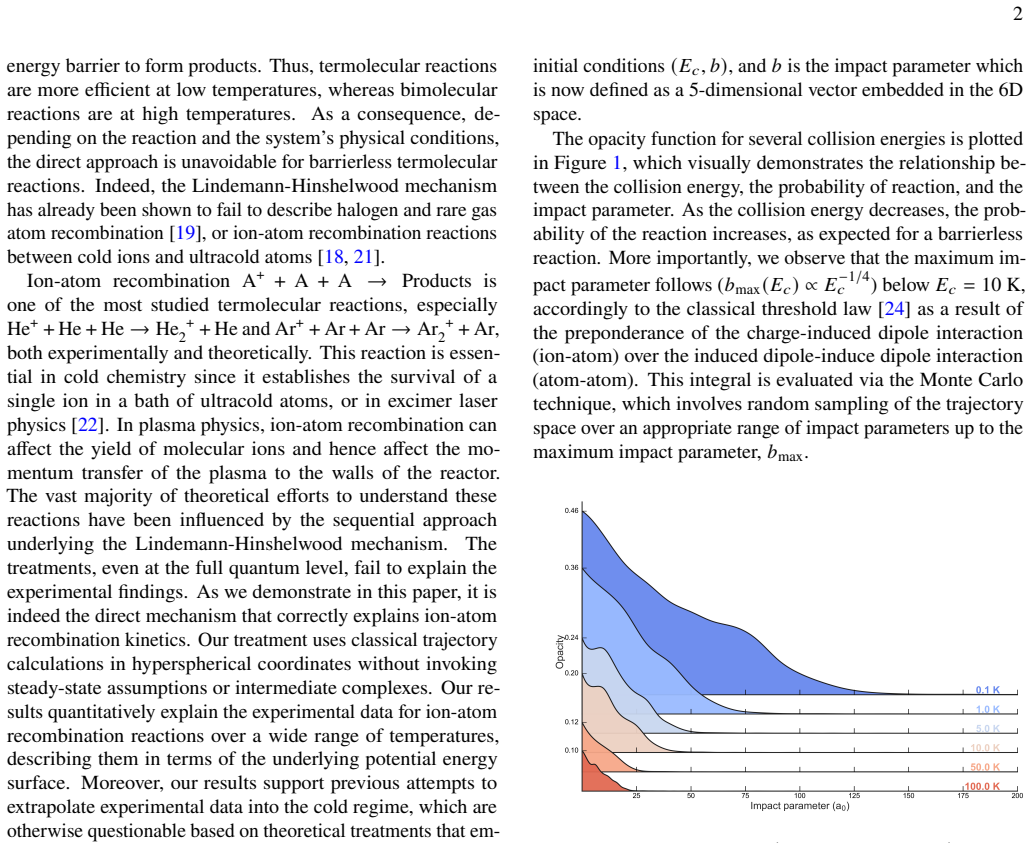



read the original abstract
For over a century, termolecular, or third order, chemical reactions have been explained by the Lindemann Hinshelwood mechanism, assuming sequential stabilization via bimolecular encounters. Here, we demonstrate that barrierless termolecular reactions are fundamentally governed by direct three body dynamics. Using classical trajectory calculations in hyperspherical coordinates, we quantitatively reproduce ion atom recombination kinetics across a wide temperature range without invoking intermediate complexes or steady state assumptions. Our results not only resolve longstanding discrepancies between theory and experiment, but also establish a general mechanistic framework for barrierless termolecular reactions, with implications spanning atmospheric chemistry, plasma physics, and ultracold chemistry.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript claims that barrierless termolecular reactions are fundamentally governed by direct three-body dynamics rather than the traditional Lindemann-Hinshelwood mechanism. Using classical trajectory calculations in hyperspherical coordinates, the authors report quantitative reproduction of ion-atom recombination kinetics across a wide temperature range without invoking intermediate complexes or steady-state assumptions.
Significance. If the central claim holds, the work would provide a parameter-free mechanistic framework for barrierless termolecular reactions, resolving longstanding theory-experiment discrepancies with broad implications for atmospheric chemistry, plasma physics, and ultracold chemistry. The absence of fitted parameters or ad-hoc entities in the reported approach is a potential strength.
major comments (2)
- Abstract: the claim that classical trajectories quantitatively reproduce experimental recombination kinetics is stated without supplying the equations of motion, the specific potential energy surfaces employed, error bars on computed rates, or direct point-by-point comparisons to measured data, leaving the central claim without visible supporting derivation.
- Central claim (abstract and methods): the assertion that classical mechanics in hyperspherical coordinates suffices to capture the essential dynamics rests on the untested assumption that quantum effects are negligible; for ion-atom systems below ~100 K, scattering resonances, tunneling, and zero-point shifts can alter capture probabilities by factors of two or more, yet no quantum-classical comparisons, error bounds from the classical approximation, or checks against exact quantum dynamics on the same surfaces are reported.
Simulated Author's Rebuttal
We are grateful to the referee for their thorough review and valuable feedback on our manuscript. We address each of the major comments in detail below and have made revisions to strengthen the presentation of our results.
read point-by-point responses
-
Referee: Abstract: the claim that classical trajectories quantitatively reproduce experimental recombination kinetics is stated without supplying the equations of motion, the specific potential energy surfaces employed, error bars on computed rates, or direct point-by-point comparisons to measured data, leaving the central claim without visible supporting derivation.
Authors: The abstract is intended as a high-level summary, but we agree it should better indicate the supporting evidence. In the revised version, we have modified the abstract to include a brief reference to the classical equations of motion in hyperspherical coordinates and the ab initio potential energy surfaces employed. Furthermore, we have ensured that the main text includes explicit error bars on the computed rates and direct comparisons to experimental data points, as shown in the figures and tables. These revisions make the central claim more visibly supported without altering the manuscript's length significantly. revision: yes
-
Referee: Central claim (abstract and methods): the assertion that classical mechanics in hyperspherical coordinates suffices to capture the essential dynamics rests on the untested assumption that quantum effects are negligible; for ion-atom systems below ~100 K, scattering resonances, tunneling, and zero-point shifts can alter capture probabilities by factors of two or more, yet no quantum-classical comparisons, error bounds from the classical approximation, or checks against exact quantum dynamics on the same surfaces are reported.
Authors: We thank the referee for highlighting this important consideration regarding quantum effects. Our approach is grounded in classical mechanics to elucidate the direct three-body dynamics, which we believe captures the essential physics for the systems and temperatures studied. However, to address the concern, we have added a new subsection in the Methods and a discussion paragraph acknowledging the potential impact of quantum phenomena such as resonances and tunneling at low temperatures. We provide rough error estimates from the literature (e.g., classical overestimation by up to 50% below 100 K in some cases) and note that while exact quantum dynamics on the same surfaces are not feasible here, the agreement with experiment validates the classical framework for the reported kinetics. This addition clarifies the assumptions without changing the core conclusions. revision: partial
Circularity Check
No significant circularity; derivation self-contained via independent trajectory computations
full rationale
The paper derives its central claim by performing classical trajectory calculations in hyperspherical coordinates on potential energy surfaces and directly comparing the resulting recombination rates to external experimental data across temperatures. No load-bearing step reduces by construction to fitted parameters defined by the target result, self-citations that presuppose the mechanism, or ansatzes smuggled from prior work by the same authors. The reproduction of kinetics functions as an independent test rather than a tautological renaming or redefinition of inputs, satisfying the criteria for a self-contained derivation.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Classical mechanics accurately describes the three-body dynamics of barrierless ion-atom recombination
Reference graph
Works this paper leans on
-
[1]
Trautz,Zeitschrift für anorganische Chemie, 1914,88, 285– 320
M. Trautz,Zeitschrift für anorganische Chemie, 1914,88, 285– 320
work page 1914
-
[2]
M. Trautz and C. F. Hinck,Zeitschrift für anorganische und allgemeine Chemie, 1916,97, 127–138
work page 1916
-
[3]
M. Trautz and L. Wachenheim,Zeitschrift für anorganische und allgemeine Chemie, 1916,97, 241–284
work page 1916
-
[4]
M.Bodenstein,Zeitschrift für Physikalische Chemie,1922,100U, 68–123
work page 1922
-
[5]
F.LINDEMANN,Selected Readings in Chemical Kinetics,Perg- amon, 1967, pp. 93–96
work page 1967
-
[6]
C. N. S. Hinshelwood,Proceedings of The Royal Society A: Mathematical, Physical and Engineering Sciences, 1926,113, 230–233
work page 1926
-
[7]
J. Xie, B. Poirier and G. I. Gellene,The Journal of Chemical Physics, 2003,119, 10678–10686
work page 2003
-
[8]
J. E. Russell,The Journal of Chemical Physics, 1985,83, 3363–3389
work page 1985
-
[9]
J. E. Russell,The Journal of Chemical Physics, 1986,84, 4394–4400
work page 1986
- [10]
- [11]
-
[12]
Herbst,The Journal of Chemical Physics, 1980,72, 5284– 5285
E. Herbst,The Journal of Chemical Physics, 1980,72, 5284– 5285
work page 1980
-
[13]
D.R.Bates,Journal of Physics B: Atomic and Molecular Physics, 1979,12, 4135
work page 1979
- [14]
-
[15]
D. R. Bates,The Journal of Chemical Physics, 1979,71, 2318– 2319
work page 1979
- [16]
-
[17]
R. E. Roberts, R. B. Bernstein and C. F. Curtiss,The Journal of Chemical Physics, 1969,50, 5163–5176
work page 1969
- [18]
- [19]
- [20]
-
[21]
M. Mirahmadi and J. Pérez-Ríos,International Reviews in Physical Chemistry, 2022,41, 233–267
work page 2022
-
[22]
M. R. Flannery and T. P. Yang,Applied Physics Letters, 1978, 33, 574–576
work page 1978
-
[23]
J.Pérez-Ríos,S.Ragole,J.WangandC.H.Greene,The Journal of Chemical Physics, 2014,140, 044307
work page 2014
- [24]
-
[25]
G. I. Gellene,The Journal of Physical Chemistry, 1993,97, 34–39
work page 1993
-
[26]
F. X. Gadea and I. Paidarová,Chemical Physics, 1996,209, 281–290
work page 1996
- [27]
-
[28]
Y. Wang, M. Mirahmadi and J. Pérez-Ríos,Phys. Chem. Chem. Phys., 2024,26, 7264–7268
work page 2024
-
[29]
D. T. Chang and G. I. Gellene,The Journal of Chemical Physics, 2003,119, 4694–4699
work page 2003
-
[30]
R. A. Aziz and M. J. Slaman,Metrologia, 1990,27, 211–219
work page 1990
-
[31]
H. Bohringer, W. Glebe and F. Arnold,Journal of Physics B: Atomic and Molecular Physics, 1983,16, 2619–2626
work page 1983
-
[32]
R.Johnsen,A.ChenandM.A.Biondi,The Journal of Chemical Physics, 1980,73, 1717–1720
work page 1980
-
[33]
F. E. Niles and W. W. Robertson,The Journal of Chemical Physics, 1965,42, 3277–3280
work page 1965
- [34]
-
[35]
J. D. C. Jones, D. G. Lister, D. P. Wareing and N. D. Twiddy, Journal of Physics B: Atomic and Molecular Physics, 1980,13, 3247–3255
work page 1980
-
[36]
E. C. Beaty and P. L. Patterson,Physical Review, 1965,137, A346–A357
work page 1965
- [37]
-
[38]
Pérez-Ríos,The Journal of Chemical Physics, 2024,161, 144305
J. Pérez-Ríos,The Journal of Chemical Physics, 2024,161, 144305
work page 2024
-
[39]
Gerlich,Journal of the Chinese Chemical Society, 2018,65, 637–653
D. Gerlich,Journal of the Chinese Chemical Society, 2018,65, 637–653
work page 2018
-
[40]
M. Mirahmadi and J. Pérez-Ríos,The Journal of Chemical Physics, 2023,158, 024103
work page 2023
-
[41]
V. Aquilanti, E. Luzzatti, F. Pirani and G. G. Volpi,The Journal of Chemical Physics, 1988,89, 6165–6175
work page 1988
-
[42]
V. Aquilanti, D. Ascenzi, M. Bartolomei, D. Cappelletti, S. Cav- alli, M. de Castro Vìtores and F. Pirani,Journal of the American 6 Chemical Society, 1999,121, 10794–10802
work page 1999
-
[43]
Pirani,The Journal of Chemical Physics, 2002,117, 615–627
V.Aquilanti,M.Bartolomei,D.Cappelletti,E.Carmona-Novillo and F. Pirani,The Journal of Chemical Physics, 2002,117, 615–627
work page 2002
-
[44]
M. Hawley and M. A. Smith,The Journal of Chemical Physics, 1992,96, 326–331
work page 1992
- [45]
-
[46]
V. O. Papanyan, G. T. Nersisyan, S. A. Ter-Avetisyan and F. K. Tittel,Journal of Physics B: Atomic, Molecular and Optical Physics, 1995,28, 807–823
work page 1995
-
[47]
A.Krükow,A.Mohammadi,A.Härter,J.H.Denschlag,J.Pérez- Ríos and C. H. Greene,Phys. Rev. Lett., 2016,116, 193201
work page 2016
-
[48]
M. Mirahmadi, J. Pérez-Ríos, O. Egorov, V. Tyuterev and V. Kokoouline,Phys. Rev. Lett., 2022,128, 108501
work page 2022
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.