Recognition: unknown
Thermodynamic Properties of Diatomic Molecules from the Frost-Musulin Potential
Pith reviewed 2026-05-09 20:36 UTC · model grok-4.3
The pith
The Frost-Musulin potential with Pekeris approximation produces accurate Gibbs free energy for H2 and LiH across temperatures.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
By combining the analytical bound-state approach to the radial Schrödinger problem with the near-equilibrium Pekeris representation, the authors obtain a validated rotation-vibration spectrum. These bound states are combined with standard translational and rotational ideal-gas contributions to construct the total partition function and the corresponding thermodynamic observables. The resulting formulation captures the Gibbs free energy deviation function for both molecules with high quantitative accuracy and provides chemically plausible trends for heat capacity and enthalpy increase over a wide temperature range, while showing that residual high-temperature errors arise mainly from the lack
What carries the argument
Frost-Musulin potential solved analytically for bound states via the near-equilibrium Pekeris representation, then used to build the molecular partition function together with ideal-gas translational and rotational terms.
If this is right
- The model recovers a large share of the observed thermochemistry for the two diatomic molecules.
- Heat capacity and enthalpy follow chemically plausible trends over a broad temperature interval.
- High-temperature errors in derivative quantities are traced to the absence of inelastic rotational and dissociation channels rather than to the bound-state calculation itself.
- The potential supplies a compact analytical description of the bound region and marks the temperature regime where fuller molecular statistical mechanics becomes necessary.
Where Pith is reading between the lines
- The same analytical route could be tried on other diatomic molecules whose potentials admit comparable closed-form solutions.
- A minimal correction for dissociation might extend reliable predictions to higher temperatures while preserving tractability.
- The clear separation between bound-state and continuum contributions offers a baseline for testing more elaborate potential models in computational thermochemistry.
Load-bearing premise
The bound-state spectrum from the Frost-Musulin potential plus Pekeris approximation, together with standard translational and rotational ideal-gas terms, is sufficient to reach high accuracy in the Gibbs free energy while high-temperature deviations can be blamed entirely on neglected inelastic and dissociation effects.
What would settle it
Direct comparison of the calculated versus measured Gibbs free energy at high temperatures after an explicit dissociation term is added to the partition function; a marked reduction in deviation would confirm the claim.
Figures
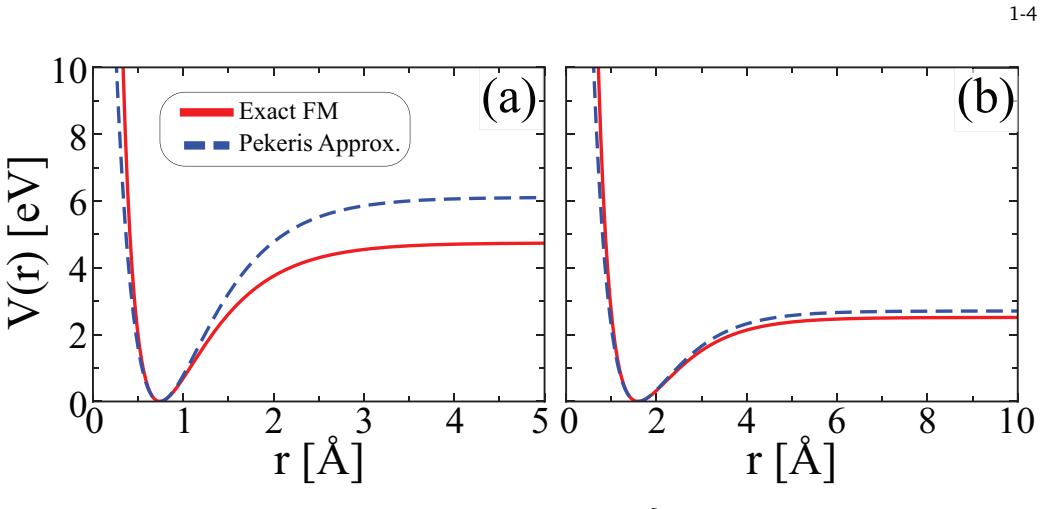


read the original abstract
In this study, we present a quantum-statistical analysis of H$_2$ and LiH diatomic molecules within the Frost--Musulin potential framework. By combining the analytical bound-state approach to the radial Schr\"odinger problem with the near-equilibrium Pekeris representation, we obtain a validated rotation-vibration spectrum that reproduces a physically consistent ordering of energy levels. These bound states are subsequently combined with standard translational and rotational ideal gas contributions to construct the total partition function and the corresponding thermodynamic observables of the ground state. The resulting formulation captures the Gibbs free energy deviation function for both molecules with high quantitative accuracy and provides chemically plausible trends for heat capacity and enthalpy increase over a wide temperature range. At the same time, residual errors become increasingly pronounced in derivative-sensitive quantities, particularly at high temperatures; this indicates that the dominant limitations now stem not from the local bound-state spectrum itself, but from the neglect of inelastic rotational, continuity contributions and dynamics close to dissociation. Consequently, the present results define the potential model as a compact and analytically tractable representation of the bound region, recovering a significant portion of the observed thermochemistry whilst also delineating the regime where more comprehensive molecular statistical mechanics is required.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper applies the Frost-Musulin potential to H2 and LiH, obtaining an analytical bound-state spectrum via the radial Schrödinger equation combined with the Pekeris approximation for the centrifugal term. These levels, together with standard translational and rotational ideal-gas contributions, are inserted into the partition function to derive thermodynamic quantities, with the central claim being that the Gibbs free energy deviation function is reproduced with high quantitative accuracy while high-T deviations in derivatives arise from neglected dissociation and inelastic channels.
Significance. If the spectrum accuracy and quantitative Gibbs claims can be independently verified, the work supplies a compact, analytically tractable representation of the bound-region contribution to diatomic thermochemistry that could serve as a useful benchmark or rapid-estimate tool, while clearly demarcating the temperature range where more complete treatments (including continuum states) become necessary.
major comments (3)
- [Abstract] Abstract: the claim that the formulation 'captures the Gibbs free energy deviation function for both molecules with high quantitative accuracy' is unsupported by any numerical tables, error metrics, direct comparisons to experimental thermochemical data, or reference calculations; without these, the accuracy assertion cannot be evaluated.
- [Abstract] Abstract and methods description of the Pekeris approximation: the approximation replaces the centrifugal term by a first-order expansion about r_e and is stated to yield a 'validated' spectrum, yet no assessment of its accuracy for high-v, high-J levels (which dominate the partition function at elevated T) or comparison to exact numerical eigenvalues is provided; this directly affects whether residual high-T errors can be attributed solely to neglected dissociation.
- [Abstract] Abstract: the Frost-Musulin parameters are fitted quantities chosen to reproduce spectroscopic data, but the manuscript supplies no independent check that the computed E(v,J) levels recover experimental spectroscopic constants to the precision required for the Gibbs free energy claim; this leaves open the possibility that the reported accuracy is partly circular.
minor comments (1)
- [Abstract] Abstract: the phrase 'inelastic rotational, continuity contributions' appears to be a typographical error and should read 'inelastic rotational and continuum contributions'.
Simulated Author's Rebuttal
We thank the referee for the careful and constructive review. We address each major comment point by point below and indicate the revisions we will make to strengthen the quantitative support and validation in the manuscript.
read point-by-point responses
-
Referee: [Abstract] Abstract: the claim that the formulation 'captures the Gibbs free energy deviation function for both molecules with high quantitative accuracy' is unsupported by any numerical tables, error metrics, direct comparisons to experimental thermochemical data, or reference calculations; without these, the accuracy assertion cannot be evaluated.
Authors: We agree that the abstract claim requires explicit quantitative backing for proper evaluation. The full manuscript presents graphical comparisons, but we will add a dedicated table in the results section listing Gibbs free energy deviation values against experimental thermochemical data for H2 and LiH, together with RMS errors and mean percentage deviations over the studied temperature range. This will directly substantiate the accuracy statement. revision: yes
-
Referee: [Abstract] Abstract and methods description of the Pekeris approximation: the approximation replaces the centrifugal term by a first-order expansion about r_e and is stated to yield a 'validated' spectrum, yet no assessment of its accuracy for high-v, high-J levels (which dominate the partition function at elevated T) or comparison to exact numerical eigenvalues is provided; this directly affects whether residual high-T errors can be attributed solely to neglected dissociation.
Authors: The referee is correct that an explicit accuracy assessment for high-v, high-J states is missing. In the revised manuscript we will include a new subsection comparing the Pekeris-approximated eigenvalues to numerically exact solutions of the radial Schrödinger equation for representative high-v and high-J states up to the energies relevant for the partition function at 2000 K. This will quantify the approximation error and reinforce our attribution of high-T deviations to dissociation and inelastic channels. revision: yes
-
Referee: [Abstract] Abstract: the Frost-Musulin parameters are fitted quantities chosen to reproduce spectroscopic data, but the manuscript supplies no independent check that the computed E(v,J) levels recover experimental spectroscopic constants to the precision required for the Gibbs free energy claim; this leaves open the possibility that the reported accuracy is partly circular.
Authors: We acknowledge the potential circularity concern. The parameters are taken from literature fits, yet the analytical spectrum with the Pekeris approximation must still be shown to recover the constants faithfully. We will add to the revised manuscript a direct comparison of the vibrational frequency ω_e and rotational constant B_e extracted from our computed E(v,J) levels against the experimental spectroscopic constants for both molecules, thereby demonstrating that the thermochemical accuracy arises from the model's physical fidelity rather than fitting alone. revision: yes
Circularity Check
No significant circularity; derivation is standard model-to-thermo chain
full rationale
The paper solves the radial Schrödinger equation for the Frost-Musulin potential using the Pekeris approximation to generate bound-state energies, inserts those energies into the rovibrational partition function together with standard translational and rotational ideal-gas factors, and computes thermodynamic functions including the Gibbs free-energy deviation. This is a conventional forward calculation from a chosen potential model to observables; no equation or step is shown to be definitionally identical to its input, no fitted parameter is relabeled as an independent prediction, and no load-bearing uniqueness theorem or ansatz is imported solely via self-citation. The abstract's claim of quantitative accuracy is therefore an empirical comparison against external data rather than a tautology.
Axiom & Free-Parameter Ledger
free parameters (1)
- Frost-Musulin potential parameters
axioms (2)
- standard math The radial Schrödinger equation with central potential admits an analytical bound-state solution under the Pekeris approximation near equilibrium.
- domain assumption Translational and rotational contributions to the partition function can be treated with classical ideal-gas statistics.
Reference graph
Works this paper leans on
-
[1]
( 23) are used
levels generated by Eq. ( 23) are used. In the numerical implementation, the vibrational zero of energy is taken at the ground state and the summation is truncated at the highest bound level satisfying En0 < De. Accordingly , the vibrational partition function is evaluated directly as qvib(T ) = Nb ∑ n=0 exp[− β (En0 − E00)] , β = 1 kBT , (24) where Nb de...
-
[2]
Table I: Spectroscopic molecular parameters for H 2, and LiH diatomic molecules
is evaluated directly in the ana- lytically obtained energy eigenvalues. Table I: Spectroscopic molecular parameters for H 2, and LiH diatomic molecules. Molecule De re ν e µ α [eV] [Å] [cm− 1] [a.m.u.] [Å− 1] H2 4.74460 0.7416 4395.2 0.50391 1.711487215 LiH 2.51508 1.5955990 1405.498 0.88013 1.086430866 III. Results and Discussion The spectroscopic const...
2000
-
[3]
Wang, C.-S
J. Wang, C.-S. Jia, C.-J. Li, X.-L. Peng, L.-H. Zhang, and J.- Y. Liu, ACS Omega 4, 19193 (2019)
2019
-
[4]
Khordad, A
R. Khordad, A. Avazpour , and A. Ghanbari, Chem. Phys. 517, 30 (2019)
2019
-
[5]
Ghanbari, R
A. Ghanbari, R. Khordad, and M. Sharifzadeh, Physica B 678, 415750 (2024)
2024
-
[6]
Ghanbari and S
A. Ghanbari and S. M. Jadbaba, J. Low T emp. Phys. 211, 109 (2023)
2023
-
[7]
Ghanbari and R
A. Ghanbari and R. Khordad, Indian J. Phys. 96, 1413 (2022)
2022
-
[8]
C.-S. Jia, J. Li, Y.-S. Liu, X.-L. Peng, X. Jia, L.-H. Zhang, R . Jiang, X.-P. Li, J.-Y. Liu, and Y.-L. Zhao, J. Mol. Liq. 315, 113751 (2020)
2020
-
[9]
Servatkhah, R
M. Servatkhah, R. Khordad, and A. Ghanbari, Int. J. Thermophys. 41, 37 (2020)
2020
-
[10]
Okorie, A
U. Okorie, A. Ikot, E. Chukwuocha, and G. Rampho, Results Phys. 17, 103078 (2020)
2020
-
[11]
Valencia-Ortega and L.-A
G. Valencia-Ortega and L.-A. Arias-Hernandez, Int. J Quantum. Chem. 118, e25589 (2018)
2018
-
[12]
Khordad, C
R. Khordad, C. Edet, and A. Ikot, Int. J. Mod. Phys. C 33, 2250106 (2022)
2022
-
[13]
Habibinejad and A
M. Habibinejad and A. Ghanbari, Eur . Phys. J. Plus 136, 400 (2021)
2021
-
[14]
Ghanbari and R
A. Ghanbari and R. Khordad, Chem. Phys. 534, 110732 (2020)
2020
-
[15]
Khordad and A
R. Khordad and A. Ghanbari, J. Low T emp. Phys. 199, 1198 (2020)
2020
-
[16]
A. N. Ikot, U. S. Okorie, R. Sever , and G. J. Rampho, Eur . Phys. J. Plus 134, 386 (2019)
2019
-
[17]
Qiang and S.-H
W.-C. Qiang and S.-H. Dong, Phys. Lett. A 368, 13 (2007)
2007
-
[18]
Dong and M
S.-H. Dong and M. Cruz-Irisson, J. Math. Chem. 50, 881 (2012)
2012
-
[19]
S.-H. Dong, M. Lozada-Cassou, J. Yu, F. Jiménez-Ángeles, an d A. L. Rivera, Int. J. Quantum Chem. 107, 366 (2007)
2007
-
[20]
Wei, S.-H
G.-F. Wei, S.-H. Dong, and V. B. Bezerra, Int. J. Mod. Phys. A 24, 161 (2009)
2009
-
[21]
Zhang, G.-H
M.-C. Zhang, G.-H. Sun, and S.-H. Dong, Phys. Lett. A 374, 704 (2010)
2010
-
[22]
M. G. Miranda, G.-H. Sun, and S.-H. Dong, Int. J. Mod. Phys. E 19, 123 (2010)
2010
-
[23]
Y. P. Varshni, Rev . Mod. Phys.29, 664 (1957)
1957
-
[24]
J. P. Araújo and M. Y. Ballester , Int. J. Quantum Chem. 121, e26808 (2021)
2021
-
[25]
V. H. Badalov and S. V. Badalov , Commun. Theor . Phys. 75, 075003 (2023)
2023
-
[26]
H. I. Ahmadov , E. A. Dadashov , N. S. Huseynova, and V. H. Badal ov , Eur . Phys. J. Plus 136, 244 (2021)
2021
-
[27]
Ahmed, EPL 141, 25003 (2023)
F. Ahmed, EPL 141, 25003 (2023)
2023
-
[28]
C. Edet, K. O. Okorie, H. Louis, and N. A. Nzeata-Ibe, Indian J. Phys. 94, 243 (2020)
2020
-
[29]
U. S. Okorie, A. N. Ikot, M. C. Onyeaju, and E. O. Chukwuocha, J. Mol. Model. 24, 289 (2018)
2018
-
[30]
A. V. Balakrishnan, Applied Functional Analysis (Springer , 2012)
2012
-
[31]
T ezcan and R
C. T ezcan and R. Sever ,Int. J. Theor . Phys. 48, 337 (2009)
2009
-
[32]
Abramowitz and I
M. Abramowitz and I. A. Stegun, eds., Handbook of Mathematical Functions with Formulas, Graphs, and Mathematical T ables, 9th ed. (Dover , New Y ork, 1964)
1964
-
[33]
A. A. Frost and B. Musulin, J. Chem. Phys. 22, 1017 (1954)
1954
-
[34]
Khordad, Phys
R. Khordad, Phys. B: Condens. Matter 690, 416297 (2024)
2024
-
[35]
Khordad, Opt
R. Khordad, Opt. Quant. Electron. 56, 596 (2024)
2024
-
[36]
Khordad, Opt
R. Khordad, Opt. Quant. Electron. 56, 963 (2024)
2024
-
[37]
M. W. Schmidt, J. Ivanic, and K. Ruedenberg, J. Chem. Phys. 140, 204104 (2014)
2014
-
[38]
Martin Pendas, D
A. Martin Pendas, D. J. L. Rodriges, and E. Francisco, Phys. Chem. Chem. Phys. 27, 1789 (2025)
2025
-
[39]
T a¸ s,Phys
A. T a¸ s,Phys. Scr .98, 125404 (2023)
2023
-
[40]
Mustafa, Phys
O. Mustafa, Phys. Scr .90, 065002 (2015)
2015
-
[41]
K. K. Irikura, in Computational Thermochemistry (American Chemical Soci- ety) Chap. 22, pp. 402–418
-
[42]
Malcolm W
J. Malcolm W. Chase, NIST-JANAF Thermochemical T ables, Fourth Edition (Journal of Physical and Chemical Reference Data, Monograp h 9, 1998) pp. 1–1951
1998
-
[43]
P. J. Linstrom and W. G. Mallard, J. Chem. Eng. Data. 46, 1059 (2001) . 1-9
2001
-
[44]
National Institute of Standards and T echnology , Hydrogen: NIST chemistry webbook, SRD 69, https://webbook.nist.gov/cgi/cbook.cgi?ID=C1333740&Plot=on&Type=JANAFG (2026), accessed 2026-03-29
2026
-
[45]
National Institute of Standards and T echnology , Lithium hydride: NIST chemistry webbook, SRD 69, https://webbook.nist.gov/cgi/cbook.cgi?ID=C7580678&Mask=3FFF (2026), accessed 2026-03-29
2026
-
[46]
Spicher and S
S. Spicher and S. Grimme, J. Chem. Theory Comput. 17, 1701 (2021)
2021
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.