Recognition: unknown
A class and home problem on electrolyte transport: constant electric field implies electroneutrality, but electroneutrality does not imply a constant electric field
Pith reviewed 2026-05-09 19:58 UTC · model grok-4.3
The pith
Electroneutrality is necessary but not sufficient for a constant electric field in electrolytes.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
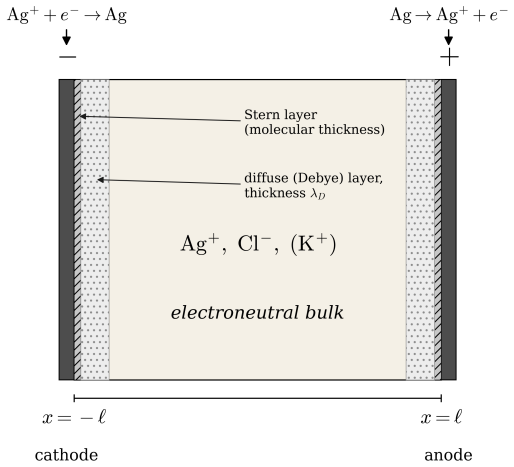
Starting with the one-dimensional Poisson-Nernst-Planck equations for a silver electroplating cell, concentration and potential profiles are obtained that satisfy the governing equations and boundary conditions. A companion problem with background electrolyte introduces an additional dimensionless ratio and admits a closed-form solution. The resulting profiles establish that electroneutrality is necessary but not sufficient for a constant electric field.
What carries the argument
The one-dimensional Poisson-Nernst-Planck equations applied to the silver electroplating cell, which couple ion concentration gradients to electric potential through Poisson's relation and the Nernst-Planck flux expressions.
Load-bearing premise
The one-dimensional Poisson-Nernst-Planck equations with the stated boundary conditions for the silver electroplating cell fully capture the relevant physics without convection, side reactions, or three-dimensional effects.
What would settle it
A direct measurement of the electric field inside the silver electroplating cell that remains constant in a region where local electroneutrality does not hold would falsify the necessity part of the claim.
Figures



read the original abstract
We present a class and home problem in graduate transport phenomena and electrochemical engineering that clarifies a common misconception: electroneutrality implies the electric field is constant. Starting with one-dimensional Poisson--Nernst--Planck equations for a silver electroplating cell, students obtain concentration and potential profiles. A companion home problem with added background electrolyte introduces a new dimensionless ratio and admits a closed-form solution. Students conclude that electroneutrality is necessary but not sufficient for a constant electric field.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript presents a pedagogical class and home problem based on the one-dimensional Poisson-Nernst-Planck equations for a silver electroplating cell. Students derive concentration and potential profiles from the governing equations; a companion home problem adds supporting electrolyte, introduces a new dimensionless ratio, and admits a closed-form solution. The central claim is that a constant electric field implies electroneutrality (via Poisson's equation) but the converse does not hold, as electroneutrality is necessary but not sufficient for a constant field when background electrolyte is present.
Significance. If the derivations hold, this provides a clear, concrete teaching example that distinguishes two concepts often conflated in electrolyte transport. The closed-form solution with supporting electrolyte is a particular strength, allowing students to see explicitly how small nonzero charge density permits field variation while satisfying steady-state current conservation. The work ships explicit solutions and a falsifiable distinction under standard 1D PNP boundary conditions, which is valuable for graduate courses in transport phenomena and electrochemical engineering.
minor comments (2)
- [Abstract] The abstract states that the home problem 'introduces a new dimensionless ratio' but does not give its explicit form (e.g., ratio of supporting-electrolyte to reacting-ion concentration); adding the definition would improve immediate readability.
- [Home-problem section] Boundary conditions for the closed-form solution in the home problem are referenced but not written out; listing them explicitly (e.g., the values of flux or potential at the electrodes) would allow readers to verify the analytic profiles without ambiguity.
Simulated Author's Rebuttal
We thank the referee for their supportive review and recommendation of minor revision. The referee's summary correctly captures the manuscript's focus on using the one-dimensional Poisson-Nernst-Planck equations to illustrate that electroneutrality is necessary but not sufficient for a constant electric field, and we appreciate the recognition of the closed-form solution with supporting electrolyte as a particular strength.
Circularity Check
No significant circularity; derivation follows directly from standard PNP equations
full rationale
The paper constructs a class problem by solving the one-dimensional Poisson-Nernst-Planck equations for a silver electroplating cell and a companion home problem with added supporting electrolyte that yields a closed-form solution. The central claim (electroneutrality necessary but not sufficient for constant E) follows immediately from the Poisson equation (constant E iff rho=0) combined with steady-state current conservation in the Nernst-Planck fluxes, which permit small nonzero rho and varying E. No fitted parameters are renamed as predictions, no self-citations are load-bearing, and no ansatz or uniqueness theorem is smuggled in. The result is self-contained against the governing equations without reduction to its own inputs by construction.
Axiom & Free-Parameter Ledger
Reference graph
Works this paper leans on
-
[1]
Electrification and decarbonization of the chemical industry,
Z. J. Schiffer and K. Manthiram, “Electrification and decarbonization of the chemical industry,” Joule, vol. 1, no. 1, pp. 10–14, 2017
2017
-
[2]
W. M. Deen,Analysis of Transport Phenomena. New York, NY: Oxford Univ. Press, 2nd ed., 2012
2012
-
[3]
A. J. Bard, L. R. Faulkner, and H. S. White,Electrochemical Methods: Fundamentals and Applications. Hoboken, NJ: Wiley, 3rd ed., 2022
2022
-
[4]
Newman and K
J. Newman and K. E. Thomas-Alyea,Electrochemical Systems. Hoboken, NJ: Wiley, 3rd ed., 2004
2004
-
[5]
Current-voltage relations for electrochemical thin films,
M. Z. Bazant, K. T. Chu, and B. J. Bayly, “Current-voltage relations for electrochemical thin films,”SIAM J. Appl. Math., vol. 65, no. 5, pp. 1463–1484, 2005
2005
-
[6]
Ion transport in an electrochemical cell: a theoretical framework to couple dynamics of double layers and redox reactions for multicomponent electrolyte solutions,
N. Jarvey, F. Henrique, and A. Gupta, “Ion transport in an electrochemical cell: a theoretical framework to couple dynamics of double layers and redox reactions for multicomponent electrolyte solutions,”J. Electrochem. Soc., vol. 169, no. 9, p. 093506, 2022
2022
-
[7]
A lithium-ion battery model including electrical double layer effects,
J. Marcicki, A. T. Conlisk, and G. Rizzoni, “A lithium-ion battery model including electrical double layer effects,”J. Power Sources, vol. 251, pp. 157–169, 2014. 11
2014
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.