Recognition: 3 theorem links
· Lean TheoremDiffusio-osmotic transport in nanochannels
Pith reviewed 2026-05-08 18:30 UTC · model grok-4.3
The pith
Diffusio-osmosis shows that osmotic flows can occur in nanochannels without any semi-permeable membrane by treating both osmosis and diffusio-osmosis as the same entropic process.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Osmosis and diffusio-osmosis are two faces of the same phenomenon, naturally embedded in an Onsager framework and quantified by local and global force balances. When diffusio-osmosis is applied to nanochannels, osmotic drivings appear without the prerequisite of semi-permeability, thereby extending the domain of entropically driven transport to a broader class of channels and membranes.
What carries the argument
The diffusio-osmotic mechanism, arising from entropic driving forces inside diffuse layers at solid boundaries and quantified by local and global force balances in an Onsager framework.
If this is right
- Osmotic drivings arise in channels and membranes without semi-permeability.
- Diffusio-osmosis produces enhanced diffusion under concentration gradients.
- The same mechanism yields mechano-sensitivity in transport through nanochannels.
- Rectified osmotic flows appear when geometry or boundary conditions break symmetry.
- Diffusio-osmosis can be used as a lever for osmotic energy conversion from single nanopores to larger membrane modules.
Where Pith is reading between the lines
- Design of artificial osmotic devices could drop the requirement for perfect membrane selectivity and rely instead on controlled surface diffuse layers.
- Symmetric nanochannels with tunable wall properties offer a clean experimental test bed for isolating the predicted velocity from concentration gradients alone.
- Similar force-balance reasoning may connect diffusio-osmosis to other interface flows such as thermo-osmosis when multiple gradients are present simultaneously.
- Scaling the single-pore results to modules suggests a route to osmotic power that tolerates imperfect selectivity at the cost of lower efficiency per pore.
Load-bearing premise
Entropic driving forces inside the diffuse layers next to solid boundaries can be mapped directly onto local and global force balances inside an Onsager framework.
What would settle it
A measurement of zero net fluid velocity inside a nanochannel subjected to a controlled concentration gradient, when surface charge and ion mobility are known and the predicted diffusio-osmotic velocity is nonzero, would falsify the central claim.
Figures






read the original abstract
In this chapter, I will enter into the roots of entropically-driven transport with a focus on diffusio-osmotic transport in nanochannels. Diffusio-osmosis is a subtle surface transport, originating in entropic driving forces occuring within the diffuse layers at solid boundaries. Specifying diffusio-osmosis to nanochannels may first look like a marginal refinement, yet it reveals that osmotic drivings can arise in channels and membranes without the prerequisite of semi-permeability, so that diffusio-osmosis extends the domain of existence of entropically driven transport. Osmosis and diffusio-osmosis are two faces of the same phenomenon, naturally embedded in an Onsager framework and quantified by local and global force balances. This perspective clarifies why nanochannels are privileged arenas where diffusio-osmosis and its consequence do flourish. Throughout the chapter, I discuss a set of conceptually relevant examples to show how diffusio-osmosis "pops up" in various situations: as enhanced diffusion, mechano-sensitivity, rectified osmotic flows and, ultimately, as a lever for osmotic energy conversion from single nanopores to membrane modules approaching industrial reality.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. This perspective chapter synthesizes concepts of entropically-driven transport with a focus on diffusio-osmosis in nanochannels. It claims that osmosis and diffusio-osmosis are two faces of the same phenomenon, naturally embedded in an Onsager framework and quantified by local and global force balances. Specifying the phenomenon to nanochannels reveals that osmotic drivings can arise without semi-permeability, thereby extending the domain of entropically driven transport. The chapter discusses examples including enhanced diffusion, mechano-sensitivity, rectified osmotic flows, and osmotic energy conversion from single nanopores to membrane modules approaching industrial scale.
Significance. If the framing holds, the manuscript offers a coherent conceptual unification of osmosis and diffusio-osmosis grounded in established thermodynamic relations. It explicitly credits the absence of free parameters, ad-hoc axioms, or invented entities, relying instead on standard Onsager relations from prior literature. This synthesis clarifies the privileged role of nanochannels for such transport and has implications for applications in osmotic energy conversion. The interpretive approach provides a parameter-free view that could guide understanding in nanofluidics without introducing new mathematical inconsistencies.
minor comments (1)
- [Abstract] Abstract: 'occuring' is a typo and should be corrected to 'occurring'.
Simulated Author's Rebuttal
We thank the referee for the positive summary and significance assessment of our perspective chapter on diffusio-osmotic transport in nanochannels. The recommendation for minor revision is noted; we are prepared to incorporate any specific minor suggestions in the revised manuscript.
Circularity Check
No significant circularity detected
full rationale
The manuscript is a perspective chapter that synthesizes established concepts of entropically driven transport, framing osmosis and diffusio-osmosis within standard Onsager relations and local/global force balances drawn from prior literature. No new mathematical derivations, fitted parameters, or falsifiable predictions are advanced that reduce by construction to inputs internal to the paper; the central interpretive claim extends known diffuse-layer physics to nanochannels without introducing self-definitional steps, fitted-input predictions, or load-bearing self-citation chains. The framework remains self-contained against external benchmarks of classical irreversible thermodynamics.
Axiom & Free-Parameter Ledger
axioms (1)
- standard math Onsager reciprocal relations apply to the coupled transport phenomena described
Lean theorems connected to this paper
-
Cost.FunctionalEquation (washburn_uniqueness_aczel)washburn_uniqueness_aczel uncleara linear relation can be written between thermodynamic forces and fluxes ... the transport matrix is symetric and positive definite according to the Onsager principle
Reference graph
Works this paper leans on
-
[1]
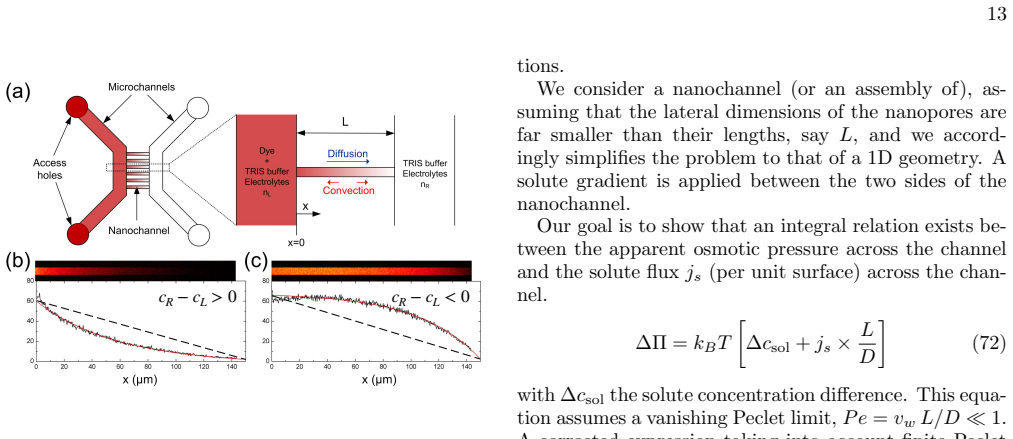
Apparent osmotic pressure Let us focus on a situation where a salt concentra- tion difference is applied across the channel,i.e.c R ext = cs + ∆cs,c L ext =c s (RandLstands for the right and left reservoirs). The concentration gradient in the reser- voirs yields a concentration difference inside the channel, but also an electric potential difference and a...
-
[2]
The integral relation As an alternative approach, we may consider the inte- gral approach from equation in Eq.(79), which expresses the apparent osmotic pressure in terms of the ionic flux js. In the present overlapping regime of the capillary pore model, the ionic fluxj s can be readily computed from from Eq.(110) and the Donnan expressions in Eqs.(112)-...
-
[3]
rejection
Apparent osmotic pressure from the Onsager symmetry From the previous calculation, we obtained that the water flow under a salt concentration gradient rewrites Qw =L w ×(−σ(Du)c s)× −2k BT∆logc s (125) with the negative of the “rejection” coefficient given by −σ(Du) = √ 1 +Du 2 −1 andc s is the bulk reservoir concentration. This result can also be infered...
-
[4]
Sinceλ≪h, the flow identifies with that on a flat surface which was discussed in Sec
Solvent DO velocity and ion fluxes Let us assume that there is a local concentration gradi- ent along the nanochannel,c 0(x), with the concentration c0(x) defined in the middle of the channel cross section (see below). Sinceλ≪h, the flow identifies with that on a flat surface which was discussed in Sec. III. Tak- ing a distancezperpendicular to the pore s...
-
[5]
standard
Diffusio-osmotic ionic currents An interesting consequence of diffusio-osmosis is the emergence of diffusio-osmotic ionic currents induced by a gradient of salt concentration. Again, current in- duced by salinty gradients is usually expected for selec- tive nanochannels/membranes, such as cation- or anion- exchange membranes (CEM/AEM). However, diffusio- ...
2013
-
[6]
1D-PNP equations integrating the diffuse layer contribution In the thin diffuse layer regime, there is a separation of length scales between the Electric Double Layer and the characteristic scales over which the geometry varies laterally (e.g.pore length). One can therefore integrate over the EDL in order to separate explicity the scales and derive one-di...
-
[7]
Non-convective contributions Let me first ignore the convection effects (terms pro- portional to the fluid velocity), which we will estimate hereafter. Using Eq.(94), and identifying thatc v =c s the value of the salt concentration outside the EDL, one get the cross-sectional integrals of the non-convective contri- butions to the solute flux and ionic den...
-
[8]
These are slightly more subtle to describe
Convective effects in the thin diffuse layer regime Now, one should add the convective contribution, which stem from the ion transport by the solvent flow. These are slightly more subtle to describe. This follows from the analysis developped in the previous sections, as well as for the capillary pore model. Gathering the pre- vious results, the Stokes equ...
-
[9]
Osmotic Transport across Surface Functionalized Carbon Nanotube Membrane
Combining all elements... Gathering results in Eqs.(143), (145) & (148) we have obtained the complete expression of the local transport matrix. Differences of pressure/concentration/voltage are applied at the two ends of the channel will in- duce pressure/concentration/voltage profiles inside the nanochannel, whose equations obeys the conservation laws ab...
-
[10]
But observations in Ref
Simple estimates Fick diffusion is associated with a flux of salt pro- portional to the salt concentration gradient, asJ Fick s = −D 2∆cs L (remembering that the total ion concentration is twice the salt concentration). But observations in Ref
-
[11]
It is easy to reconcile this observation with the effect of diffusio-osmosis
show an enhanced salt diffusion at low concentration, see Fig.5. It is easy to reconcile this observation with the effect of diffusio-osmosis. According to the various de- scriptions above, a DO flow will be induced under the concentration gradient obeying vDO =D DO × −∆ logc s L (149) where the DO mobility has the dimension of a diffusion coefficient. Fo...
-
[12]
However it misses some subtleties of the inner transport
More insights into the inner transport The above approach gives the main trends for the effect of DO on diffusive transport. However it misses some subtleties of the inner transport. This is what I briefly discuss here. The diffusio-osmotic flow is expected to flush the con- centration profiles, hence affecting the concentration gra- dient along the nanoc...
2018
-
[13]
Architecting mechanosensi- tive nanofluidic transport in graphite nanoslits
Beyond: consequences on the conductance The detailed response under a voltage drop ∆Vfollows the same type of analysis. We leave the full analysis to the reader. To my knowledge it has not been exhaustively performed. I rather explore here the effect of DO on the conductance, which is the linear response to ∆V: I=G R(−∆V) In a simple analysis, the conduct...
-
[14]
Charge contrast as a source of mechano-sensitivity The origin of the phenomenon is the charge discon- tinuity at the entrance of the nanochannel, which is at the origin of a cascade of transport couplings. In a few words, the ion in the EDL close to the activated surface are flushed towards (or away from) the nanochannel but entering the nanochannels thes...
-
[15]
Nanofluidic Osmotic Diodes: Theory and Molecular Dynamics Simulations
The cherry on the cake: a huge diffusio-osmotic boost The approach above however misses an important in- gredient. Indeed the created salinity gradient inside the nanochannel under flow is expected to induce a diffusio- osmotic velocity,v DO =−K DO ×(c s(L)−c s(0)) with KDO =D DO/cs. In the presence of slippage with b≫λ D, this yieldsv DO =− kB T η λDb×(c...
2025
-
[16]
This mix- ing process is associated with a substantial entropy gain, corresponding to an ideal extractable energy of about 0.8 kWh per cubic metre of freshwater mixed with seawater
Osmotic energy, a quick recap Osmotic, or “blue”, energy denotes the free energy that is released when solutions of different salinity mix, for example where rivers discharge into the sea. This mix- ing process is associated with a substantial entropy gain, corresponding to an ideal extractable energy of about 0.8 kWh per cubic metre of freshwater mixed w...
1954
-
[17]
Diffusio-osmosis as an alternative lever for osmotic energy harvesting The concept of diffusio-osmosis has changed the per- spective for osmotic energy conversion by showing that it does not require strict molecular selectivity as previ- ously conceived and can instead be driven by interfacially controlled mechanisms such as diffusio-osmosis. This di- rec...
-
[18]
nanofluidic-informed
Scaling up nanofluidics Exploiting such surface-driven transport in highly charged nanopores actually provides a route to circum- vent the conventional permeability–selectivity compro- mise and to boost osmotic power densities as compared with PRO and RED. Interestingly, while existing PRO and RED plant are highly optimized in terms of engi- neering, the ...
-
[19]
J. L. Anderson,Annual review of fluid mechanics, 1989, 21, 61–99
1989
-
[20]
Marbach and L
S. Marbach and L. Bocquet,Chemical Society Reviews, 2019,48, 3102–3144
2019
-
[21]
Shim,Chemical Reviews, 2022,122, 6986–7009
S. Shim,Chemical Reviews, 2022,122, 6986–7009
2022
-
[22]
Derjaguin, G
B. Derjaguin, G. Sidorenkov, E. Zubashchenko and E. Kiseleva,Progress in Surface Science, 1993,43, 138– 152
1993
-
[23]
Bacchin, K
P. Bacchin, K. Glavatskiy and V. Gerbaud,Physical Chemistry Chemical Physics, 2019,21, 10114–10124
2019
-
[24]
Ab´ ecassis, C
B. Ab´ ecassis, C. Cottin-Bizonne, C. Ybert, A. Ajdari and L. Bocquet,Nature materials, 2008,7, 785
2008
-
[25]
Ab´ ecassis, C
B. Ab´ ecassis, C. Cottin-Bizonne, C. Ybert, A. Ajdari and L. Bocquet,New Journal of Physics, 2009,11, 075022
2009
-
[26]
Palacci, C
J. Palacci, C. Cottin-Bizonne, C. Ybert and L. Bocquet, Soft Matter, 2012,8, 980–994
2012
-
[27]
Bocquet,Nature materials, 2020,19, 254–256
L. Bocquet,Nature materials, 2020,19, 254–256
2020
-
[28]
Kavokine, R
N. Kavokine, R. R. Netz and L. Bocquet,Annual Review of Fluid Mechanics, 2021,53, 377–410
2021
-
[29]
Siria, P
A. Siria, P. Poncharal, A.-L. Biance, R. Fulcrand, X. Blase, S. T. Purcell and L. Bocquet,Nature, 2013, 494, 455
2013
-
[30]
J. Feng, M. Graf, K. Liu, D. Ovchinnikov, D. Dumcenco, M. Heiranian, V. Nandigana, N. R. Aluru, A. Kis and A. Radenovic,Nature, 2016,536, 197
2016
-
[31]
Emmerich, K
T. Emmerich, K. S. Vasu, A. Nigu` es, A. Keerthi, B. Radha, A. Siria and L. Bocquet,Nature materials, 2022,21, 696–702
2022
-
[32]
C. Lee, C. Cottin-Bizonne, A.-L. Biance, P. Joseph, L. Bocquet and C. Ybert,Physical Review Letters, 2014, 112, 244501
2014
-
[33]
C. Lee, C. Cottin-Bizonne, R. Fulcrand, L. Joly and C. Ybert,The journal of physical chemistry letters, 2017,8, 478–483
2017
-
[34]
S. R. De Groot and P. Mazur,Non-equilibrium thermo- dynamics, Courier Corporation, 2013
2013
-
[35]
Herrero, M
C. Herrero, M. De San F´ eliciano, S. Merabia and L. Joly, Nanoscale, 2022,14, 626–631
2022
-
[36]
Ganti, Y
R. Ganti, Y. Liu and D. Frenkel,Physical review letters, 2017,119, 038002
2017
-
[37]
Kedem and A
O. Kedem and A. Katchalsky,The Journal of general physiology, 1961,45, 143–179
1961
-
[38]
G. S. Manning and A. R. Kay,Journal of General Phys- 34 iology, 2023,155, e202313332
2023
-
[39]
K. S. Pitzer,The Journal of physical chemistry, 1973, 77, 268–277
1973
-
[40]
K. S. Pitzer and G. Mayorga,The Journal of Physical Chemistry, 1973,77, 2300–2308
1973
-
[41]
Debye,Recueil des Travaux Chimiques des Pays-Bas, 1923,42, 597–604
P. Debye,Recueil des Travaux Chimiques des Pays-Bas, 1923,42, 597–604
1923
-
[42]
G. S. Manning,The Journal of Chemical Physics, 1968, 49, 2668–2675
1968
-
[43]
Marbach, H
S. Marbach, H. Yoshida and L. Bocquet,The Journal of Chemical Physics, 2017,146, 194701
2017
-
[44]
Yoshida, S
H. Yoshida, S. Marbach and L. Bocquet,The Journal of Chemical Physics, 2017,146, 194702
2017
-
[45]
Marbach, N
S. Marbach, N. Kavokine and L. Bocquet,The Journal of Chemical Physics, 2020,152, 054704
2020
-
[46]
C. B. Picallo, S. Gravelle, L. Joly, E. Charlaix and L. Bocquet,Physical Review Letters, 2013,111, 244501
2013
-
[47]
Prieve, J
D. Prieve, J. Anderson, J. Ebel and M. Lowell,Journal of Fluid Mechanics, 1984,148, 247–269
1984
-
[48]
B. J. Kirby and E. F. Hasselbrink Jr,Electrophoresis, 2004,25, 187–202
2004
-
[49]
D. M. Huang, C. Cottin-Bizonne, C. Ybert and L. Boc- quet,Physical review letters, 2007,98, 177801
2007
-
[50]
L. Joly, R. H. Meißner, M. Iannuzzi and G. Tocci,ACS nano, 2021,15, 15249–15258
2021
-
[51]
Bocquet and E
L. Bocquet and E. Charlaix,Chemical Society Reviews, 2010,39, 1073–1095
2010
-
[52]
N. R. Aluru, F. Aydin, M. Z. Bazant, D. Blankschtein, A. H. Brozena, J. P. de Souza, M. Elimelech, S. Faucher, J. T. Fourkas, V. B. Komanet al.,Chemical reviews, 2023,123, 2737–2831
2023
-
[53]
Kavokine, M.-L
N. Kavokine, M.-L. Bocquet and L. Bocquet,Nature, 2022,602, 84–90
2022
-
[54]
Ajdari and L
A. Ajdari and L. Bocquet,Physical Review Letters, 2006,96, 186102
2006
-
[55]
L. Joly, C. Ybert, E. Trizac and L. Bocquet,The Jour- nal of chemical physics, 2006,125, 204716
2006
-
[56]
Y. Xie, L. Fu, T. Niehaus and L. Joly,Physical review letters, 2020,125, 014501
2020
-
[57]
Mouterde, A
T. Mouterde, A. Keerthi, A. Poggioli, S. A. Dar, A. Siria, A. K. Geim, L. Bocquet and B. Radha,Na- ture, 2019,567, 87–90
2019
- [58]
-
[59]
Architecting mechanosensitive nanofluidic transport in graphite nanoslits
M. Lizee, Z. Zhang, B. Coquinot, Q. Yang and L. Boc- quet,under review; arXiv preprint ArXiv: 2605.00661, 2026
work page internal anchor Pith review Pith/arXiv arXiv 2026
-
[60]
Ybert, C
C. Ybert, C. Barentin, C. Cottin-Bizonne, P. Joseph and L. Bocquet,Physics of fluids, 2007,19, 123601
2007
-
[61]
J. Ou, B. Perot and J. P. Rothstein,Physics of fluids, 2004,16, 4635–4643
2004
-
[62]
D. M. Huang, C. Cottin-Bizonne, C. Ybert and L. Boc- quet,Physical Review Letters, 2008,101, 064503
2008
-
[63]
Macha, S
M. Macha, S. Marion, V. V. Nandigana and A. Raden- ovic,Nature Reviews Materials, 2019,4, 588–605
2019
-
[64]
D. J. Rankin, L. Bocquet and D. M. Huang,The Journal of chemical physics, 2019,151, 044705
2019
-
[65]
Baldock and D
H. Baldock and D. M. Huang,Physics of Fluids, 2025, 37, 022032
2025
-
[66]
Bilichenko, M
M. Bilichenko, M. Iannuzzi and G. Tocci,ACS nano, 2024,18, 24118–24127
2024
-
[67]
Yoshida, H
H. Yoshida, H. Mizuno, T. Kinjo, H. Washizu and J.- L. Barrat,The Journal of chemical physics, 2014,140, 214701
2014
-
[68]
Monet, M.-L
G. Monet, M.-L. Bocquet and L. Bocquet,The Journal of Chemical Physics, 2023,159, 014501
2023
-
[69]
J. L. Anderson and D. M. Malone,Biophysical journal, 1974,14, 957–982
1974
-
[70]
Hill,Quarterly reviews of biophysics, 1979,12, 67– 99
A. Hill,Quarterly reviews of biophysics, 1979,12, 67– 99
1979
-
[71]
Pendse, S
A. Pendse, S. Cetindag, P. Rehak, S. Behura, H. Gao, N. H. L. Nguyen, T. Wang, V. Berry, P. Kr´ al, J. Shanet al.,Advanced Functional Materials, 2021,31, 2009586
2021
-
[72]
Pascual, N
M. Pascual, N. Chapuis, S. Abdelghani-Idrissi, M.-C. Jullien, A. Siria and L. Bocquet,Energy & Environ- mental Science, 2023,16, 4539–4548
2023
-
[73]
Lokesh, S
M. Lokesh, S. K. Youn and H. G. Park,Nano letters, 2018,18, 6679–6685
2018
-
[75]
Secchi, S
E. Secchi, S. Marbach, A. Nigu` es, D. Stein, A. Siria and L. Bocquet,Nature, 2016,537, 210
2016
-
[76]
Marcotte, T
A. Marcotte, T. Mouterde, A. Nigu` es, A. Siria and L. Bocquet,Nature materials, 2020,19, 1057–1061
2020
-
[77]
R. J. Gross and J. Osterle,The Journal of chemical physics, 1968,49, 228–234
1968
-
[78]
Fair and J
J. Fair and J. Osterle,The Journal of Chemical Physics, 1971,54, 3307–3316
1971
-
[79]
Westermann-Clark and J
G. Westermann-Clark and J. Anderson,Journal of the Electrochemical Society, 1983,130, 839
1983
-
[80]
Peters, R
P. Peters, R. Van Roij, M. Z. Bazant and P. Biesheuvel, Physical review E, 2016,93, 053108
2016
-
[81]
Karnik, C
R. Karnik, C. Duan, K. Castelino, H. Daiguji and A. Majumdar,Nano letters, 2007,7, 547–551
2007
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.