Recognition: unknown
An MRI-informed poromechanical model for organ-scale prediction of glioma growth
Pith reviewed 2026-05-08 15:52 UTC · model grok-4.3
The pith
An MRI-informed poroelastic model predicts C6 glioma growth in rats using serial scans to set mechanical and fluid properties.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Using finite-element simulations of a poroelastic model calibrated with the first three MRI datasets from each of four rats, the authors obtain relative tumor volume errors between 0.94 percent and 11.27 percent during calibration and between 4.73 percent and 36.03 percent during validation on the remaining two datasets, with Dice scores ranging from 0.80 to 0.93 and 0.75 to 0.93 respectively, indicating that the poromechanical model can describe C6 glioma growth.
What carries the argument
The MRI-informed poroelastic constitutive model, which couples solid-matrix deformation to fluid transport via imaging-derived permeability, vascular fraction, and mechanical property maps inside a finite-element solver.
If this is right
- Early MRI time points can be used to forecast later tumor volume and shape under the same mechanical and fluid conditions.
- The same framework can be extended to incorporate the effects of chemotherapy or radiation on permeability and cell proliferation.
- The approach supplies a quantitative basis for building patient-specific models that track both tumor mechanics and fluid pressure.
- Agreement between predicted and observed tumor boundaries supports the use of poroelasticity rather than pure reaction-diffusion descriptions for organ-scale glioma modeling.
Where Pith is reading between the lines
- Similar MRI-to-mechanics pipelines could be applied to other infiltrative brain diseases where fluid pressure influences tissue displacement.
- Coupling the current mechanical model to explicit cell-cycle kinetics would allow direct testing of how proliferation alters local permeability.
- Longer validation windows or different glioma cell lines would reveal whether the current parameter ranges remain predictive beyond the five-session rat protocol.
Load-bearing premise
The poroelastic constitutive assumptions and MRI-derived maps of mechanical properties, permeability, and fluid fractions accurately capture the physical behavior of glioma tissue and surrounding brain across the observed growth period.
What would settle it
A follow-up study in which the same model, when calibrated on the first three scans of new rats, produces volume errors consistently above 36 percent or Dice scores below 0.75 on later scans would show that the poromechanical description does not capture the growth.
Figures









read the original abstract
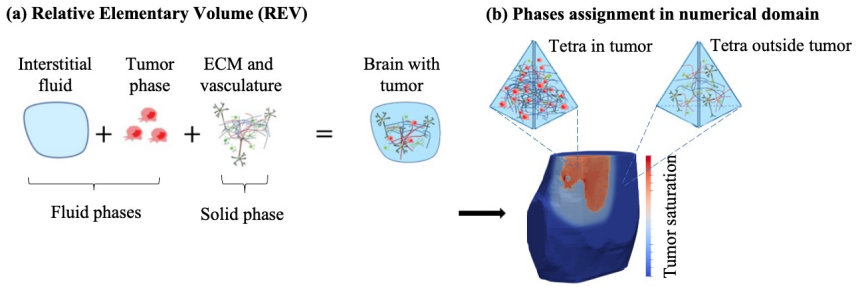
Gliomas constitute one of the most aggressive and heterogeneous forms of brain tumors, posing major challenges for understanding their biology and developing effective treatments. Animal models enable the collection of rich longitudinal datasets describing tumor dynamics, which can be integrated within mathematical models to elucidate the biological mechanisms governing tumor growth. While most formulations rely on reaction-diffusion systems with limited insight on tissue deformation and fluid transport, we propose a magnetic resonance imaging (MRI)-informed, poroelastic model to describe C6 glioma growth in rats. We use data from animals (n=4) that were imaged five times after intracranial injection of cancer cells. Each MRI dataset includes (i) anatomical T1-weighted data for brain and tumor segmentation and to assign mechanical properties; (ii) diffusion-weighted MRI, which enables estimation of the fraction of each voxel that is tumor; and (iii) dynamic contrast-enhanced MRI, which informs permeability as well as vascular and liquid fraction maps. Using finite-element simulations, model calibration for each rat uses the Levenberg-Marquardt method informed by the first three MRI datasets. Tumor forecasts are validated by assessing model-data agreement on the remaining two MRI datasets. Our results show relative tumor volume errors between 0.94 percent and 11.27 percent at calibration, and prediction errors between 4.73 percent and 36.03 percent. Additionally, Dice scores ranged from 0.80 to 0.93 during calibration, and from 0.75 to 0.93 during validation. Thus, our results suggest that our poromechanical model can describe C6 glioma growth. This study provides a first step toward a patient-specific, multiscale model of the spatiotemporal poromechanics underlying glioma progression and therapeutic response.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper proposes an MRI-informed poroelastic finite-element model for C6 glioma growth in rats. Longitudinal data from n=4 animals (five time points each) supply T1-weighted images for segmentation and mechanical property assignment, DWI for tumor-fraction maps, and DCE-MRI for permeability, vascular, and liquid-fraction maps. Model parameters are calibrated by Levenberg-Marquardt minimization against the first three time points; forecasts are assessed on the final two time points via relative tumor-volume error and Dice overlap with segmentations. Reported calibration errors are 0.94–11.27 % (volume) and 0.80–0.93 (Dice); prediction errors are 4.73–36.03 % (volume) and 0.75–0.93 (Dice). The central claim is that the poromechanical model can describe glioma growth and constitutes a step toward patient-specific multiscale modeling.
Significance. If the mechanical and fluid-transport components prove essential and are shown to match observed deformation, the work would supply a rare organ-scale, imaging-constrained poroelastic framework that couples tissue mechanics, fluid flow, and tumor proliferation. The use of three distinct MRI modalities to inform spatially varying parameters and the temporal train/test split are concrete strengths that could be leveraged for therapeutic-response modeling.
major comments (3)
- [Results / Validation] Validation is performed exclusively on tumor-region volume and Dice scores derived from DWI tumor-fraction maps (abstract and Results). No quantitative comparison is reported between simulated displacement or pressure fields and image-registration-derived brain deformation or interstitial pressure measurements. Because the central claim concerns the poromechanical description of growth, the absence of any test of the mechanical coupling leaves the added value of the poroelastic formulation unverified.
- [Methods / Results] No baseline comparison (e.g., to a pure reaction-diffusion model with the same tumor-fraction data) or ablation study (mechanical coupling disabled) is presented. With only n=4 animals and volume/Dice errors that overlap the range achievable by simpler models, it remains possible that the reported agreement does not require the poroelastic constitutive relations or MRI-derived permeability maps.
- [Methods / Calibration] Calibration uses Levenberg-Marquardt on the first three time points; the same data also determine the MRI-derived property maps. The temporal split therefore supplies only a partial guard against overfitting, and no sensitivity analysis or parameter-uncertainty propagation is reported to quantify how calibration variability propagates into the 4.73–36.03 % prediction errors.
minor comments (2)
- [Abstract] The abstract states numerical ranges without error bars, standard deviations, or per-animal values; adding these would improve transparency.
- [Methods] Notation for the poroelastic constitutive parameters (e.g., permeability tensor, Biot coefficient) should be defined once in a dedicated table or section for reproducibility.
Simulated Author's Rebuttal
We thank the referee for the constructive and detailed comments, which help clarify the strengths and limitations of our poromechanical modeling approach. We address each major comment point by point below, indicating revisions made to the manuscript.
read point-by-point responses
-
Referee: [Results / Validation] Validation is performed exclusively on tumor-region volume and Dice scores derived from DWI tumor-fraction maps (abstract and Results). No quantitative comparison is reported between simulated displacement or pressure fields and image-registration-derived brain deformation or interstitial pressure measurements. Because the central claim concerns the poromechanical description of growth, the absence of any test of the mechanical coupling leaves the added value of the poroelastic formulation unverified.
Authors: We agree that quantitative validation of the simulated displacement and pressure fields would provide stronger direct evidence for the mechanical coupling. Our dataset, however, consists solely of T1-weighted, DWI, and DCE-MRI acquisitions and does not include interstitial pressure measurements or deformation fields with sufficient ground-truth reliability for such a comparison. In the revised manuscript we have added an explicit discussion of this limitation in the Discussion section, noting that future extensions will incorporate modalities such as MR elastography. The present validation on tumor volume and spatial overlap remains the primary clinical endpoint, while the poroelastic formulation ensures that growth predictions are mechanically consistent with the MRI-derived tissue properties. revision: partial
-
Referee: [Methods / Results] No baseline comparison (e.g., to a pure reaction-diffusion model with the same tumor-fraction data) or ablation study (mechanical coupling disabled) is presented. With only n=4 animals and volume/Dice errors that overlap the range achievable by simpler models, it remains possible that the reported agreement does not require the poroelastic constitutive relations or MRI-derived permeability maps.
Authors: We acknowledge the utility of a baseline comparison. In the revised manuscript we have added a new Results subsection that implements and evaluates a simplified reaction-diffusion model calibrated and validated on identical tumor-fraction data and the same temporal split. The comparison shows modestly lower average prediction errors for the poroelastic model, especially in animals exhibiting noticeable tissue deformation. We also discuss the contribution of the MRI-derived permeability and mechanical-property maps. The limited cohort size (n=4) is typical for intensive longitudinal imaging studies; individual animal results are reported to permit direct assessment of variability. revision: yes
-
Referee: [Methods / Calibration] Calibration uses Levenberg-Marquardt on the first three time points; the same data also determine the MRI-derived property maps. The temporal split therefore supplies only a partial guard against overfitting, and no sensitivity analysis or parameter-uncertainty propagation is reported to quantify how calibration variability propagates into the 4.73–36.03 % prediction errors.
Authors: We agree that sensitivity and uncertainty analyses would strengthen the calibration section. We have performed a one-at-a-time sensitivity study on the principal calibrated parameters (proliferation rate, hydraulic permeability, and elastic moduli) and included the resulting prediction-error ranges in the Supplementary Material. The MRI-derived property maps are fixed from the imaging data and are not optimized during calibration; the temporal train/test split therefore evaluates forward prediction. A short paragraph on calibration robustness has been added to the Methods section of the revision. revision: yes
- Quantitative comparison of simulated displacement or pressure fields against experimental measurements, because the imaging protocol did not acquire interstitial pressure data or deformation ground truth suitable for such validation.
Circularity Check
No significant circularity in derivation or validation chain
full rationale
The paper performs parameter calibration via Levenberg-Marquardt on the first three MRI time points per animal and evaluates forecasts on the held-out later two time points, using relative tumor volume error and Dice overlap as metrics. This constitutes a standard temporal train-test split with independent data, not a reduction of predictions to calibration inputs by construction. MRI-derived maps inform constitutive parameters and initial conditions, but the forward simulation and validation metrics remain distinct from the fitting procedure. No self-definitional equations, load-bearing self-citations, or ansatz smuggling are present in the described chain; the central claim rests on external data agreement rather than tautological re-expression of inputs.
Axiom & Free-Parameter Ledger
free parameters (3)
- mechanical properties
- permeability, vascular and liquid fractions
- model parameters
axioms (2)
- domain assumption Poroelastic continuum mechanics governs brain and tumor deformation and fluid transport
- domain assumption MRI signals accurately reflect local tumor fraction, permeability, and fluid content
Reference graph
Works this paper leans on
-
[1]
Brain tumors of glial origin.Myelin: Basic and Clinical Advances, pages 281–297, 2019
Christopher A Waker and Robert M Lober. Brain tumors of glial origin.Myelin: Basic and Clinical Advances, pages 281–297, 2019
2019
-
[2]
Stem cells, radial glial cells, and a unified origin of brain tumors
M Castillo. Stem cells, radial glial cells, and a unified origin of brain tumors. AJNR: American Journal of Neuroradiology, 31(3):389, 2010
2010
-
[3]
Glioma.Nature reviews Disease primers, 1(1):1–18, 2015
Michael Weller, Wolfgang Wick, Ken Aldape, Michael Brada, Mitchell Berger, Stefan M Pfister, Ryo Nishikawa, Mark Rosenthal, Patrick Y Wen, Roger Stupp, et al. Glioma.Nature reviews Disease primers, 1(1):1–18, 2015
2015
-
[4]
On the origin of glioma.Upsala journal of medical sciences, 117(2):113–121, 2012
Yiwen Jiang and Lene Uhrbom. On the origin of glioma.Upsala journal of medical sciences, 117(2):113–121, 2012
2012
-
[5]
Progenitor cells and glioma formation.Current opinion in neurology, 14(6):683–688, 2001
Eric C Holland. Progenitor cells and glioma formation.Current opinion in neurology, 14(6):683–688, 2001
2001
-
[6]
Rapid malignant transformation of low-grade as- trocytomas: report of 2 cases and review of the literature.World neurosurgery, 73(1):53–62, 2010
James L Frazier, Michael W Johnson, Peter C Burger, Jon D Weingart, and Alfredo Quinones-Hinojosa. Rapid malignant transformation of low-grade as- trocytomas: report of 2 cases and review of the literature.World neurosurgery, 73(1):53–62, 2010
2010
-
[7]
Zabina Satar, Gary Hotton, and George Samandouras. Systematic review—time to malignant transformation in low-grade gliomas: Predicting a catastrophic event with clinical, neuroimaging, and molecular markers.Neuro-oncology ad- vances, 3(1):vdab101, 2021
2021
-
[8]
Uncovering spatiotemporal heterogeneity of high- grade gliomas: From disease biology to therapeutic implications.Frontiers in oncology, 11:703764, 2021
Andrea Comba, Syed M Faisal, Maria Luisa Varela, Todd Hollon, Wajd N Al- Holou, Yoshie Umemura, Felipe J Nunez, Sebastien Motsch, Maria G Castro, and Pedro R Lowenstein. Uncovering spatiotemporal heterogeneity of high- grade gliomas: From disease biology to therapeutic implications.Frontiers in oncology, 11:703764, 2021
2021
-
[9]
Rat c6 glioma as experi- mental model system for the study of glioblastoma growth and invasion.Cell and tissue research, 310(3):257–270, 2002
Bert Grobben, Peter De Deyn, and Herman Slegers. Rat c6 glioma as experi- mental model system for the study of glioblastoma growth and invasion.Cell and tissue research, 310(3):257–270, 2002. 36
2002
-
[10]
Glioma: experimental models and reality.Acta neuropathologica, 133(2):263–282, 2017
Krissie Lenting, Roel Verhaak, Mark Ter Laan, Pieter Wesseling, and William Leenders. Glioma: experimental models and reality.Acta neuropathologica, 133(2):263–282, 2017
2017
-
[11]
Large animal models of glioma: Current status and future prospects.Anticancer Research, 41(11):5343–5353, 2021
William H Hicks, Cylaina E Bird, Mark N Pernik, Ali S Haider, Aksharku- mar Dobariya, Kalil G Abdullah, Salah G Aoun, R Timothy Bentley, Aaron A Cohen-Gadol, Robert M Bachoo, et al. Large animal models of glioma: Current status and future prospects.Anticancer Research, 41(11):5343–5353, 2021
2021
-
[12]
Jana Lipková, Panagiotis Angelikopoulos, Stephen Wu, Esther Alberts, Benedikt Wiestler, Christian Diehl, Christine Preibisch, Thomas Pyka, Stephanie E Combs, Panagiotis Hadjidoukas, et al. Personalized radiother- apy design for glioblastoma: integrating mathematical tumor models, multi- modal scans, and bayesian inference.IEEE transactions on medical imag...
2019
-
[13]
Towardpatient-specific, biologicallyoptimizedradiationtherapyplans for the treatment of glioblastoma.PloS one, 8(11):e79115, 2013
David Corwin, Clay Holdsworth, Russell C Rockne, Andrew D Trister, Maciej M Mrugala, Jason K Rockhill, Robert D Stewart, Mark Phillips, and Kristin R Swanson. Towardpatient-specific, biologicallyoptimizedradiationtherapyplans for the treatment of glioblastoma.PloS one, 8(11):e79115, 2013
2013
-
[14]
David A Hormuth II, Maguy Farhat, Bikash Panthi, Holly Langshaw, Mi- hir D Shanker, Wasif Talpur, Sara Thrower, Jodi Goldman, Sophia Ty, Calliope Custer, et al. Forecasting chemoradiation response mid-treatment for high-grade gliomas through patient-specific biology-based modeling.International Journal of Radiation Oncology* Biology* Physics, 2025
2025
-
[15]
Minimally sufficient experimental design using identifiability analysis.npj Systems Biology and Applications, 10(1):2, 2024
Jana L Gevertz and Irina Kareva. Minimally sufficient experimental design using identifiability analysis.npj Systems Biology and Applications, 10(1):2, 2024
2024
-
[16]
On optimal temozolomide scheduling for slowly growing glioblastomas.Neuro-Oncology Ad- vances, 4(1):vdac155, 2022
Berta Segura-Collar, Juan Jiménez-Sánchez, Ricardo Gargini, Miodrag Dragoj, Juan M Sepúlveda-Sánchez, Milica Pešić, María A Ramírez, Luis E Ayala- Hernández, Pilar Sánchez-Gómez, and Víctor M Pérez-García. On optimal temozolomide scheduling for slowly growing glioblastomas.Neuro-Oncology Ad- vances, 4(1):vdac155, 2022
2022
-
[17]
Integrated biophysical modeling and image analysis: applica- tion to neuro-oncology.Annual review of biomedical engineering, 22(1):309–341, 2020
Andreas Mang, Spyridon Bakas, Shashank Subramanian, Christos Davatzikos, and George Biros. Integrated biophysical modeling and image analysis: applica- tion to neuro-oncology.Annual review of biomedical engineering, 22(1):309–341, 2020. 37
2020
-
[18]
Oncology and mechanics: landmark studies and promising clinical applications
Stéphane Urcun, Guillermo Lorenzo, Davide Baroli, Pierre-Yves Rohan, Giuseppe Sciumè, Wafa Skalli, Vincent Lubrano, and Stéphane PA Bordas. Oncology and mechanics: landmark studies and promising clinical applications. Advances in Applied Mechanics, 55:513–571, 2022
2022
-
[19]
Chemoradiotherapy in malignant glioma: standard of care and future direc- tions.Journal of Clinical Oncology, 25(26):4127–4136, 2007
Roger Stupp, Monika E Hegi, Mark R Gilbert, and Arnab Chakravarti. Chemoradiotherapy in malignant glioma: standard of care and future direc- tions.Journal of Clinical Oncology, 25(26):4127–4136, 2007
2007
-
[20]
Stan- dards of care for treatment of recurrent glioblastoma—are we there yet?Neuro- oncology, 15(1):4–27, 2013
Michael Weller, Timothy Cloughesy, James R Perry, and Wolfgang Wick. Stan- dards of care for treatment of recurrent glioblastoma—are we there yet?Neuro- oncology, 15(1):4–27, 2013
2013
-
[21]
Predicting in vivo glioma growth with the reaction diffusion equation constrained by quantitative mag- netic resonance imaging data.Physical biology, 12(4):046006, 2015
David A Hormuth II, Jared A Weis, Stephanie L Barnes, Michael I Miga, Erin C Rericha, Vito Quaranta, and Thomas E Yankeelov. Predicting in vivo glioma growth with the reaction diffusion equation constrained by quantitative mag- netic resonance imaging data.Physical biology, 12(4):046006, 2015
2015
-
[22]
David A Hormuth, Jared A Weis, Stephanie L Barnes, Michael I Miga, Erin C Rericha, Vito Quaranta, and Thomas E Yankeelov. A mechanically cou- pled reaction–diffusion model that incorporates intra-tumoural heterogeneity to predict in vivo glioma growth.Journal of The Royal Society Interface, 14(128):20161010, 2017
2017
-
[23]
Heterogeneous mechanical stress and interstitial fluid flow predictions derived from dce-mri for rat u251n orthotopic gliomas.Annals of Biomedical Engineering, pages 1–14, 2024
Julian A Rey, Katelynn G Spanick, Glauber Cabral, Isabel N Rivera-Santiago, Tavarekere N Nagaraja, Stephen L Brown, James R Ewing, and Malisa Sarnti- noranont. Heterogeneous mechanical stress and interstitial fluid flow predictions derived from dce-mri for rat u251n orthotopic gliomas.Annals of Biomedical Engineering, pages 1–14, 2024
2024
-
[24]
Classical mathematical models for description and prediction of experimental tumor growth.PLoS computational biology, 10(8):e1003800, 2014
Sébastien Benzekry, Clare Lamont, Afshin Beheshti, Amanda Tracz, John ML Ebos, Lynn Hlatky, and Philip Hahnfeldt. Classical mathematical models for description and prediction of experimental tumor growth.PLoS computational biology, 10(8):e1003800, 2014
2014
-
[25]
Predicting regrowth of low-grade gliomas after radiotherapy.PLOS Computational Biology, 19(3):e1011002, 2023
Stéphane Plaszczynski, Basile Grammaticos, Johan Pallud, Jean-Eric Cam- pagne, and Mathilde Badoual. Predicting regrowth of low-grade gliomas after radiotherapy.PLOS Computational Biology, 19(3):e1011002, 2023
2023
-
[26]
Sarah C Brüningk, Jeffrey Peacock, Christopher J Whelan, Renee Brady- Nicholls, Hsiang-Hsuan M Yu, Solmaz Sahebjam, and Heiko Enderling. Inter- mittent radiotherapy as alternative treatment for recurrent high grade glioma: 38 a modeling study based on longitudinal tumor measurements.Scientific reports, 11(1):20219, 2021
2021
-
[27]
Overcoming chemotherapy resis- tance in low-grade gliomas: A computational approach.PLoS computational biology, 19(11):e1011208, 2023
Thibault Delobel, Luis E Ayala-Hernández, Jesús J Bosque, Julián Pérez- Beteta, Salvador Chulián, Manuel García-Ferrer, Pilar Piñero, Philippe Schucht, Michael Murek, and Víctor M Pérez-García. Overcoming chemotherapy resis- tance in low-grade gliomas: A computational approach.PLoS computational biology, 19(11):e1011208, 2023
2023
-
[28]
Christina H Wang, Jason K Rockhill, Maciej Mrugala, Danielle L Peacock, Albert Lai, Katy Jusenius, Joanna M Wardlaw, Timothy Cloughesy, Alexan- der M Spence, Russ Rockne, et al. Prognostic significance of growth kinetics in newly diagnosed glioblastomas revealed by combining serial imaging with a novel biomathematical model.Cancer research, 69(23):9133–9140, 2009
2009
-
[29]
Patient-specific mathematical neuro-oncology: using a simple proliferation and invasion tumor model to inform clinical practice
Pamela R Jackson, Joseph Juliano, Andrea Hawkins-Daarud, Russell C Rockne, and Kristin R Swanson. Patient-specific mathematical neuro-oncology: using a simple proliferation and invasion tumor model to inform clinical practice. Bulletin of mathematical biology, 77(5):846–856, 2015
2015
-
[30]
Individualizing glioma radiotherapy planning by optimization of a data and physics-informed discrete loss.Nature Communica- tions, 16(1):5982, 2025
Michal Balcerak, Jonas Weidner, Petr Karnakov, Ivan Ezhov, Sergey Litvinov, Petros Koumoutsakos, Tamaz Amiranashvili, Ray Zirui Zhang, John S Lowen- grub, Igor Yakushev, et al. Individualizing glioma radiotherapy planning by optimization of a data and physics-informed discrete loss.Nature Communica- tions, 16(1):5982, 2025
2025
-
[31]
Inducing biomechanical heterogeneity in brain tumor model- ing by mr elastography: effects on tumor growth, vascular density and delivery of therapeutics.Cancers, 14(4):884, 2022
Constantinos Harkos, Siri Fløgstad Svensson, Kyrre E Emblem, and Triantafyl- los Stylianopoulos. Inducing biomechanical heterogeneity in brain tumor model- ing by mr elastography: effects on tumor growth, vascular density and delivery of therapeutics.Cancers, 14(4):884, 2022
2022
-
[32]
A personalized mathematical tool for neuro-oncology: A clinical case study.International Journal of Non-Linear Mechanics, 107:170–181, 2018
Abramo Agosti, Chiara Giverso, Elena Faggiano, Aymeric Stamm, and Pasquale Ciarletta. A personalized mathematical tool for neuro-oncology: A clinical case study.International Journal of Non-Linear Mechanics, 107:170–181, 2018
2018
-
[33]
Selection and validation of predictive mod- els of radiation effects on tumor growth based on noninvasive imaging data
EABF Lima, JT Oden, B Wohlmuth, A Shahmoradi, DA Hormuth II, TE Yan- keelov, L Scarabosio, and T Horger. Selection and validation of predictive mod- els of radiation effects on tumor growth based on noninvasive imaging data. Computer methods in applied mechanics and engineering, 327:277–305, 2017. 39
2017
-
[34]
Advances in magnetic resonance imaging of brain tumours.Cur- rent opinion in neurology, 16(6):643–650, 2003
Jeremy Rees. Advances in magnetic resonance imaging of brain tumours.Cur- rent opinion in neurology, 16(6):643–650, 2003
2003
-
[35]
Advanced mri in the management of adult gliomas.British Journal of Neurosurgery, 21(6):550–561, 2007
MD Jenkinson, DG Du Plessis, C Walker, and TS Smith. Advanced mri in the management of adult gliomas.British Journal of Neurosurgery, 21(6):550–561, 2007
2007
-
[36]
Beyond conventional imaging: Advancements in mri for glioma malignancy pre- diction and molecular profiling.Magnetic Resonance Imaging, 112:63–81, 2024
Paulina Śledzińska-Bebyn, Jacek Furtak, Marek Bebyn, and Zbigniew Serafin. Beyond conventional imaging: Advancements in mri for glioma malignancy pre- diction and molecular profiling.Magnetic Resonance Imaging, 112:63–81, 2024
2024
-
[37]
Functional mri for surgery of gliomas.Current treatment options in neurology, 19(10):34, 2017
Antonella Castellano, Sara Cirillo, Lorenzo Bello, Marco Riva, and Andrea Falini. Functional mri for surgery of gliomas.Current treatment options in neurology, 19(10):34, 2017
2017
-
[38]
Multiclass segmentation of brain tumor from mri images
PK Bhagat and Prakash Choudhary. Multiclass segmentation of brain tumor from mri images. InApplications of Artificial Intelligence Techniques in Engi- neering: SIGMA 2018, Volume 1, pages 543–553. Springer, 2018
2018
-
[39]
Image-based personalization of computational models for predicting response of high-grade glioma to chemoradiation.Sci- entific reports, 11(1):8520, 2021
David A Hormuth, Karine A Al Feghali, Andrew M Elliott, Thomas E Yan- keelov, and Caroline Chung. Image-based personalization of computational models for predicting response of high-grade glioma to chemoradiation.Sci- entific reports, 11(1):8520, 2021
2021
-
[40]
An image-based modeling framework for predicting spatiotemporal brain cancer biology within individual patients
Kamila M Bond, Lee Curtin, Sara Ranjbar, Ariana E Afshari, Leland S Hu, Joshua B Rubin, and Kristin R Swanson. An image-based modeling framework for predicting spatiotemporal brain cancer biology within individual patients. Frontiers in Oncology, 13:1185738, 2023
2023
-
[41]
Quantitative in vivo imaging to enable tumour forecasting and treatment optimization
Guillermo Lorenzo, David A Hormuth II, Angela M Jarrett, Ernesto ABF Lima, Shashank Subramanian, George Biros, J Tinsley Oden, Thomas JR Hughes, and Thomas E Yankeelov. Quantitative in vivo imaging to enable tumour forecasting and treatment optimization. InCancer, complexity, computation, pages 55–97. Springer, 2022
2022
-
[42]
Multiscale modeling of solid stress and tumor cell invasion in response to dynamic mechanical microenvironment
H Chen, Y Cai, Qiang Chen, and Zhiyong Li. Multiscale modeling of solid stress and tumor cell invasion in response to dynamic mechanical microenvironment. Biomechanics and modeling in mechanobiology, 19:577–590, 2020
2020
-
[43]
Current trends in mathematical modeling of tumor–microenvironment interactions: a survey of tools and appli- cations.Experimental Biology and Medicine, 235(4):411–423, 2010
Katarzyna A Rejniak and Lisa J McCawley. Current trends in mathematical modeling of tumor–microenvironment interactions: a survey of tools and appli- cations.Experimental Biology and Medicine, 235(4):411–423, 2010. 40
2010
-
[44]
Modeling the mechanics of cancer: effect of changes in cellular and extra-cellular mechanical properties.Frontiers in oncology, 3:145, 2013
Parag Katira, Roger T Bonnecaze, and Muhammad H Zaman. Modeling the mechanics of cancer: effect of changes in cellular and extra-cellular mechanical properties.Frontiers in oncology, 3:145, 2013
2013
-
[45]
The role of mechanical forces in tumor growth and therapy.Annual review of biomedical engineering, 16(1):321–346, 2014
Rakesh K Jain, John D Martin, and Triantafyllos Stylianopoulos. The role of mechanical forces in tumor growth and therapy.Annual review of biomedical engineering, 16(1):321–346, 2014
2014
-
[46]
On the poroelastic biot coefficient for a granitic rock.Geo- sciences, 11(5):219, 2021
APS Selvadurai. On the poroelastic biot coefficient for a granitic rock.Geo- sciences, 11(5):219, 2021
2021
-
[47]
General theory of three-dimensional consolidation.Journal of applied physics, 12(2):155–164, 1941
Maurice A Biot. General theory of three-dimensional consolidation.Journal of applied physics, 12(2):155–164, 1941
1941
-
[48]
John wiley & sons, 1996
Karl Terzaghi, Ralph B Peck, and Gholamreza Mesri.Soil mechanics in engi- neering practice. John wiley & sons, 1996
1996
-
[49]
A tumor growth model with deformable ecm.Physical biology, 11(6):065004, 2014
Giuseppe Sciumè, R Santagiuliana, Mauro Ferrari, P Decuzzi, and BA Schrefler. A tumor growth model with deformable ecm.Physical biology, 11(6):065004, 2014
2014
-
[50]
Three phase flow dynamics in tumor growth.Computa- tional Mechanics, 53:465–484, 2014
Giuseppe Sciumè, WG Gray, Fazle Hussain, Mauro Ferrari, P Decuzzi, and BA3167634 Schrefler. Three phase flow dynamics in tumor growth.Computa- tional Mechanics, 53:465–484, 2014
2014
-
[51]
Mechanistic modeling of vascular tumor growth: an extension of biot’s theory to hierarchical bi-compartment porous medium systems.Acta Mechanica, 232(4):1445–1478, 2021
Giuseppe Sciumè. Mechanistic modeling of vascular tumor growth: an extension of biot’s theory to hierarchical bi-compartment porous medium systems.Acta Mechanica, 232(4):1445–1478, 2021
2021
-
[52]
Non-operable glioblastoma: proposition of patient-specific forecasting by image-informed poromechanical model.Brain Multiphysics, 4:100067, 2023
Stéphane Urcun, Davide Baroli, Pierre-Yves Rohan, Wafa Skalli, Vincent Lu- brano, Stéphane PA Bordas, and Giuseppe Sciumè. Non-operable glioblastoma: proposition of patient-specific forecasting by image-informed poromechanical model.Brain Multiphysics, 4:100067, 2023
2023
-
[53]
Barnes, Jennifer G
Stephanie L. Barnes, Jennifer G. Whisenant, Mary E. Loveless, and Thomas E. Yankeelov. Practical dynamic contrast enhanced mri in small animal models of cancer: Data acquisition, data analysis, and interpretation.Pharmaceutics, 4(3):442–478, 2012
2012
-
[54]
A comparison of individual and population-derived vascular input functions for quantitative dce-mri in rats.Magnetic resonance imaging, 32(4):397–401, 2014
David A Hormuth II, Jack T Skinner, Mark D Does, and Thomas E Yankeelov. A comparison of individual and population-derived vascular input functions for quantitative dce-mri in rats.Magnetic resonance imaging, 32(4):397–401, 2014. 41
2014
-
[55]
Tumor growth modeling from the perspective of multiphase porous media mechanics.Molecular & cellular biomechanics: MCB, 9(3):193, 2012
Giuseppe Sciumè, SE Shelton, WG Gray, CT Miller, F Hussain, M Ferrari, P Decuzzi, and BA Schrefler. Tumor growth modeling from the perspective of multiphase porous media mechanics.Molecular & cellular biomechanics: MCB, 9(3):193, 2012
2012
-
[56]
Digital twinning of cellular capsule technology: Emerging outcomes from the perspective of porous media mechanics.PLoS One, 16(7):e0254512, 2021
Stéphane Urcun, Pierre-Yves Rohan, Wafa Skalli, Pierre Nassoy, Stéphane PA Bordas, and Giuseppe Sciumè. Digital twinning of cellular capsule technology: Emerging outcomes from the perspective of porous media mechanics.PLoS One, 16(7):e0254512, 2021
2021
-
[57]
A model for hysteretic constitutive relations govern- ing multiphase flow: 1
JC Parker and RJ Lenhard. A model for hysteretic constitutive relations govern- ing multiphase flow: 1. saturation-pressure relations.Water Resources Research, 23(12):2187–2196, 1987
1987
-
[58]
Determining three-phase permeabil- ity—saturation—pressure relations from two-phase system measurements.Jour- nal of Petroleum Science and Engineering, 4(1):57–65, 1990
JC Parker and RJ Lenhard. Determining three-phase permeabil- ity—saturation—pressure relations from two-phase system measurements.Jour- nal of Petroleum Science and Engineering, 4(1):57–65, 1990
1990
-
[59]
An avascular tumor growth model based on porous media mechanics and evolving natural states.Mathematics and Mechanics of Solids, 23(4):686– 712, 2018
Pietro Mascheroni, Melania Carfagna, Alfio Grillo, DP Boso, and Bernhard A Schrefler. An avascular tumor growth model based on porous media mechanics and evolving natural states.Mathematics and Mechanics of Solids, 23(4):686– 712, 2018
2018
-
[60]
Raffaella Santagiuliana, Rui C Pereira, Bernhard A Schrefler, and Paolo De- cuzzi. Predicting the role of microstructural and biomechanical cues in tumor growth and spreading.International journal for numerical methods in biomedi- cal engineering, 34(3):e2935, 2018
2018
-
[61]
Mechanotransduction in tumor dynamics modeling.Physics of Life Reviews, 44:279–301, 2023
Beatriz Blanco, Héctor Gómez, Juan Melchor, Roberto Palma, Juan Soler, and Guillermo Rus. Mechanotransduction in tumor dynamics modeling.Physics of Life Reviews, 44:279–301, 2023
2023
-
[62]
Hormuth, Angela M
D. Hormuth, Angela M. Jarrett, Xinzeng Feng, and T. Yankeelov. Calibrating a predictive model of tumor growth and angiogenesis with quantitative mri. Annals of Biomedical Engineering, 2019
2019
-
[63]
Rat brain interstitial fluid pressure measured with mi- cropipettes.American Journal of Physiology-Heart and Circulatory Physiology, 244(2):H239–H246, 1983
H Wiig and RK Reed. Rat brain interstitial fluid pressure measured with mi- cropipettes.American Journal of Physiology-Heart and Circulatory Physiology, 244(2):H239–H246, 1983. 42
1983
-
[64]
Non-invasive imaging of interstitial fluid transport parameters in solid tumors in vivo.Sci- entific Reports, 13(1):7132, 2023
Sharmin Majumder, Md Tauhidul Islam, and Raffaella Righetti. Non-invasive imaging of interstitial fluid transport parameters in solid tumors in vivo.Sci- entific Reports, 13(1):7132, 2023
2023
-
[65]
Non-invasive in vivo mapping of tumour vascular and interstitial volume fractions.European Journal of Cancer, 34(9):1448–1454, 1998
R Weissleder, HC Cheng, E Marecos, K Kwong, and A Bogdanov. Non-invasive in vivo mapping of tumour vascular and interstitial volume fractions.European Journal of Cancer, 34(9):1448–1454, 1998
1998
-
[66]
The estimation of tumor cell percentage for molecular testing by pathologists is not accurate.Modern Pathology, 27(2):168– 174, 2014
Alexander JJ Smits, J Alain Kummer, Peter C De Bruin, Mijke Bol, Jan G Van Den Tweel, Kees A Seldenrijk, Stefan M Willems, G Johan A Offerhaus, Roel A De Weger, Paul J Van Diest, et al. The estimation of tumor cell percentage for molecular testing by pathologists is not accurate.Modern Pathology, 27(2):168– 174, 2014
2014
-
[67]
Interstitial fluid pressure in intracranial tumours in patients and in rodents.British journal of cancer, 75(6):829–836, 1997
Y Boucher, H Salehi, B Witwer, GRT Harsh, and RK Jain. Interstitial fluid pressure in intracranial tumours in patients and in rodents.British journal of cancer, 75(6):829–836, 1997
1997
-
[68]
Rasha Elmghirbi, Tavarekere N Nagaraja, Stephen L Brown, Kelly A Keenan, Swayamprava Panda, Glauber Cabral, Hassan Bagher-Ebadian, George W Di- vine, Ian Y Lee, and James R Ewing. Toward a noninvasive estimate of intersti- tial fluid pressure by dynamic contrast-enhanced mri in a rat model of cerebral tumor.Magnetic resonance in medicine, 80(5):2040–2052, 2018
2040
-
[69]
Diffusion in brain extracellular space.Phys- iological reviews, 88(4):1277–1340, 2008
Eva Syková and Charles Nicholson. Diffusion in brain extracellular space.Phys- iological reviews, 88(4):1277–1340, 2008
2008
-
[70]
Extra- cellular space parameters in the rat neocortex and subcortical white matter during postnatal development determined by diffusion analysis.Neuroscience, 55(2):339–351, 1993
A Lehmenkühler, E Syková, J Svoboda, K Zilles, and Ch Nicholson. Extra- cellular space parameters in the rat neocortex and subcortical white matter during postnatal development determined by diffusion analysis.Neuroscience, 55(2):339–351, 1993
1993
-
[71]
The need for mathematical modelling of spatial drug distribution within the brain.Fluids and Barriers of the CNS, 16:1–33, 2019
Esmée Vendel, Vivi Rottschäfer, and Elizabeth CM de Lange. The need for mathematical modelling of spatial drug distribution within the brain.Fluids and Barriers of the CNS, 16:1–33, 2019
2019
-
[72]
Mr-derived cerebral blood volume maps: issues regarding histological validation and assessment of tumor angiogenesis
Arvind P Pathak, Kathleen M Schmainda, B Douglas Ward, JR Linderman, Kelly J Rebro, and Andrew S Greene. Mr-derived cerebral blood volume maps: issues regarding histological validation and assessment of tumor angiogenesis. Magnetic Resonance in Medicine: An Official Journal of the International So- ciety for Magnetic Resonance in Medicine, 46(4):735–747, 2001. 43
2001
-
[73]
Transport phenomena in biological systems
George A Truskey, Fan Yuan, and David F Katz. Transport phenomena in biological systems. 2004
2004
-
[74]
Me- chanical difference between white and gray matter in the rat cerebellum mea- sured by scanning force microscopy.Journal of biomechanics, 43(15):2986–2992, 2010
Andreas F Christ, Kristian Franze, Helene Gautier, Pouria Moshayedi, James Fawcett, Robin JM Franklin, Ragnhildur T Karadottir, and Jochen Guck. Me- chanical difference between white and gray matter in the rat cerebellum mea- sured by scanning force microscopy.Journal of biomechanics, 43(15):2986–2992, 2010
2010
-
[75]
Viscoelastic properties of the rat brain in the sagittal plane: effects of anatomical structure and age.Annals of biomedical engineering, 40:70–78, 2012
JohnDFinan, BenjaminSElkin, EricaMPearson, IreneLKalbian, andBarclay Morrison. Viscoelastic properties of the rat brain in the sagittal plane: effects of anatomical structure and age.Annals of biomedical engineering, 40:70–78, 2012
2012
-
[76]
Malignant transformation of low-grade astrocytomas through a mechano-biological trigger- ing hypothesis: Imaging-informed modelling
Meryem Abbad Andaloussi, Stéphane Urcun, Andreas Husch, Isabel Fernan- des Arroteia, Giuseppe Sciumè, Stéphane Bordas, and Frank Hertel. Malignant transformation of low-grade astrocytomas through a mechano-biological trigger- ing hypothesis: Imaging-informed modelling. InModelling and Computational Approaches for Multi-Scale Phenomena in Cancer Research: ...
2025
-
[77]
Usefulness of diffusion/perfusion-weighted mri in rat gliomas: Correlation with histopathology1.Academic radiology, 12(5):640–651, 2005
Guoguang Fan, Peizhuo Zang, Fengdong Jing, Zhenhua Wu, and Qiyong Guo. Usefulness of diffusion/perfusion-weighted mri in rat gliomas: Correlation with histopathology1.Academic radiology, 12(5):640–651, 2005
2005
-
[78]
Interstitial fluid flow: the mechanical environment of cells and foundation of meridians.Evidence-Based Complemen- tary and Alternative Medicine, 2012(1):853516, 2012
Wei Yao, Yabei Li, and Guanghong Ding. Interstitial fluid flow: the mechanical environment of cells and foundation of meridians.Evidence-Based Complemen- tary and Alternative Medicine, 2012(1):853516, 2012
2012
-
[79]
A multiphase model for three- dimensional tumor growth.New journal of physics, 15(1):015005, 2013
Giuseppe Sciume, S Shelton, William G Gray, Cass T Miller, Fazle Hussain, Mauro Ferrari, Paolo Decuzzi, and BA Schrefler. A multiphase model for three- dimensional tumor growth.New journal of physics, 15(1):015005, 2013
2013
-
[80]
Assessment of the experimental model of transplanted c6 glioblastoma in wistar rats.Journal of neuro-oncology, 7:299–304, 1989
F San-Galli, P Vrignaud, J Robert, JM Coindre, and F Cohadon. Assessment of the experimental model of transplanted c6 glioblastoma in wistar rats.Journal of neuro-oncology, 7:299–304, 1989
1989
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.