Recognition: unknown
Solvent-induced memory effects in a model electrolyte
Pith reviewed 2026-05-08 04:20 UTC · model grok-4.3
The pith
A Brownian-particle model of ions and solvent yields memory kernels that simplify charge fluctuations for fast solvents but produce two-step relaxation when solvents move slowly.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
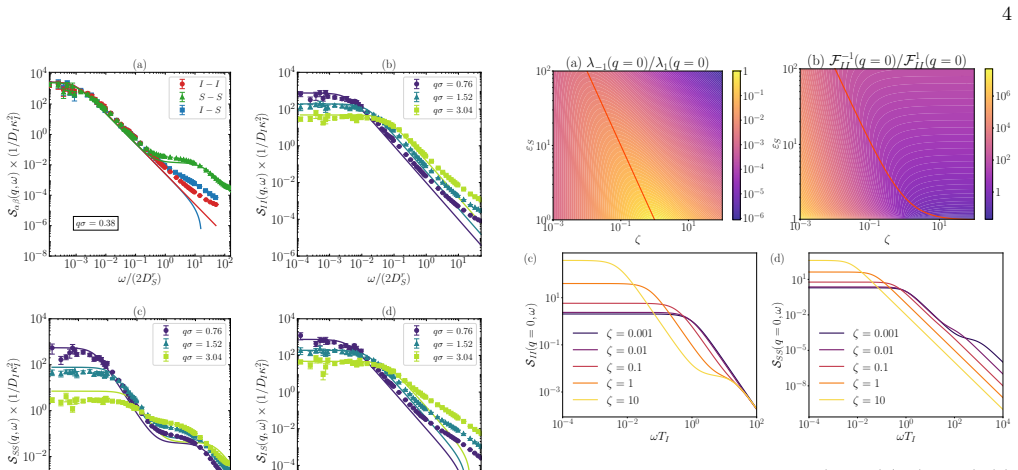
By representing ions and solvent as Brownian particles, stochastic density functional theory produces a generalized Langevin equation for the ionic charge density that incorporates solvent memory. In the limit of fast solvent relative to ions the memory kernel yields simple analytic forms for the dynamical charge structure factors that are confirmed by simulation; when solvent relaxation is slow the same kernel generates a two-step relaxation in the ionic dynamics.
What carries the argument
The generalized Langevin equation for the ionic charge density that carries an explicit solvent-mediated memory kernel derived via stochastic density functional theory
If this is right
- Dynamical charge structure factors acquire closed-form expressions once solvent and ion timescales separate.
- Ionic relaxation displays an emergent two-step process when solvent motion is comparable to or slower than ion motion.
- The same framework directly supplies the memory kernel that couples solvent polarization to ionic density fluctuations.
- Fluctuation-induced effects in electrolytes become accessible to mesoscopic analytic treatment without full molecular resolution.
Where Pith is reading between the lines
- The two-step relaxation signature could be searched for in dielectric or neutron-scattering spectra of electrolytes in viscous solvents.
- The approach might be extended to charged or polarizable solvent models to predict how dielectric response alters ionic transport.
- Similar memory kernels could appear in other systems with fast and slow Brownian species, such as colloidal suspensions with molecular solvents.
Load-bearing premise
Ions and solvent molecules can be represented as interacting Brownian particles whose dynamics admit a clear separation of timescales so that the memory kernel can be treated in the stated limits.
What would settle it
Brownian-dynamics simulations with deliberately slow solvent particles that fail to exhibit the predicted two-step decay in the ionic charge structure factor or velocity autocorrelation would falsify the memory-effect prediction.
Figures
read the original abstract
The fluctuations of ions in polar solvents remain poorly understood theoretically due to the complex coupling between ionic motion and solvent polarization. Indeed, while all-atom resolution can be achieved in numerical simulations, analytical approaches require suitable levels of coarse-graining. In this work, we describe ions and solvent molecules as interacting Brownian particles and use stochastic density functional theory to derive a generalized Langevin equation for the ionic charge density, explicitly accounting for solvent-mediated memory effects. In the regime where there is a clear timescale separation between fast solvent and slow ion dynamics, we obtain simple expressions for dynamical charge structure factors, which are validated by BD simulations. For slow solvents, we predict an emerging two-step relaxation in ionic dynamics. These results provide a mesoscopic approach for ion-solvent dynamics and open pathways to study fluctuation-induced phenomena in electrolytes.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper models ions and solvent molecules as interacting Brownian particles and applies stochastic density functional theory to derive a generalized Langevin equation for the ionic charge density that incorporates solvent-mediated memory effects. Under the assumption of clear timescale separation between fast solvent and slow ion dynamics, it obtains simple closed-form expressions for dynamical charge structure factors, validates these against Brownian dynamics simulations, and predicts an emerging two-step relaxation in ionic dynamics for slow solvents.
Significance. If the results hold, the work provides a useful mesoscopic analytical framework for ion-solvent coupling in electrolytes that bridges microscopic simulations and continuum descriptions. Strengths include the derivation of memory kernels from the underlying Brownian-particle model without fitted parameters, the closed-form limits under timescale separation, the use of BD simulations for validation, and the falsifiable prediction of two-step relaxation, all of which could inform studies of fluctuation-induced effects in electrolytes.
major comments (1)
- [Abstract and results] The central claim that the derived expressions for dynamical charge structure factors are validated by BD simulations lacks any quantitative match metrics, error analysis, or explicit discussion of how well the timescale-separation assumption holds in the reported simulations (see abstract and results sections). This leaves the support for the expressions only moderately strong and is load-bearing for the validation aspect of the main result.
Simulated Author's Rebuttal
We thank the referee for their positive evaluation of the significance of our work and for the constructive comment on strengthening the validation. We have revised the manuscript to address this point directly.
read point-by-point responses
-
Referee: [Abstract and results] The central claim that the derived expressions for dynamical charge structure factors are validated by BD simulations lacks any quantitative match metrics, error analysis, or explicit discussion of how well the timescale-separation assumption holds in the reported simulations (see abstract and results sections). This leaves the support for the expressions only moderately strong and is load-bearing for the validation aspect of the main result.
Authors: We agree that the original manuscript presented the agreement between theory and BD simulations primarily through visual inspection of the figures without accompanying quantitative metrics or an explicit assessment of the timescale separation. In the revised version we have added the following: (i) error bars on all simulation data points obtained from 20 independent runs, (ii) root-mean-square deviation values between the theoretical curves and the simulation data for each wave-vector, and (iii) a dedicated paragraph in the results section that reports the ratio of solvent to ionic relaxation times (approximately 15–25 depending on the parameter set) together with the decay time of the memory kernel extracted from the underlying Brownian-particle model. These additions confirm that the timescale-separation assumption is well satisfied in the reported simulations and provide a quantitative basis for the claimed validation. revision: yes
Circularity Check
No significant circularity identified
full rationale
The paper starts from an explicit modeling premise (ions and solvent as interacting Brownian particles) and applies stochastic density functional theory to derive a GLE for ionic charge density that includes solvent-mediated memory. Under the additional input assumption of clear timescale separation, it obtains closed-form limits for dynamical charge structure factors; these are presented as derived results and checked against independent BD simulations rather than being fitted or redefined from the outputs. No load-bearing self-citations, self-definitional steps, or ansatzes smuggled via prior work are required for the central claims, which remain self-contained against the stated premises and external validation.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Ions and solvent molecules can be modeled as interacting Brownian particles
- domain assumption There exists a clear timescale separation between fast solvent and slow ion dynamics
Reference graph
Works this paper leans on
-
[1]
H. S. Harned and B. B. Owen,The Physical Chemistry of Electrolyte Solutions(American Chemical Society Mono- graph Series, 1943)
1943
-
[2]
R´ esibois,Electrolyte Theory: An Elementary Introduc- tion to a Microscopic Approach(Harper and Row Pub- lishers, New York, Evanston and London, 1968)
P. R´ esibois,Electrolyte Theory: An Elementary Introduc- tion to a Microscopic Approach(Harper and Row Pub- lishers, New York, Evanston and London, 1968)
1968
-
[3]
R. A. Robinson and R. H. Stokes,Electrolyte Solutions (Dover Publications, 2002)
2002
-
[4]
Yamaguchi, T
T. Yamaguchi, T. Matsuoka, and S. Koda, A theoretical study on the frequency-dependent electric conductivity of electrolyte solutions, J. Chem. Phys.127, 234501 (2007)
2007
-
[5]
S. Roy, S. Yashonath, and B. Bagchi, Mode coupling the- ory analysis of electrolyte solutions: Time dependent dif- fusion, intermediate scattering function, and ion solva- tion dynamics, J. Chem. Phys.142, 124502 (2015)
2015
-
[6]
Bagchi and R
B. Bagchi and R. Biswas, Ionic Mobility and Ultrafast Solvation: Control of a Slow Phenomenon by Fast Dy- namics, Acc. Chem. Res.31, 181 (1998)
1998
-
[7]
Chandra and B
A. Chandra and B. Bagchi, Ion conductance in electrolyte solutions, J. Chem. Phys.110, 10024 (1999)
1999
-
[8]
Chandra and B
A. Chandra and B. Bagchi, Frequency dependence of ionic conductivity of electrolyte solutions, J. Chem. Phys. 112, 1876 (2000)
2000
-
[9]
Dufrˆ eche, O
J.-F. Dufrˆ eche, O. Bernard, P. Turq, A. Mukherjee, and B. Bagchi, Ionic Self-Diffusion in Concentrated Aque- ous Electrolyte Solutions, Phys. Rev. Lett.88, 095902 (2002)
2002
-
[10]
Dufrˆ eche, O
J.-F. Dufrˆ eche, O. Bernard, and P. Turq, Transport in electrolyte solutions: Are ions Brownian particles?, Jour- nal of Molecular Liquids118, 189 (2005)
2005
-
[11]
D. S. Dean, Langevin equation for the density of a system of interacting Langevin processes, J. Phys. A: Math. Gen. 29, L613 (1996)
1996
-
[12]
Kawasaki, Stochastic model of slow dynamics in su- percooled liquids and dense colloidal suspensions, Phys- ica A208, 35 (1994)
K. Kawasaki, Stochastic model of slow dynamics in su- percooled liquids and dense colloidal suspensions, Phys- ica A208, 35 (1994)
1994
-
[13]
Illien, The Dean–Kawasaki equation and stochastic density functional theory, Rep
P. Illien, The Dean–Kawasaki equation and stochastic density functional theory, Rep. Prog. Phys.88, 086601 (2025)
2025
-
[14]
D´ emery and D
V. D´ emery and D. S. Dean, The conductivity of strong electrolytes from stochastic density functional theory, J. Stat. Mech.2016, 023106 (2016)
2016
-
[15]
Y. Avni, D. Andelman, and H. Orland, Conductance of concentrated electrolytes: Multivalency and the Wien ef- fect, J. Chem. Phys.157, 154502 (2022)
2022
-
[16]
Y. Avni, R. M. Adar, D. Andelman, and H. Orland, Con- ductivity of Concentrated Electrolytes, Phys. Rev. Lett. 128, 098002 (2022)
2022
-
[17]
Bonneau, V
H. Bonneau, V. D´ emery, and ´E. Rapha¨ el, Temporal re- sponse of the conductivity of electrolytes, J. Stat. Mech. 2023, 073205 (2023)
2023
-
[18]
Bonneau, Y
H. Bonneau, Y. Avni, D. Andelman, and H. Orland, Frequency-dependent conductivity of concentrated elec- trolytes: A stochastic density functional theory, J. Chem. Phys.161, 244501 (2024)
2024
-
[19]
Bernard, M
O. Bernard, M. Jardat, B. Rotenberg, and P. Illien, On analytical theories for conductivity and self-diffusion in concentrated electrolytes, J. Chem. Phys.159, 164105 (2023)
2023
-
[20]
Bonneau, V
H. Bonneau, V. D´ emery, and E. Rapha¨ el, Stationary and transient correlations in driven electrolytes, J. Stat. Mech.2025, 033201 (2025)
2025
-
[21]
Donev, A
A. Donev, A. L. Garcia, J. P. P´ eraud, A. J. Nonaka, and J. B. Bell, Fluctuating Hydrodynamics and Debye- H¨ uckel-Onsager Theory for Electrolytes, Curr. Opin. Electrochem.13, 1 (2019)
2019
-
[22]
J. P. P´ eraud, A. J. Nonaka, J. B. Bell, A. Donev, and A. L. Garcia, Fluctuation-enhanced electric conductiv- ity in electrolyte solutions, Proc. Natl. Acad. Sci. U.S.A. 114, 10829 (2017)
2017
-
[23]
Zorkot, R
M. Zorkot, R. Golestanian, and D. J. Bonthuis, The Power Spectrum of Ionic Nanopore Currents: The Role of Ion Correlations, Nano Lett.16, 2205 (2016)
2016
-
[24]
Zorkot, R
M. Zorkot, R. Golestanian, and D. J. Bonthuis, Current fluctuations in nanopores: The effects of electrostatic and hydrodynamic interactions, Eur. Phys. J. Spec. Top.225, 1583 (2016)
2016
-
[25]
Zorkot and R
M. Zorkot and R. Golestanian, Current fluctuations across a nano-pore, J. Phys.: Condens. Matter30, 134001 (2018)
2018
-
[26]
Robin, Correlation-induced viscous dissipation in concentrated electrolytes, J
P. Robin, Correlation-induced viscous dissipation in concentrated electrolytes, J. Chem. Phys.160, 064503 (2024)
2024
-
[27]
D. S. Dean and R. Podgornik, Relaxation of the ther- mal Casimir force between net neutral plates containing Brownian charges, Phys. Rev. E89, 032117 (2014)
2014
-
[28]
B.-S. Lu, D. S. Dean, and R. Podgornik, Out-of- equilibrium thermal Casimir effect between Brownian conducting plates, EPL112, 20001 (2015)
2015
-
[29]
Mahdisoltani and R
S. Mahdisoltani and R. Golestanian, Long-Range Fluctuation-Induced Forces in Driven Electrolytes, Phys. Rev. Lett.126, 158002 (2021)
2021
-
[30]
Mahdisoltani and R
S. Mahdisoltani and R. Golestanian, Transient fluctuation-induced forces in driven electrolytes af- ter an electric field quench, New J. Phys.23, 073034 (2021)
2021
-
[31]
G. Du, D. S. Dean, B. Miao, and R. Podgornik, Correla- 6 tion Decoupling of Casimir Interaction in an Electrolyte Driven by External Electric Fields, Phys. Rev. Lett.133, 238002 (2024)
2024
-
[32]
G. Du, D. S. Dean, B. Miao, and R. Podgornik, Repul- sive thermal van der Waals interaction in multispecies asymmetric electrolytes driven by external electric fields, Phys. Rev. E111, 044108 (2025)
2025
-
[33]
Hubbard and L
J. Hubbard and L. Onsager, Dielectric dispersion and dielectric friction in electrolyte solutions. I., J. Chem. Phys.67, 4850 (1977)
1977
-
[34]
Buchner and G
R. Buchner and G. Hefter, Interactions and dynamics in electrolyte solutions by dielectric spectroscopy, Phys. Chem. Chem. Phys.11, 8984 (2009)
2009
-
[35]
J. Yang, S. Kondrat, C. Lian, H. Liu, A. Schlaich, and C. Holm, Solvent Effects on Structure and Screening in Confined Electrolytes, Physical Review Letters131, 118201 (2023)
2023
-
[36]
D´ emery, D
V. D´ emery, D. Toquer, and H. Berthoumieux, Ef- fect of solvent structure on the Wien effect and ionic correlations at the nanoscale, Faraday Discuss. , 10.1039.D5FD00149H (2026)
2026
-
[37]
Belloni, D
L. Belloni, D. Borgis, and M. Levesque, Screened Coulombic Orientational Correlations in Dilute Aqueous Electrolytes, J. Phys. Chem. Lett.9, 1985 (2018)
1985
-
[38]
Jungwirth and D
P. Jungwirth and D. Laage, Ion-Induced Long-Range Orientational Correlations in Water: Strong or Weak, Physiologically Relevant or Unimportant, and Unique to Water or Not?, J. Phys. Chem. Lett.9, 2056 (2018)
2056
-
[39]
Borgis, L
D. Borgis, L. Belloni, and M. Levesque, What Does Second-Harmonic Scattering Measure in Diluted Elec- trolytes?, J. Phys. Chem. Lett.9, 3698 (2018)
2018
-
[40]
Duboisset and P.-F
J. Duboisset and P.-F. Brevet, Salt-induced Long-to- Short Range Orientational Transition in Water, Phys. Rev. Lett.120, 263001 (2018)
2018
-
[41]
Duboisset, F
J. Duboisset, F. Rondepierre, and P.-F. Brevet, Long- Range Orientational Organization of Dipolar and Steric Liquids, J. Phys. Chem. Lett.11, 9869 (2020)
2020
-
[42]
Chandra and B
A. Chandra and B. Bagchi, The role of translational diffu- sion in the polarization relaxation in dense polar liquids, Chem. Phys. Lett.151, 47 (1988)
1988
-
[43]
Bagchi and A
B. Bagchi and A. Chandra, Polarization relaxation, di- electric dispersion, and solvation dynamics in dense dipo- lar liquid, J. Chem. Phys.90, 7338 (1989)
1989
-
[44]
Vijayadamodar, A
G. Vijayadamodar, A. Chandra, and B. Bagchi, Effects of translational diffusion on dielectric friction in a dipolar liquid, Chem. Phys. Lett.161, 413 (1989)
1989
-
[45]
Chandra and Biman
Amalendu. Chandra and Biman. Bagchi, Relationship between microscopic and macroscopic orientational re- laxation times in polar liquids, J. Phys. Chem.94, 3152 (1990)
1990
-
[46]
Varghese, P
S. Varghese, P. Illien, and B. Rotenberg, Dynamic cor- relations in a polar fluid: Confronting stochastic density functional theory to simulations, J. Chem. Phys.163, 124107 (2025)
2025
-
[47]
D. S. Dean and H. Diamant, Dielectric response and vis- cosity due to dipolar interactions (2026)
2026
-
[48]
Schr¨ oder, M
C. Schr¨ oder, M. Haberler, and O. Steinhauser, On the computation and contribution of conductivity in molec- ular ionic liquids, The Journal of Chemical Physics128, 134501 (2008)
2008
-
[49]
D. Roy, N. Patel, S. Conte, and M. Maroncelli, Dynam- ics in an Idealized Ionic Liquid Model, The Journal of Physical Chemistry B114, 8410 (2010)
2010
-
[50]
M. Sega, S. S. Kantorovich, A. Arnold, and C. Holm, On the calculation of the dielectric properties of liquid ionic systems, inRecent Advances in Broadband Dielec- tric Spectroscopy, edited by P. Kalmykov, Y (Springer Netherlands, 2013) pp. 103–122
2013
-
[51]
M. Sega, S. S. Kantorovich, C. Holm, and A. Arnold, Communication: Kinetic and pairing contributions in the dielectric spectra of electrolyte solutions, Journal of Chemical Physics140, 211101 (2014)
2014
-
[52]
T. H. N. Minh, J. Kim, G. Pireddu, I. Chubak, S. Nair, and B. Rotenberg, Electrical noise in electrolytes: A theoretical perspective, Faraday Discussions246, 198 (2023)
2023
-
[53]
Pireddu, C
G. Pireddu, C. J. Fairchild, S. P. Niblett, S. J. Cox, and B. Rotenberg, Impedance of nanocapacitors from molec- ular simulations to understand the dynamics of confined electrolytes, Proceedings of the National Academy of Sci- ences121, e2318157121 (2024)
2024
-
[54]
M. R. Becker, R. R. Netz, P. Loche, D. J. Bonthuis, D. Mouhanna, and H. Berthoumieux, Dielectric Prop- erties of Aqueous Electrolytes at the Nanoscale, Phys. Rev. Lett.134, 158001 (2025)
2025
-
[55]
Illien, A
P. Illien, A. Carof, and B. Rotenberg, Stochastic Density Functional Theory for Ions in a Polar Solvent, Phys. Rev. Lett.133, 268002 (2024)
2024
-
[56]
Within our description and approxi- mations, thetransverserelaxation time is simply given by 1/(DSq2 + 2Dr S) [46, 55]
Note thatτ S is thelongitudinalrelaxation time of the polarization field. Within our description and approxi- mations, thetransverserelaxation time is simply given by 1/(DSq2 + 2Dr S) [46, 55]
-
[57]
H. J. Bakker and J. L. Skinner, Vibrational Spectroscopy as a Probe of Structure and Dynamics in Liquid Water, Chemical Reviews110, 1498 (2010)
2010
-
[58]
Laage, G
D. Laage, G. Stirnemann, F. Sterpone, R. Rey, and J. T. Hynes, Reorientation and Allied Dynamics in Water and Aqueous Solutions, Annual Review of Physical Chem- istry62, 395 (2011)
2011
-
[59]
Supplemental Material
-
[60]
A. P. Thompson, H. M. Aktulga, R. Berger, D. S. Bolin- tineanu, W. M. Brown, P. S. Crozier, P. J. in ’t Veld, A. Kohlmeyer, S. G. Moore, T. D. Nguyen, R. Shan, M. J. Stevens, J. Tranchida, C. Trott, and S. J. Plimpton, LAMMPS - a flexible simulation tool for particle-based materials modeling at the atomic, meso, and continuum scales, Comput. Phys. Commun....
2022
-
[61]
N. C. Maity, S. Mitra, and R. Biswas, What Dictates the Optimal Concentration of Li-Based Battery Electrolytes? A Combined Experimental and Simulation Study (2025)
2025
-
[62]
Nakamoto, K
M. Nakamoto, K. Endo, S. Katayama, J. Han, E. Otani, H. Watanabe, I. Okae, and Y. Umebayashi, Dielectric behavior of propylene carbonate solutions with LiPF6, LiClO4, and LiBF4. II. Molecular dynamics simulation study, J. Chem. Phys.163, 204504 (2025)
2025
-
[63]
8εS(εS −1)(q 2 +κ 2 I /εS) κ2 I(q2a2 + 2)3 TS TI 3 1 1 + [ω¯τS(q)]2 + κ2 I /εS q2 +κ 2 I /εS 1 1 + [ω¯τI(q)]2 # (S10) SIS (q, ω)≃2T I
M. Sha, X. Ma, N. Li, F. Luo, G. Zhu, and M. D. Fayer, Dynamical properties of a room temperature ionic liquid: Using molecular dynamics simulations to implement a dynamic ion cage model, J. Chem. Phys.151, 154502 (2019). Solvent-induced memory effects in a model electrolyte Supplemental Material Sleeba Varghese,1 Benjamin Rotenberg, 1, 2 and Pierre Illie...
2019
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.