Recognition: no theorem link
Discovery of High-Voltage Magnesium-Ion Cathodes using Machine Learning and First-Principles Calculations
Pith reviewed 2026-05-13 04:59 UTC · model grok-4.3
The pith
Machine learning screens topological quantum materials to yield magnesium cathodes with average voltages of 3.66 V and 4.06 V.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
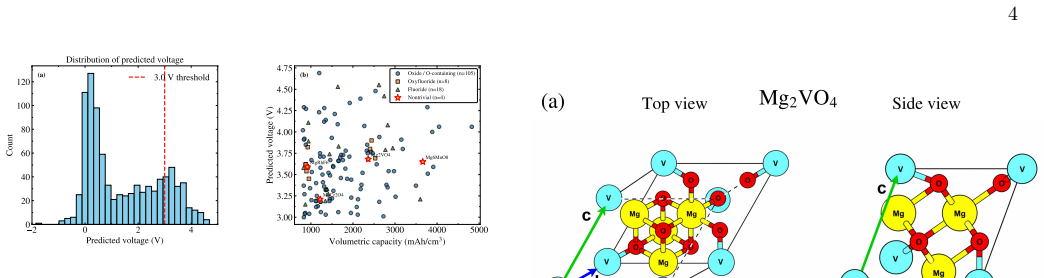
Screening 917 Mg-containing topological quantum materials with a modified crystal graph convolutional neural network identifies Mg₂VO₄ and Mg₆MnO₈ as candidates. Formation-energy and convex-hull analysis shows MgₓVO₄ follows a fully stable magnesiation pathway while MgₓMnO₈ shows only minor metastability at intermediate stages. DFT voltage profiles give averages of 3.66 V for Mg₂VO₄ and 4.06 V for Mg₆MnO₈, matching the machine-learning predictions. Both materials are semiconducting, with valence bands dominated by O 2p states and conduction bands by transition-metal d states, pointing to a charge-transfer redox mechanism. These TQMs therefore deliver higher voltages and competitive gravimetr
What carries the argument
The modified crystal graph convolutional neural network (mCGCNN) that ranks 917 Mg-containing topological quantum materials by predicted voltage and volumetric capacity, followed by DFT verification of formation energies, voltage profiles, and electronic structure.
If this is right
- Mg₂VO₄ exhibits a fully stable magnesiation pathway that supports reversible insertion without phase decomposition.
- Both materials remain semiconducting throughout the cycle, with oxygen 2p and transition-metal d states driving the redox activity.
- The calculated voltages exceed those of conventional magnesium cathodes while retaining competitive capacities.
- The close agreement between machine-learning predictions and DFT results validates the screening workflow for multivalent cathodes.
Where Pith is reading between the lines
- The same ML-plus-DFT pipeline could be reused to hunt for calcium- or aluminum-ion cathodes in topological materials.
- The identified charge-transfer character implies that oxygen redox participation may need surface coatings to prevent gas evolution in full cells.
- If these compositions prove synthesizable at scale, they could be paired with existing magnesium electrolytes to prototype full cells within months.
Load-bearing premise
The modified crystal graph convolutional neural network accurately predicts voltages and capacities for the Mg-containing topological quantum materials that lie outside its original training data.
What would settle it
Synthesizing polycrystalline Mg₂VO₄ or Mg₆MnO₈ and recording its galvanostatic discharge curve versus a magnesium metal anode in a non-aqueous electrolyte cell would directly test whether the measured average voltage matches the calculated 3.66 V or 4.06 V.
Figures




read the original abstract
Developing high-performance cathode materials for magnesium-ion batteries (MIBs) remains challenging because Mg$^{2+}$ ions move slowly, and conventional materials exhibit low voltage outputs. In this study, machine learning and first-principles calculations were combined to investigate topological quantum materials (TQMs) as a new class of cathode candidates. A modified crystal graph convolutional neural network (mCGCNN) was used to screen 917 Mg-containing TQMs, identifying a small subset of materials with predicted voltages above 3 V and high volumetric capacities. Among these, Mg$_2$VO$_4$ and Mg$_6$MnO$_8$ were selected for detailed density functional theory (DFT) analysis. Formation energy and convex-hull calculations indicate that Mg$_x$VO$_4$ exhibits a fully stable magnesiation pathway, whereas Mg$_x$MnO$_8$ demonstrates minor metastability at intermediate compositions. The calculated voltage profiles yield average voltages of 3.66 V for Mg$_2$VO$_4$ and 4.06 V for Mg$_6$MnO$_8$, in good agreement with machine learning predictions. Electronic structure analysis, supported by Wannier interpolation, confirms that both materials are semiconducting, with valence bands dominated by O $2p$ states and conduction bands by transition-metal $d$ states, indicating a charge-transfer redox mechanism. Compared to conventional Mg cathodes, these TQMs exhibit higher voltages and competitive capacities, underscoring their potential for next-generation multivalent batteries. This study demonstrates that integrating machine learning with first-principles calculations offers an efficient approach for discovering and understanding novel cathode materials.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper claims to discover two high-voltage Mg-ion cathode materials (Mg₂VO₄ with 3.66 V average voltage and Mg₆MnO₈ with 4.06 V) by screening 917 Mg-containing topological quantum materials with a modified crystal graph convolutional neural network (mCGCNN), followed by DFT validation of formation energies, convex-hull stability, voltage profiles, and electronic structure confirming a charge-transfer redox mechanism. These are presented as superior to conventional Mg cathodes in voltage while maintaining competitive capacities.
Significance. If the ML screening step is reliable, the workflow offers an efficient route to identify new multivalent battery cathodes, and the DFT results provide independent support for the two selected materials' voltages and stability. The reported agreement between mCGCNN predictions and DFT calculations is a strength of the validation approach.
major comments (1)
- The abstract states that the mCGCNN screened 917 Mg-containing TQMs to identify candidates with predicted voltages above 3 V, but provides no information on training-set composition for Mg compounds, the source of the voltage labels, validation metrics such as MAE or R², or whether the TQMs were held out from training. This is load-bearing for the central claim because the selection of Mg₂VO₄ and Mg₆MnO₈ for DFT (and the assertion of an efficient ML+DFT discovery method) depends on the model's generalization; without these details, the agreement on two points cannot confirm that the screening was not due to overfitting or arbitrary ranking.
Simulated Author's Rebuttal
We thank the referee for their careful reading of our manuscript and for highlighting an important point about the machine learning component of our workflow. We agree that additional details are needed to fully substantiate the screening results and have revised the manuscript accordingly.
read point-by-point responses
-
Referee: The abstract states that the mCGCNN screened 917 Mg-containing TQMs to identify candidates with predicted voltages above 3 V, but provides no information on training-set composition for Mg compounds, the source of the voltage labels, validation metrics such as MAE or R², or whether the TQMs were held out from training. This is load-bearing for the central claim because the selection of Mg₂VO₄ and Mg₆MnO₈ for DFT (and the assertion of an efficient ML+DFT discovery method) depends on the model's generalization; without these details, the agreement on two points cannot confirm that the screening was not due to overfitting or arbitrary ranking.
Authors: We agree that these details are essential to demonstrate the reliability and generalization of the mCGCNN screening. The original manuscript describes the mCGCNN architecture and its application but does not explicitly detail the training-set composition for Mg compounds, the origin of the voltage labels, quantitative validation metrics, or the hold-out status of the 917 TQMs. In the revised manuscript we have added a new subsection to the Methods section that provides this information: the training set consists of Mg-containing compounds drawn from established materials databases with voltage labels obtained from prior DFT calculations; we report the MAE and R² on both cross-validation and an independent test set; and we explicitly state that the 917 topological quantum materials were excluded from training to ensure the screening constitutes a true prediction on unseen structures. We have also updated the abstract to reference the model validation performance. These changes directly address the concern and reinforce the validity of selecting Mg₂VO₄ and Mg₆MnO₈ for DFT follow-up. revision: yes
Circularity Check
No circularity: ML screening and independent DFT validation are separate steps
full rationale
The paper trains an mCGCNN on (presumably external) data to screen 917 TQMs, selects two candidates, and then runs separate first-principles DFT calculations to obtain voltage profiles. The DFT results are presented as independent confirmation that happens to agree with the ML output; no equation, parameter fit, or self-citation reduces the DFT voltages to the ML predictions or vice versa. No self-definitional loops, fitted-input-as-prediction, or load-bearing self-citations are present in the provided text. The derivation chain remains self-contained against the external DFT benchmark.
Axiom & Free-Parameter Ledger
axioms (2)
- standard math Standard density functional theory approximations (exchange-correlation functional, pseudopotentials) are sufficient for accurate formation energies and voltage profiles in these oxide materials
- domain assumption The mCGCNN model, trained on prior materials data, generalizes to the 917 Mg-containing topological quantum materials without large extrapolation errors
Reference graph
Works this paper leans on
-
[1]
H. D. Yoo, I. Shterenberg, Y. Gofer, G. Gershinsky, N. Pour, and D. Aurbach, Mg rechargeable batteries: an on-going challenge, Energy & Environmental Science6, 2265 (2013)
work page 2013
-
[2]
R. Mohtadi and F. Mizuno, Magnesium batteries: Cur- rent state of the art, issues and future perspectives, Beil- stein journal of nanotechnology5, 1291 (2014)
work page 2014
-
[3]
Y. Chen, Y. Kang, Y. Zhao, L. Wang, J. Liu, Y. Li, Z. Liang, X. He, X. Li, N. Tavajohi, and L. Baohua, A review of lithium-ion battery safety concerns: The is- sues, strategies, and testing standards, Journal of Energy Chemistry59, 83 (2021)
work page 2021
-
[4]
I. T. Adebanjo, J. Eko, A. G. Agbeyegbe, S. F. Yuk, S. V. Cowart, E. A. Nagelli, F. J. Burpo, J. L. Allen, D. T. Tran, N. Bhattarai, K. Shah, J.-Y. Hwang, and H. H. Sun, A comprehensive review of lithium-ion battery components degradation and operational considerations: a safety perspective, Energy Advances4, 820 (2025)
work page 2025
-
[5]
S. Yagi, T. Ichitsubo, Y. Shirai, S. Yanai, T. Doi, K. Murase, and E. Matsubara, A concept of dual-salt polyvalent-metal storage battery, Journal of Materials Chemistry A2, 1144 (2014)
work page 2014
-
[6]
E. Levi, M. Levi, O. Chasid, and D. Aurbach, A review on the problems of the solid state ions diffusion in cathodes for rechargeable mg batteries, Journal of Electroceramics 22, 13 (2009)
work page 2009
-
[7]
S. H. Lapidus, N. N. Rajput, X. Qu, K. W. Chapman, K. A. Persson, and P. J. Chupas, Solvation structure and energetics of electrolytes for multivalent energy storage, Physical Chemistry Chemical Physics16, 21941 (2014)
work page 2014
-
[8]
N. N. Rajput, T. J. Seguin, B. M. Wood, X. Qu, and K. A. Persson, Elucidating solvation structures for ratio- nal design of multivalent electrolytes—a review, Model- ing Electrochemical Energy Storage at the Atomic Scale , 79 (2018)
work page 2018
- [9]
-
[10]
L. F. Wan, B. R. Perdue, C. A. Apblett, and D. Pren- dergast, Mg desolvation and intercalation mechanism at the Mo6S8 chevrel phase surface, Chemistry of Materials 27, 5932 (2015)
work page 2015
-
[11]
P. Saha, M. K. Datta, O. I. Velikokhatnyi, A. Manivan- nan, D. Alman, and P. N. Kumta, Rechargeable magne- sium battery: Current status and key challenges for the future, Progress in Materials Science66, 1 (2014)
work page 2014
-
[12]
D. Aurbach, Z. Lu, A. Schechter, Y. Gofer, H. Gizbar, R. Turgeman, Y. Cohen, M. Moshkovich, and E. Levi, Prototype systems for rechargeable magnesium batteries, Nature407, 724 (2000)
work page 2000
-
[13]
S. Okamoto, T. Ichitsubo, T. Kawaguchi, Y. Kuma- gai, F. Oba, S. Yagi, K. Shimokawa, N. Goto, T. Doi, and E. Matsubara, Intercalation and push-out process with spinel-to-rocksalt transition on Mg insertion into spinel oxides in magnesium batteries, Advanced Science 2, 1500072 (2015)
work page 2015
-
[14]
J. Zeng, Y. Yang, S. Lai, J. Huang, Y. Zhang, J. Wang, and J. Zhao, A promising high-voltage cathode mate- rial based on mesoporous Na 3V2(PO4)3/C for recharge- able magnesium batteries, Chemistry–A European Jour- nal23, 16898 (2017)
work page 2017
-
[15]
Y. Orikasa, T. Masese, Y. Koyama, T. Mori, M. Hat- tori, K. Yamamoto, T. Okado, Z.-D. Huang, T. Minato, 9 C. Tassel, J. Kim, Y. Kobayashi, T. Abe, H. Kageyama, and Y. Uchimoto, High energy density rechargeable mag- nesium battery using earth-abundant and non-toxic ele- ments, Scientific reports4, 5622 (2014)
work page 2014
-
[16]
T. Kaewmaraya, M. Ramzan, J. Osorio-Guill´ en, and R. Ahuja, Electronic structure and ionic diffusion of green battery cathode material: Mg 2Mo6S8, Solid State Ionics 261, 17 (2014)
work page 2014
-
[17]
R. Deng, C. Dai, Z. Wang, Y. Wang, G. Lu, C. Li, X. Huang, C. Chen, J. Huang, Z. Gao, L. Luo, S. Tan, H. Li, J. Wang, J. Wang, and F. Pan, Ad- vanced cathode for synergistic anion-cation redox reac- tions in magnesium-ion batteries—a pathway to fast dif- fusion/reaction kinetics, Composites Part B: Engineering 293, 112107 (2025)
work page 2025
-
[18]
P. Saranya, D. Vanitha, K. Sundaramahalingam, A. Shameem, and N. Nallamuthu, Comparative perfor- mance analysis of nanostructured metal oxides as cath- ode in solid state magnesium battery, Inorganic Chem- istry Communications178, 114458 (2025)
work page 2025
-
[19]
W. Chen, Z. Lin, X. Zhang, H. Zhou, and Y. Zhang, Ai- driven accelerated discovery of intercalation-type cath- ode materials for magnesium batteries, Journal of Energy Chemistry108, 40 (2025)
work page 2025
-
[20]
M. M. Obeid and Q. Sun, Recent advances in topological quantum anode materials for metal-ion batteries, Journal of Power Sources540, 231655 (2022)
work page 2022
-
[21]
X. Yi, W. Li, Z. Li, P. Zhou, Z. Ma, and L. Sun, Topolog- ical dual double node-line semimetals NaAlSi (Ge) and their potential as cathode material for sodium ion bat- teries, Journal of Materials Chemistry C7, 15375 (2019)
work page 2019
-
[22]
Z. Wu, G. Liang, W. K. Pang, T. Zhou, Z. Cheng, W. Zhang, Y. Liu, B. Johannessen, and Z. Guo, Coupling topological insulator SnSb 2Te4 nanodots with highly doped graphene for high-rate energy storage, Advanced Materials32, 1905632 (2020)
work page 2020
-
[23]
J. Liu, S. Wang, and Q. Sun, All-carbon-based porous topological semimetal for Li-ion battery anode material, Proceedings of the National Academy of Sciences114, 651 (2017)
work page 2017
-
[24]
Y. Zhao, Y. Lu, H. Li, Y. Zhu, Y. Meng, N. Li, D. Wang, F. Jiang, F. Mo, C. Long, Y. Gou, X. Li, Z. Huang, Q. Li, J. C. Ho, J. Fan, M. Sui, F. Chen, W. Zhu, W. Liu, and C. Zhi, Few-layer bismuth selenide cathode for low- temperature quasi-solid-state aqueous zinc metal batter- ies, Nature communications13, 752 (2022)
work page 2022
-
[25]
Y. Wang, J. Liu, P.-H. Du, Z. Sun, and Q. Sun, Screening topological quantum cathode materials for K-ion batter- ies by graph neural network and first-principles calcula- tions, ACS Applied Energy Materials6, 4503 (2023)
work page 2023
- [26]
- [27]
-
[28]
K. Choudhary and B. DeCost, Atomistic line graph neu- ral network for improved materials property predictions, npj Computational Materials7, 185 (2021)
work page 2021
-
[29]
C. Chen, W. Ye, Y. Zuo, C. Zheng, and S. P. Ong, Graph networks as a universal machine learning framework for molecules and crystals, Chemistry of Materials31, 3564 (2019)
work page 2019
-
[30]
K. T. Sch¨ utt, H. E. Sauceda, P.-J. Kindermans, A. Tkatchenko, and K.-R. M¨ uller, Schnet–a deep learn- ing architecture for molecules and materials, The Journal of chemical physics148(2018)
work page 2018
-
[31]
S. Dick and M. Fernandez-Serra, Machine learning ac- curate exchange and correlation functionals of the elec- tronic density, Nature communications11, 3509 (2020)
work page 2020
-
[32]
J. Zeng, D. Zhang, D. Lu, P. Mo, Z. Li, Y. Chen, M. Rynik, L. Huang, Z. Li, S. Shi, Y. Wang, H. Ye, P. Tuo, J. Yang, Y. Ding, Y. Li, D. Tisi, Q. Zeng, H. Bao, Y. Xia,et al., DeePMD-kit v2: A software package for deep potential models, The Journal of Chemical Physics 159(2023)
work page 2023
-
[33]
X. Gong, H. Li, N. Zou, R. Xu, W. Duan, and Y. Xu, General framework for e (3)-equivariant neural network representation of density functional theory hamiltonian, Nature Communications14, 2848 (2023)
work page 2023
-
[34]
A. Jain, S. P. Ong, G. Hautier, W. Chen, W. D. Richards, S. Dacek, S. Cholia, D. Gunter, D. Skinner, G. Ceder, and K. A. Persson, Commentary: The materials project: A materials genome approach to accelerating materials innovation, APL materials1(2013)
work page 2013
-
[35]
M. Hellenbrandt, The inorganic crystal structure database (ICSD)—present and future, Crystallography Reviews10, 17 (2004)
work page 2004
-
[36]
S. Wang, Y. Ji, J. Liu, Z. Liu, X. Zhang, Y. Guo, J. Lin, J. Tao, J. Kasemchainan, Y. Jiang, and H. Gao, Integrat- ing crystal structure and numerical data for predictive models of lithium-ion battery materials: A modified crys- tal graph convolutional neural networks approach, Jour- nal of Energy Storage80, 110220 (2024)
work page 2024
- [37]
-
[38]
B. Bradlyn, L. Elcoro, J. Cano, M. G. Vergniory, Z. Wang, C. Felser, M. I. Aroyo, and B. A. Bernevig, Topological quantum chemistry, Nature547, 298 (2017)
work page 2017
-
[39]
M. Vergniory, L. Elcoro, C. Felser, N. Regnault, B. A. Bernevig, and Z. Wang, A complete catalogue of high- quality topological materials, Nature566, 480 (2019)
work page 2019
-
[40]
M. G. Vergniory, B. J. Wieder, L. Elcoro, S. S. Parkin, C. Felser, B. A. Bernevig, and N. Regnault, All topolog- ical bands of all nonmagnetic stoichiometric materials, Science376, eabg9094 (2022)
work page 2022
-
[41]
A. Urban, D.-H. Seo, and G. Ceder, Computational un- derstanding of Li-ion batteries, npj Computational Ma- terials2, 16002 (2016)
work page 2016
-
[42]
P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococ- cioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalji, M. Lazzeri, L. Martin-Samos,et al., Quantum espresso: a modular and open-source software project for quantumsimulations of ...
work page 2009
-
[43]
J. P. Perdew and Y. Wang, Accurate and simple ana- lytic representation of the electron-gas correlation energy, Physical review B45, 13244 (1992)
work page 1992
-
[44]
J. P. Perdew, K. Burke, and M. Ernzerhof, Generalized gradient approximation made simple, Physical review let- ters77, 3865 (1996)
work page 1996
-
[45]
H. J. Monkhorst and J. D. Pack, Special points for brillouin-zone integrations, Physical review B13, 5188 10 (1976)
work page 1976
-
[46]
F. Zhou, M. Cococcioni, C. A. Marianetti, D. Morgan, and G. Ceder, First-principles prediction of redox poten- tials in transition-metal compounds with LDA+ U, Phys- ical Review B—Condensed Matter and Materials Physics 70, 235121 (2004)
work page 2004
-
[47]
M. Cococcioni and S. De Gironcoli, Linear response approach to the calculation of the effective interaction parameters in the lda+ u method, Physical Review B—Condensed Matter and Materials Physics71, 035105 (2005)
work page 2005
-
[48]
L. Wang, T. Maxisch, and G. Ceder, Oxidation energies of transition metal oxides within the GGA+ U frame- work, Physical Review B—Condensed Matter and Mate- rials Physics73, 195107 (2006)
work page 2006
-
[49]
A. Jain, G. Hautier, S. P. Ong, C. J. Moore, C. C. Fis- cher, K. A. Persson, and G. Ceder, Formation enthalpies by mixing GGA and GGA+ U calculations, Physical Re- view B—Condensed Matter and Materials Physics84, 045115 (2011)
work page 2011
-
[50]
J. M. Munro, K. Latimer, M. K. Horton, S. Dwarak- nath, and K. A. Persson, An improved symmetry-based approach to reciprocal space path selection in band struc- ture calculations, npj Computational Materials6, 112 (2020)
work page 2020
-
[51]
D. Ebrahimzadeh, S. S. Sharif, and Y. M. Banad, Ac- celerated discovery of vanadium oxide compositions: A WGAN-VAE framework for materials design, Materials Today Electronics13, 100155 (2025)
work page 2025
-
[52]
M. Wang and A. Navrotsky, Enthalpy of formation of LiNiO2, LiCoO2 and their solid solution, LiNi1−xCoxO2, Solid State Ionics166, 167 (2004)
work page 2004
- [53]
-
[54]
W. Sun, S. T. Dacek, S. P. Ong, G. Hautier, A. Jain, W. D. Richards, A. C. Gamst, K. A. Persson, and G. Ceder, The thermodynamic scale of inorganic crys- talline metastability, Science advances2, e1600225 (2016)
work page 2016
-
[55]
X. Han, C. Liu, J. Sun, A. D. Sendek, and W. Yang, Density functional theory calculations for evaluation of phosphorene as a potential anode material for magnesium batteries, RSC advances8, 7196 (2018)
work page 2018
-
[56]
T. Chen, G. Sai Gautam, W. Huang, and G. Ceder, First- principles study of the voltage profile and mobility of Mg intercalation in a chromium oxide spinel, Chemistry of Materials30, 153 (2018)
work page 2018
-
[57]
Q. D. Truong, M. Kempaiah Devaraju, P. D. Tran, Y. Gambe, K. Nayuki, Y. Sasaki, and I. Honma, Unrav- elling the surface structure of MgMn 2O4 cathode mate- rials for rechargeable magnesium-ion battery, Chemistry of Materials29, 6245 (2017)
work page 2017
-
[58]
A. Mukherjee, S. Taragin, H. Aviv, I. Perelshtein, and M. Noked, Rationally Designed Vanadium Pentoxide as High Capacity Insertion Material for Mg-Ion, Advanced Functional Materials30, 2003518 (2020)
work page 2020
-
[59]
M. M. Huie, D. C. Bock, E. S. Takeuchi, A. C. Marschilok, and K. J. Takeuchi, Cathode materials for magnesium and magnesium-ion based batteries, Coordi- nation Chemistry Reviews287, 15 (2015)
work page 2015
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.