Recognition: no theorem link
Competing crystallization pathways and cold crystallization kinetics in 10OS5 liquid crystal
Pith reviewed 2026-05-13 04:29 UTC · model grok-4.3
The pith
The energy released during cold crystallization of 10OS5 liquid crystal can be tuned by its thermal history, making it a candidate for thermal energy storage.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Varying the thermal history of 10OS5 directs it toward either vitrification of the smectic Y phase or crystallization into Cr2 which transforms to Cr1, with the subsequent cold crystallization releasing an energy amount that depends on the chosen pathway and can thus be adjusted for thermal storage purposes.
What carries the argument
The competing crystallization pathways between the glass of smectic Y and the metastable Cr2 and stable Cr1 phases, studied through differential scanning calorimetry and fitted with Avrami, Augis-Bennett, and isoconversional models.
If this is right
- Cooling rates determine whether the material forms a glass or crystallizes partially or fully into Cr2.
- Reheating triggers cold crystallization to Cr1 or Cr1/Cr2 mixtures, releasing different energies.
- The melting entropies and dielectric spectra confirm conformational disorder in the crystal phases.
- Tunable energy release by thermal history enables 10OS5 for thermal energy storage applications.
Where Pith is reading between the lines
- Other liquid crystals with similar metastable phases might exhibit comparable tunability for energy storage.
- Device integration would require verifying performance over many cycles to ensure no loss of capacity.
- The kinetic analysis methods could be applied to predict behavior in related compounds for optimized storage materials.
Load-bearing premise
The phase assignments from DSC and the extracted energy values accurately capture the long-term repeatable performance without degradation or side reactions in practical use.
What would settle it
Repeated thermal cycling experiments on 10OS5 samples that show progressive reduction in the cold crystallization energy or emergence of unexpected phases.
Figures



read the original abstract
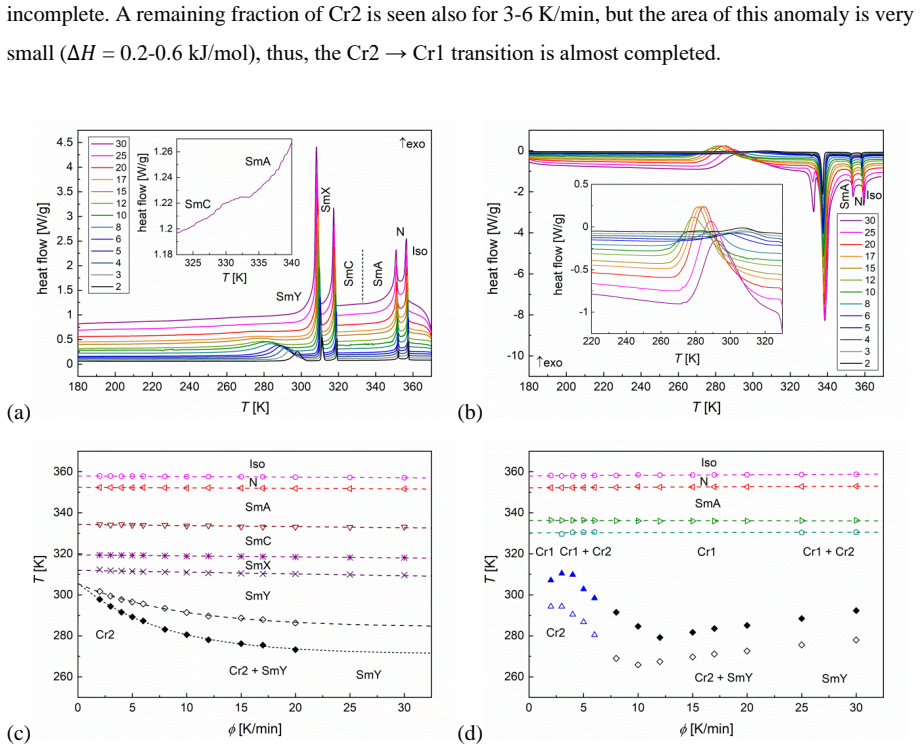
The liquid crystalline 4-pentylphenyl-4'-decyloxythiobenzoate is investigated in various temperature programs for determination of crystallization kinetics and glassforming properties. The Avrami model, Augis-Bennett method and isoconversional method are used. Cooling at the 25-30 K/min rate results in formation of the glass of the tilted smectic Y phase with the herring-bone order within layers. Slower cooling leads to the partial or total (2 K/min) crystallization of the metastable Cr2 phase, which during subsequent heating or annealing in a proper temperature transforms to another Cr1 phase. Heating from the vitrified smectic Y leads to cold crystallization of the pure Cr1 phase or the Cr1/Cr2 mix. Both Cr1 and Cr2 are conformationally disordered crystal phases, which is indicated both by the melting entropy values and the dielectric spectra. The results demonstrate that the energy released during cold crystallization can be tuned by thermal history, highlighting 10OS5 as a candidate for thermal energy storage applications.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript investigates the crystallization kinetics and glass-forming behavior of the liquid crystal 10OS5 (4-pentylphenyl-4'-decyloxythiobenzoate) using differential scanning calorimetry (DSC) and dielectric spectroscopy under varied thermal programs. Rapid cooling (25-30 K/min) produces a glass of the tilted smectic Y phase with herring-bone order, while slower cooling yields partial or complete formation of the metastable Cr2 phase that transforms to Cr1 upon heating or annealing. Heating from the vitrified smectic Y induces cold crystallization to pure Cr1 or Cr1/Cr2 mixtures. Kinetic parameters are derived via the Avrami model, Augis-Bennett method, and isoconversional analysis. Both Cr1 and Cr2 are identified as conformationally disordered crystals based on melting entropy values and dielectric spectra. The central result is that the enthalpy released during cold crystallization can be tuned by thermal history, with the work positioning 10OS5 as a candidate for thermal energy storage applications.
Significance. If the reported tunability of cold-crystallization enthalpy holds under the described conditions, the work provides a clear experimental demonstration of competing crystallization pathways in a liquid crystal system and how thermal history can select between them. Credit is due for the consistent application of three independent kinetic methods (Avrami, Augis-Bennett, and isoconversional) together with cross-validation of phase assignments via both thermodynamic (entropy) and spectroscopic (dielectric) observables. This multi-method approach strengthens the internal consistency of the kinetic and phase conclusions. The potential relevance to thermal energy storage is noted but remains provisional pending further validation.
major comments (1)
- Abstract (final sentence) and the discussion of applications: The claim that the results highlight 10OS5 as a candidate for thermal energy storage applications is load-bearing for the manuscript's concluding emphasis. This inference rests on the assumption that the observed cold-crystallization enthalpies and phase behavior (Cr1, Cr2) remain reproducible and stable without degradation, fatigue, or loss of tunability over repeated cycles. No multi-cycle DSC data or long-term stability tests are reported, despite the explicit identification of both crystal phases as conformationally disordered. This leaves the practical candidacy resting on an untested assumption that could be addressed by adding cycling experiments within the existing experimental scope.
minor comments (2)
- The abstract provides no error bars, raw DSC traces, or quantitative enthalpy values, which would allow readers to directly assess the magnitude and reproducibility of the reported tunability.
- A summary table listing the specific cooling/heating rates, resulting phases, and extracted cold-crystallization enthalpies would improve clarity and facilitate comparison across thermal histories.
Simulated Author's Rebuttal
We thank the referee for the careful review and for acknowledging the consistency of our multi-method kinetic analysis. We address the single major comment below.
read point-by-point responses
-
Referee: Abstract (final sentence) and the discussion of applications: The claim that the results highlight 10OS5 as a candidate for thermal energy storage applications is load-bearing for the manuscript's concluding emphasis. This inference rests on the assumption that the observed cold-crystallization enthalpies and phase behavior (Cr1, Cr2) remain reproducible and stable without degradation, fatigue, or loss of tunability over repeated cycles. No multi-cycle DSC data or long-term stability tests are reported, despite the explicit identification of both crystal phases as conformationally disordered. This leaves the practical candidacy resting on an untested assumption that could be addressed by adding cycling experiments within the existing experimental scope.
Authors: We thank the referee for this constructive observation. The core contribution of the manuscript is the experimental demonstration of competing crystallization pathways and the tunability of cold-crystallization enthalpy through thermal history. The reference to thermal-energy-storage candidacy is presented as a potential implication of this tunability rather than a claim of application readiness. We agree that no multi-cycle DSC data are reported and that long-term stability remains untested. In the revised manuscript we will qualify the final sentence of the abstract and the corresponding discussion paragraph to state explicitly that the candidacy is provisional and that cycling stability and fatigue resistance require separate validation. This textual adjustment will be made while preserving the reported kinetic and phase results. revision: partial
Circularity Check
No circularity: purely experimental analysis with standard models
full rationale
The paper reports DSC measurements of phase behavior and crystallization kinetics in 10OS5, applying the standard Avrami, Augis-Bennett, and isoconversional methods to extract parameters from observed traces. No derivation chain exists that reduces a claimed prediction or result to a fitted input by construction, no self-citations are load-bearing for central claims, and no ansatz or uniqueness theorem is smuggled in. Phase assignments and enthalpy values are direct experimental outputs, rendering the work self-contained against external benchmarks with no self-referential reduction.
Axiom & Free-Parameter Ledger
free parameters (1)
- cooling/heating rates
axioms (2)
- domain assumption Avrami model and isoconversional methods correctly describe the crystallization kinetics in this liquid crystal
- domain assumption Melting entropy and dielectric spectra reliably indicate conformational disorder in Cr1 and Cr2
Reference graph
Works this paper leans on
-
[1]
A.L. Dorfner, D.P. Locoteta, C.D. Messinger, M.R. Ramsey, N.Y. Kim, E. Sadzaglishvili, J.C. Kranick, J.S. Kuehner, C.J. Timony, M. Langton, J.E. Winklarek, L.J. Tucker, J.L. O’Donnell, Non -isothermal cold crystallization of liquid crystalline porphyrins, Soft Matter 19 (2023) 6414-6422, https://doi.org/10.1039/D3SM00760J
-
[2]
A. Dołęga, P.M. Zieliński, Kinetics of non-isothermal cold-crystallization of carbamazepine in the glassy state studied by DSC, J. Non.-Cryst. Sol. 575 (2022) 121198, https://doi.org/10.1016/j.jnoncrysol.2021.121198
-
[3]
K.N. Raftopoulos, I. Łukaszewska, C.B. Calduch, P. Stachak, S. Lalik, E. Hebda, M. Marzec, K. Pielichowski, Hydration and glass transition of hybrid non -isocyanate polyurethanes with POSS inclusions, Polymer 253 (2022) 125010, https://doi.org/10.1016/j.polymer.2022.125010
-
[4]
K. Koperwas, W. Tu, F. Affouard, K. Adrjanowicz, F. Kaskosz, M. Paluch, Pressure Dependence of the Crystallization Rate for the S -Enantiomer and a Racemic Mixture of Ibuprofen, Cryst. Growth Des. 21 (2021) 7075 -7086, https://doi.org/10.1021/acs.cgd.1c00980
-
[5]
K. Turunen, M.R. Yazdani, A. Santasalo -Aarnio, A. Seppälä, Exceptional cold -crystallization kinetics of erythritol - polyelectrolyte enables long -term thermal energy storage, Sol. Energy Mater. Sol. Cells 230 (2021) 111273, https://doi.org/10.1016/j.solmat.2021.111273
-
[6]
S. Puupponen, A. Seppälä, Cold -crystallization of polyelectrolyte absorbed polyol for long -term thermal energy storage, Sol. Energy Mater. Sol. Cells 180 (2018) 59-66, https://doi.org/10.1016/j.solmat.2018.02.013
-
[7]
K. Ishino, H. Shingai, Y. Hikita, I. Yoshikawa, H. Houjou, K. Iwase, Cold Crystallization and the Molecular Structure of Imidazolium-Based Ionic Liquid Crystals with a p -Nitroazobenzene Moiety, ACS Omega 6 (2021) 32869 -32878, https://doi.org/10.1021/acsomega.1c04866
-
[8]
D. Georgopoulos, S. Kripotou, E. Argyraki, A. Kyritsis, P. Pissis, Study of Isothermal Crystallization Kinetics of 5CB with Differential Scanning Calorimetry and Broadband Dielectric Spectroscopy, Mol. Cryst. Liq. Cryst. 611 (2015) 197 -207, https://doi.org/10.1080/15421406.2015.1030259
- [9]
-
[10]
R. Puertas, M.A. Rute, J. Salud, D.O. López, S. Diez, J. Kees van Miltenburg, L.C. Pardo, J.Ll. Tamarit, M. Barrio, M.A. Pérez-Jubindo, M.R. de la Fuente, Thermodynamic, crystallographic, and dielectric study of the nature of glass transitions in cyclo-octanol, Phys. Rev. B 69 (2004) 224202, https://doi.org/10.1103/PhysRevB.69.224202
-
[11]
A.J. Leadbetter, P.A. Tucker, G.W. Gray, A.R. Tajbakhsh, The Phase Behaviour of 4 -n-Hexylphenyl 4 -n-Tetra- Decyloxybenzthiolate (14S6) and 4 -n-Pentylphenyl 4-n-Decyloxybenzthiolate (10S5), Mol. Cryst. Liq. Cryst. Lett. 1 (1985) 19-24
work page 1985
-
[12]
B.M. Ocko, R.J. Birgeneau, J.D. Litster, Crossover to tricritical behavior at the nematic to smectic A transition: An x-ray scattering study, Z. Phys. B Condens. Matter 62 (1986) 487-497, https://doi.org/10.1007/BF01303581
-
[13]
J. Chruściel, H. Kresse, S. Urban, Megahertz dielectric relaxation process in the nematic and smectic phases of two thiol esters (9S5 and 10S5), Liq. Cryst. 11 (1992) 711-718, https://doi.org/10.1080/02678299208029022
-
[14]
A. Żywociński, S.A. Wieczorek, Critical Exponents for Thermal Expansion and Isothermal Compressibility near the Nematic to Smectic-A Phase Transition, 101 (1997) 6970-6976, https://doi.org/10.1021/jp971234g
-
[15]
E. Anesta, G.S. Iannacchione, C.W. Garland, Critical linear thermal expansion in the smectic -A phase near the nematic- smectic phase transition, Phys. Rev. E 70 (2004) 041703, https://doi.org/10.1103/PhysRevE.70.041703
-
[16]
D. Bauman, A. Zięba, E. Mykowska, Oriental behaviour of some homologues of 4 -n-pentyl-phenylthio-4′-n- alkoxybenzoate doped with dichroic dye, Opto-Electron. Rev. 16 (2008) 244-250, https://doi.org/10.2478/s11772-008-0020-5
-
[17]
A. Deptuch, B. Sęk, S. Lalik, M.D. Ossowska-Chruściel, J. Chruściel, M. Marzec, Structural investigation of the liquid crystalline phases of three homologues from the series of 4-pentylphenyl-4’-n-alkyloxythiobenzoates (n = 9, 10, 11), Liq. Cryst. 52 (2025) 157-169, https://doi.org/10.1080/02678292.2024.2417958. 19
-
[18]
M.D. Ediger, P. Harrowell, L. Yu, Crystal growth kinetics exhibit a fragility -dependent decoupling from viscosity, J. Chem. Phys. 128 (2008) 034709, https://doi.org/10.1063/1.2815325
-
[19]
E. Piorkowska, A. Galeski, J. -M. Haudin, Critical assessment of overall crystallization kinetics theories and predictions, Prog. Polym. Sci. 31 (2006) 549-575, https://doi.org/10.1016/j.progpolymsci.2006.05.001
-
[20]
T. Rozwadowski, Y. Yamamura, K. Saito, Interplay between Melt and Cold Crystallization in a Smectic Liquid Crystal, 4‑Pentylphenyl 4‑(trans -4-Pentylcyclohexyl)benzoate, Cryst. Growth Des. 21 (2021) 2777 -2785, https://doi.org/10.1021/acs.cgd.0c01682
-
[21]
Avrami, Kinetics of Phase Change
M. Avrami, Kinetics of Phase Change. II Transformation‐Time Relations for Random Distribution of Nuclei, J. Chem. Phys. 8 (1940) 212-224, https://doi.org/10.1063/1.1750631
-
[22]
I. Avramov, K. Avramova, C. Rüssel, New method to analyze data on overall crystallization kinetics, Cryst. Growth Des. 285 (2005) 394-399, https://doi.org/10.1016/j.jcrysgro.2005.08.024
-
[23]
R. Patki, K. Mezghani, P.J. Philips, Crystallization Kinetics of Polymers, in J.E. Mark (Ed.), Physical Properties of Polymers Handbook, Springer-Verlag, New York 2007, https://doi.org/10.1007/978-0-387-69002-5
-
[24]
A. Sanz, K. Niss, Coupling between Molecular Mobility and Kinetics of Crystal Growth in a Hydrogen -Bonded Liquid, Cryst. Growth Des. 17 (2017) 4628-4636, https://doi.org/10.1021/acs.cgd.7b00484
-
[25]
K. Kołodziejczyk, M. Paluch, K. Grzybowska, A. Grzybowski, Z. Wojnarowska, L. Hawelek, J.D. Ziolo , Relaxation Dynamics and Crystallization Study of Sildenafil in the Liquid and Glassy States , Mol. Pharmaceutics 10 (2013) 2270 -2282, https://doi.org/10.1021/mp300479r
-
[26]
J.A. Augis, J.E. Bennett, Calculation of the Avrami parameters for heterogeneous solid state reactions using a modification of the Kissinger method, J. Therm. Anal. 13 (1978) 283-292, https://doi.org/10.1007/BF01912301
-
[27]
Kissinger, Variation of peak temperature with heating rate in differential thermal analysis, J
H.E. Kissinger, Variation of peak temperature with heating rate in differential thermal analysis, J. Res. Natl. Bur. Stand., 57 (1956) 217-221, https://doi.org/10.6028/jres.057.026
-
[28]
A.A. Abu -Sehly, Study of the kinetics of non -isothermal crystallization in Ge 20Te80 chalcogenide glass, Physica B Condens. Matter 325 (2003) 372-379, https://doi.org/10.1016/S0921-4526(02)01552-1
-
[29]
Ł. Kolek, M. Massalska -Arodź, K. Adrjanowicz, T. Rozwadowski, K. Dychtoń, M. Drajewicz , P. Kula, Molecular dynamics and cold crystallization process in a liquid -crystalline substance with para -, ferro- and antiferro-electric phases as studied by dielectric spectroscopy and scanning calorimetry, J. Mol. Liq. 297 (2020) 111913, https://doi.org/10.1016/j...
-
[30]
Friedman, Kinetics of thermal degradation of char-forming plastics from thermogravimetry
H.L. Friedman, Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to a phenolic plastic, J. Polym. Sci., Part C: Polym. Symp. 6 (1964) 183-195, https://doi.org/10.1002/polc.5070060121
-
[31]
S. Vyazovkin, N. Sbirrazzuoli, Isoconversional Kinetic Analysis of Thermally Stimulated Processes in Polymers, Macromol. Rapid Commun. 27 (2006) 1515-1532, https://doi.org/10.1002/marc.200600404
- [32]
-
[33]
T. Rozwadowski, Ł. Kolek, Design of Crystal Growth Dimensionality in Synthetic Wax: The Kinetics of Nonisothermal Crystallization Processes, J. Phys. Chem. B 127 (2023) 8697-9706, https://doi.org/10.1021/acs.jpcb.3c05158
-
[34]
M.E. Neubert, R.E. Cline, M.J. Zawaski, P.J. Wildman, A. Ekachai, The Effect on Mesomorphic Properties of Substituting a Sulfur for the Ether Oxygen Atom in the Ester Linkage of 4 -Alkylphenyl-4′-Alkyl or Alkoxybenzoates, Mol. Cryst. Liq. Cryst. 76 (1981) 43-77, https://doi.org/10.1080/00268948108074675
-
[35]
Gaussian 16, Revision C.01, M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, G.A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A.V. Marenich, J. Bloino, B.G. Janesko, R. Gomperts, B. Mennucci, H.P. Hratchian, J.V. Ortiz, A.F. Izmaylov, J.L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. E...
work page 2019
-
[36]
C. Lee, W. Yang, R.G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B, 37 (1988) 785-789, https://doi.org/10.1103/PhysRevB.37.785
-
[37]
Becke, Density‐functional thermochemistry
A.D. Becke, Density‐functional thermochemistry. III. The role of exact exchange, J. Chem. Phys. 98 (1993) 5648 -5652, https://doi.org/10.1063/1.464913
-
[38]
P.J. Stephens, F.J. Devlin, C.F. Chabalowski, M.J. Frisch, Ab Initio Calculation of Vibrational Absorption and Circular Dichroism Spectra Using Density Functional Force Fields, J. Phys. Chem. 98 (1994) 11623 -11627, https://doi.org/10.1021/j100096a001
-
[39]
URL https://onlinelibrary.wiley.com/ doi/abs/10.1002/jcc.21759
S. Grimme, S. Ehrlich, L. Goerigk, Effect of the damping function in dispersion corrected density functional theory, J. Comput. Chem. 32 (2011) 1456-1465, https://doi.org/10.1002/jcc.21759
-
[40]
F. Weigend, R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy, Phys. Chem. Chem. Phys. 7 (2005) 3297-3305, https://doi.org/10.1039/B508541A
-
[41]
M.D. Hanwell, D.E. Curtis, D.C. Lonie, T. Vandermeersch , E. Zurek, G.R. Hutchison, Avogadro: an advanced semantic chemical editor, visualization, and analysis platform, J. Cheminf. 4 (2012) 17, https://doi.org/10.1186/1758-2946-4-17
-
[42]
K.S. Cole, R. H. Cole, Dispersion and Absorption in Dielectrics I. Alternating Current Characteristics, J. Chem. Phys. 9 (1941) 341-351, https://doi.org/10.1063/1.1750906
-
[43]
W. Haase, S. Wróbel (Eds.), Relaxation phenomena. Liquid crystals, magnetic systems, polymers, high -Tc superconductors, metallic glasses, Springer-Verlag, Berlin Heidelberg 2003, https://doi.org/10.1007/978-3-662-09747-2
-
[44]
S. Urban, M. Geppi, Comparison of the dielectric and NMR results for liquid crystals: dynamic aspects, Magn. Reson. Chem. 52 (2014) 656-663, https://doi.org/10.1002/mrc.4100
-
[45]
J. Czub, U. Gubernat, B. Gestblom, R. Dąbrowski, S. Urban, Dielectric Studies of Smectogenic Members of the 4’-alkyl- 4-cyanobiphenyl (nCB) Homologous Series, Z. Naturforsch. A 59 (2004) 316-324, https://doi.org/10.1515/zna-2004-0602
-
[46]
M. Samet, V. Levchenko, G. Boiteux, G. Seytre, A. Kallel, A. Serghe, Electrode polarization vs. Maxwell-Wagner-Sillars interfacial polarization in dielectric spectra of materials: Characteristic frequencies and scaling laws, J. Chem. Phys. 142 (2015) 194703, https://doi.org/10.1063/1.4919877
-
[47]
R. Böhmer, K.L. Ngai, C.A. Angell, D.J. Plazek, Nonexponential relaxations in strong and fragile glass formers, J. Chem. Phys. 99 (1993) 4201-4209, https://doi.org/10.1063/1.466117
-
[48]
A. Drzewicz, M. Jasiurkowska -Delaporte, P. Kula, E. Juszyńska-Gałązka, Effect of the linking bridge type on the self- assembly behaviour of 2’,3’ -difluoroterphenyl derivatives, Phys. Chem. Chem. Phys. 26 (2024) 8748 -8760, https://doi.org/10.1039/D4CP00133H
-
[49]
B. Wunderlich, A classification of molecules, phases, and transitions as recognized by thermal analysis, Thermochim. Acta 340-341 (1999) 37-52, https://doi.org/10.1016/S0040-6031(99)00252-X
-
[50]
M. Jasiurkowska-Delaporte, M. Massalska-Arodź, Molecular dynamics of 4-propyl-4’-thiocyanatobiphenyl (3BT) in the strong glass-forming smectic E phase, J. Mol. Liq. 241 (2017) 355-358, https://doi.org/10.1016/j.molliq.2017.06.027
-
[51]
R. Brand, P. Lunkenheimer, A. Loidl, Relaxation dynamics in plastic crystals, J. Chem. Phys. 116 (2002) 10386 -10401, https://doi.org/10.1063/1.1477186
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.