Recognition: 2 theorem links
· Lean TheoremMechanical Softening of Vero Cells Induced by an Attenuated Measles Vaccine Virus
Pith reviewed 2026-05-13 17:41 UTC · model grok-4.3
The pith
Attenuated measles vaccine virus causes Vero cells to soften by about 35 percent within 24 hours.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
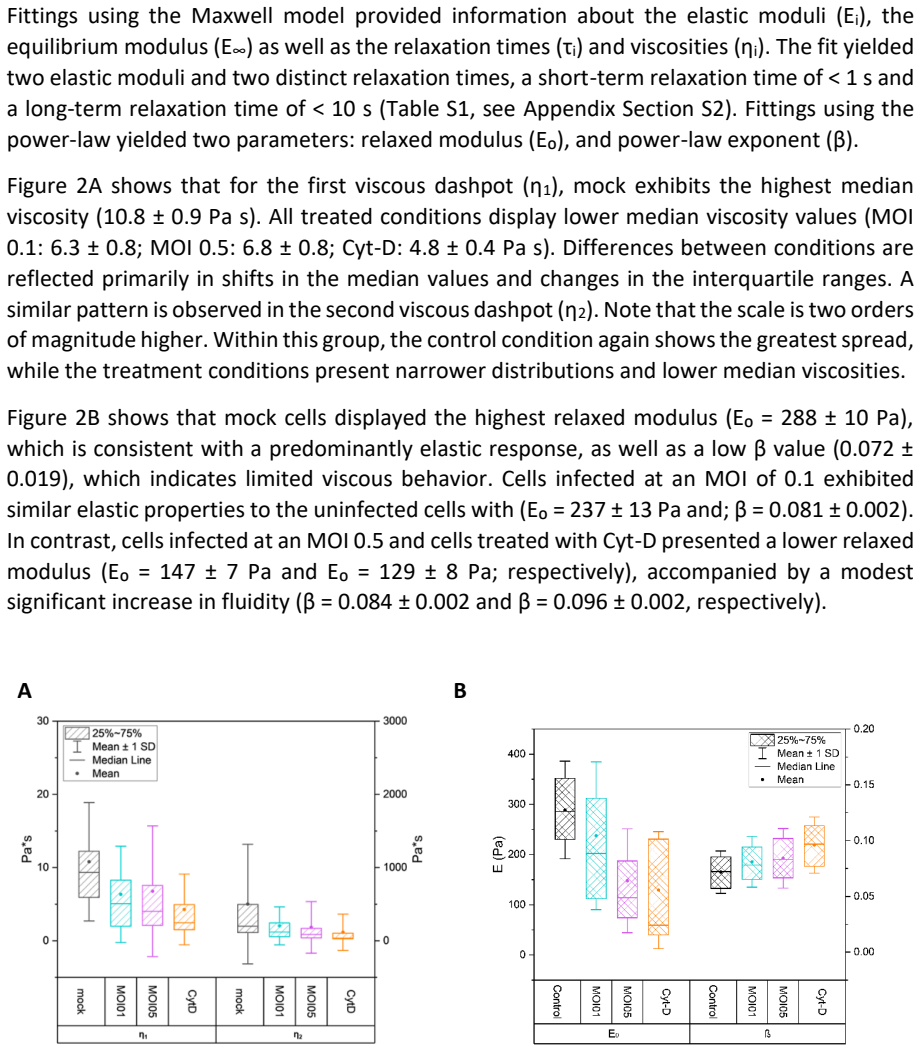
Within 24 hours post infection, cells infected at a multiplicity of infection of 0.5 exhibited an approximately thirty five percent decrease in median of the Young modulus relative to uninfected controls, indicating substantial cellular softening. Corresponding shifts in viscoelastic behavior were observed, including reductions in the relaxed modulus and in viscosities, effects comparable to those induced by cytochalasin D mediated actin depolymerization. Confocal microscopy further revealed a dependent reorganization of the cytoskeleton upon infection, marked by altered F actin distribution and changes in filament architecture, suggesting that actin remodeling contributes to the altered vis
What carries the argument
AFM force-distance measurements in the perinuclear region combined with confocal microscopy of F-actin, used to compare elastic modulus, viscoelastic parameters, and cytoskeletal architecture between infected and control cells.
Load-bearing premise
The measured softening and viscoelastic shifts result from virus-induced actin remodeling rather than cell density differences, culture conditions, or AFM calibration artifacts.
What would settle it
AFM measurements on virus-infected cells with artificially stabilized actin or on density-matched uninfected controls that show no softening would falsify the central claim.
Figures

read the original abstract
Quantitative characterization of biophysical alterations caused by viral infection remains at an early stage. In this study, we examined the mechanical response of Vero cells after exposure to an attenuated Measles Vaccine Virus using atomic force microscopy (AFM) in combination with confocal microscopy. AFM force distance measurements were conducted in the perinuclear region to evaluate changes in elastic and viscoelastic properties. Within 24 hours post infection, cells infected at a multiplicity of infection of 0.5 exhibited an approximately thirty five percent decrease in median of the Young modulus relative to uninfected controls, indicating substantial cellular softening. Corresponding shifts in viscoelastic behavior were observed, including reductions in the relaxed modulus and in viscosities (effects comparable to those induced by cytochalasin D mediated actin depolymerization). Confocal microscopy further revealed a dependent reorganization of the cytoskeleton upon infection, marked by altered F actin distribution and changes in filament architecture. These findings suggest that actin remodeling contributes to the altered viscoelastic properties observed during infection. This work proposes a straightforward and complementary approach for characterizing virus cell interactions by integrating AFM with confocal imaging.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript reports that Vero cells infected with attenuated measles vaccine virus (MOI 0.5) exhibit an approximately 35% decrease in median Young's modulus in the perinuclear region within 24 hours post-infection relative to uninfected controls, as quantified by AFM force-distance measurements. This softening is accompanied by shifts in viscoelastic parameters (relaxed modulus and viscosities) comparable to cytochalasin D treatment, and confocal imaging shows F-actin reorganization, which the authors link mechanistically to the observed biophysical changes.
Significance. If the central result holds after addressing controls, the work supplies quantitative AFM-based evidence for virus-induced cellular softening tied to cytoskeletal remodeling. The integration of AFM force spectroscopy with confocal microscopy provides a complementary experimental approach for characterizing virus-cell mechanical interactions, with the reported 35% modulus drop and viscoelastic parallels to actin depolymerization offering a concrete, falsifiable observation.
major comments (2)
- [Results (AFM force-distance analysis)] Results section on AFM measurements: the 35% median Young-modulus reduction is reported without quantitative controls for cell spread area, height, or local monolayer density at the 24 h measurement time point. Because the Hertz (or similar) contact model used for perinuclear indentation is sensitive to thin-layer geometry on stiff substrates, differences in these parameters between infected and control cohorts could produce apparent softening even without intrinsic cytoskeletal change; the manuscript must either demonstrate morphological matching or apply appropriate thin-layer corrections.
- [Methods and Results] Methods and Results: sample sizes, exact statistical tests for the median comparison, error bars or confidence intervals on the modulus values, and the precise fitting model (including any thin-layer or viscoelastic extensions) are not reported. These omissions prevent assessment of whether the observed shift is statistically robust and reproducible.
minor comments (2)
- [Abstract] Abstract and text: the phrase 'dependent reorganization' is unclear; specify whether the actin changes are statistically dependent on infection or simply observed in parallel.
- [Figures] Figure captions and text: ensure all AFM curves and confocal images include scale bars, number of cells/curves per condition, and explicit indication of which data correspond to the reported median values.
Simulated Author's Rebuttal
We thank the referee for the constructive comments, which help clarify the robustness of our AFM measurements. We address each major point below and will revise the manuscript accordingly to strengthen the presentation of controls and statistical details.
read point-by-point responses
-
Referee: [Results (AFM force-distance analysis)] Results section on AFM measurements: the 35% median Young-modulus reduction is reported without quantitative controls for cell spread area, height, or local monolayer density at the 24 h measurement time point. Because the Hertz (or similar) contact model used for perinuclear indentation is sensitive to thin-layer geometry on stiff substrates, differences in these parameters between infected and control cohorts could produce apparent softening even without intrinsic cytoskeletal change; the manuscript must either demonstrate morphological matching or apply appropriate thin-layer corrections.
Authors: We agree that morphological parameters could influence apparent modulus values under the Hertz model. In the revised manuscript we will add quantitative measurements of cell spread area, height, and local monolayer density at the 24 h time point for both cohorts, obtained from the same AFM and confocal datasets. We will also report indentation depths relative to cell height and, if needed, apply thin-layer corrections or restrict analysis to regions where substrate effects are negligible. These additions will confirm that the observed 35% softening reflects cytoskeletal changes rather than geometric differences. revision: yes
-
Referee: [Methods and Results] Methods and Results: sample sizes, exact statistical tests for the median comparison, error bars or confidence intervals on the modulus values, and the precise fitting model (including any thin-layer or viscoelastic extensions) are not reported. These omissions prevent assessment of whether the observed shift is statistically robust and reproducible.
Authors: We acknowledge these reporting gaps. The revised manuscript will explicitly state the sample sizes (number of cells and force curves per condition), the statistical test applied to the median comparison (Mann-Whitney U test), and will include error bars or 95% confidence intervals on all modulus and viscoelastic parameter values. We will also provide a complete description of the contact model (Hertzian fit to the initial 10-30% of the force curve) together with the viscoelastic model parameters and any thin-layer considerations. These details will allow full assessment of statistical robustness. revision: yes
Circularity Check
No circularity: purely experimental measurements with no derivation chain
full rationale
The manuscript presents direct AFM force-distance measurements and confocal imaging of Vero cells post-infection, reporting an observed ~35% drop in median Young modulus as a raw experimental outcome. No equations, models, or derivations are invoked that reduce this result to a fitted parameter or self-referential input; the viscoelastic shifts are likewise reported from raw data curves without any predictive step that loops back to the same measurements by construction. Self-citations, if present, are not load-bearing for any central claim, and the work contains no uniqueness theorems or ansatzes imported from prior author work.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption AFM force-distance curves in the perinuclear region can be converted to Young's modulus and viscoelastic parameters using established contact-mechanics models
- domain assumption Changes in F-actin distribution observed by confocal microscopy reflect functional cytoskeletal remodeling that alters mechanical properties
Lean theorems connected to this paper
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclear?
unclearRelation between the paper passage and the cited Recognition theorem.
Within 24 hours post infection, cells infected at a multiplicity of infection of 0.5 exhibited an approximately thirty five percent decrease in median of the Young modulus relative to uninfected controls
-
IndisputableMonolith/Foundation/RealityFromDistinction.leanreality_from_one_distinction unclear?
unclearRelation between the paper passage and the cited Recognition theorem.
AFM force-distance measurements were conducted in the perinuclear region... fitted to a 5-element Maxwell model
What do these tags mean?
- matches
- The paper's claim is directly supported by a theorem in the formal canon.
- supports
- The theorem supports part of the paper's argument, but the paper may add assumptions or extra steps.
- extends
- The paper goes beyond the formal theorem; the theorem is a base layer rather than the whole result.
- uses
- The paper appears to rely on the theorem as machinery.
- contradicts
- The paper's claim conflicts with a theorem or certificate in the canon.
- unclear
- Pith found a possible connection, but the passage is too broad, indirect, or ambiguous to say the theorem truly supports the claim.
Reference graph
Works this paper leans on
-
[1]
Elasticity of normal and cancerous human bladder cells studied by scanning force microscopy
Lekka M, Laidler P, Gil D, Lekki J, Stachura Z, Hrynkiewicz AZ. Elasticity of normal and cancerous human bladder cells studied by scanning force microscopy. Eur Biophys J Biophy. 1999;28(4):312-6
work page 1999
-
[2]
Guck J, Schinkinger S, Lincoln B, Wottawah F, Ebert S, Romeyke M, et al. Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys J. 2005;88(5):3689-98
work page 2005
-
[3]
Harnessing atomic force microscopy -based single -cell analysis to advance physical oncology
Li M. Harnessing atomic force microscopy -based single -cell analysis to advance physical oncology. Microsc Res Techniq. 2024;87(4):631-59
work page 2024
-
[4]
Measuring (biological) materials mechanics with atomic force microscopy
Weber A, Zbiral B, Iturri J, Benitez R, Toca-Herrera JL. Measuring (biological) materials mechanics with atomic force microscopy. 2. Influence of the loading rate and applied force (colloidal particles). Microsc Res Techniq. 2021;84(5):1078-88
work page 2021
-
[5]
afmToolkit: an R Package for Automated AFM Force-Distance Curves Analysis
Benitez R, Bolos VJ, Toca -Herrera JL. afmToolkit: an R Package for Automated AFM Force-Distance Curves Analysis. The R Journal, 2017
work page 2017
-
[6]
Determination of elastic moduli of thin layers of soft material using the atomic force microscope
Dimitriadis EK, Horkay F, Maresca J, Kachar B, Chadwick RS. Determination of elastic moduli of thin layers of soft material using the atomic force microscope. Biophys J. 2002;82(5):2798-810
work page 2002
-
[7]
Rotsch C, Radmacher M. Drug-induced changes of cytoskeletal structure and mechanics in fibroblasts: An atomic force microscopy study. Biophys J. 2000;78(1):520-35
work page 2000
-
[8]
Measuring (biological) materials mechanics with atomic force microscopy
Weber A, Benitez R, Toca-Herrera JL. Measuring (biological) materials mechanics with atomic force microscopy. 4. Determination of viscoelastic cell properties from stress relaxation experiments. Microsc Res Techniq. 2022;85(10):3284-95
work page 2022
-
[9]
Celebrating the inauguration of the journal: Biomechanics and Modeling in Mechanobiology
Fung YC. Celebrating the inauguration of the journal: Biomechanics and Modeling in Mechanobiology. Biomech Model Mechan. 2002;1(1):3-4
work page 2002
-
[10]
Estrogen Modulates Epithelial Breast Cancer Cell Mechanics and Cell-to-Cell Contacts
Zbiral B, Weber A, Iturri J, Vivanco MDM, Toca-Herrera JL. Estrogen Modulates Epithelial Breast Cancer Cell Mechanics and Cell-to-Cell Contacts. Materials. 2021;14(11)
work page 2021
-
[11]
Abuhattum S, Mokbel D, Müller P, Soteriou D, Guck J, Aland S. Article An explicit model to extract viscoelastic properties of cells from AFM force-indentation curves. Iscience. 2022;25(4)
work page 2022
-
[12]
Substrate stiffness modulates the viscoelastic properties of MCF-7 cells
Gil-Redondo JC, Weber A, Zbiral B, Vivanco MD, Toca-Herrera JL. Substrate stiffness modulates the viscoelastic properties of MCF-7 cells. J Mech Behav Biomed. 2022;125
work page 2022
-
[13]
Scaling the microrheology of living cells - art
Fabry B, Maksym GN, Butler JP, Glogauer M, Navajas D, Fredberg JJ. Scaling the microrheology of living cells - art. no. 148102. Phys Rev Lett. 2001;87(14)
work page 2001
-
[14]
Nonlinear viscoelasticity of adherent cells is controlled by cytoskeletal tension
Kollmannsberger P, Mierke CT, Fabry B. Nonlinear viscoelasticity of adherent cells is controlled by cytoskeletal tension. Soft Matter. 2011;7(7):3127-32
work page 2011
-
[15]
Hauke, L., Primeßnig, A., Eltzner, B., Radwitz, J., Huckemann, S.F., Rehfeldt, F. FilamentSensor 2.0: An open-source modular toolbox for 2D/3D cytoskeletal filament tracking PLOS ONE 18(2): e0279336
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.