Recognition: 2 theorem links
· Lean TheoremTranscriptomic Models for Immunotherapy Response Prediction Show Limited Cross-cohort Generalisability
Pith reviewed 2026-05-10 19:13 UTC · model grok-4.3
The pith
Transcriptomic models for predicting immune checkpoint inhibitor response show limited generalisability across independent patient cohorts.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Benchmarking of five bulk RNA-seq models and four scRNA-seq models on unseen cohorts demonstrates modest predictive performance overall, with bulk models near chance level and scRNA-seq models showing only slight improvement, accompanied by sparse and non-reproducible immune-related pathway signals across models and datasets.
What carries the argument
Cross-cohort benchmarking of transcriptomic ICI response predictors on independent public datasets, exposing gaps in performance and biomarker consistency.
If this is right
- Bulk RNA-seq models need substantial refinement to exceed near-chance prediction on new groups.
- Single-cell models require targeted adjustments to convert marginal gains into reliable performance.
- Greater emphasis on consistent immune-related biological signals would improve model reproducibility.
- Standardised preprocessing and domain adaptation methods are required to enhance transfer across cohorts.
Where Pith is reading between the lines
- Transcriptomic signals alone may need supplementation with additional data types to overcome cohort-specific noise.
- Unmeasured technical variations between studies could explain much of the observed inconsistency in biomarkers.
- Focusing future models on core immune mechanisms shared across cohorts could raise transferability without larger datasets.
Load-bearing premise
The independent test datasets represent real-world clinical variation without batch effects, selection biases, or preprocessing differences that distort measured performance.
What would settle it
Observing that any of the nine models achieves high accuracy consistently across several additional unseen cohorts after minimal adaptation would challenge the limited generalisability conclusion.
Figures




read the original abstract
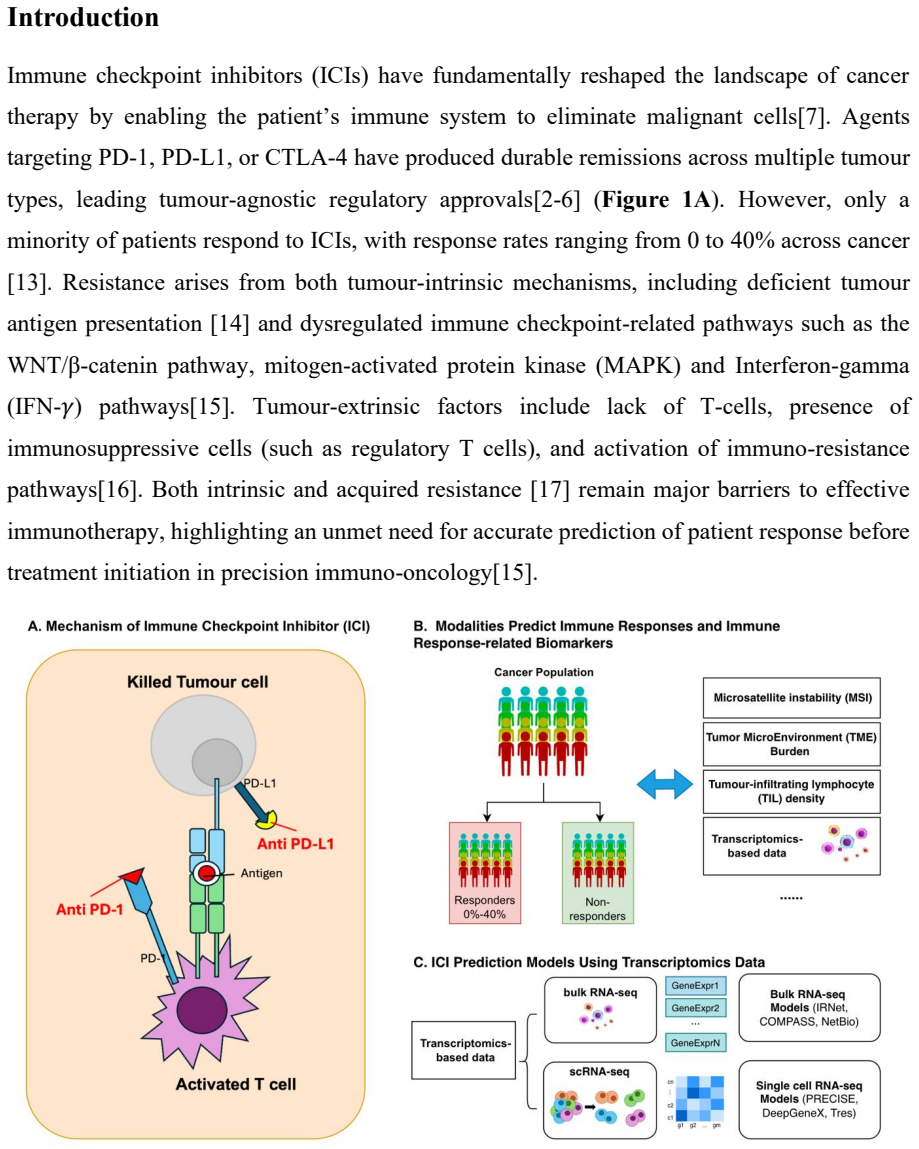
Immune checkpoint inhibitors (ICIs) have transformed cancer therapy; yet substantial proportion of patients exhibit intrinsic or acquired resistance, making accurate pre-treatment response prediction a critical unmet need. Transcriptomics-based biomarkers derived from bulk and single-cell RNA sequencing (scRNA-seq) offer a promising avenue for capturing tumour-immune interactions, yet the cross-cohort generalisability of existing prediction models remains unclear.We systematically benchmark nine state-of-the-art transcriptomic ICI response predictors, five bulk RNA-seq-based models (COMPASS, IRNet, NetBio, IKCScore, and TNBC-ICI) and four scRNA-seq-based models (PRECISE, DeepGeneX, Tres and scCURE), using publicly available independent datasets unseen during model development. Overall, predictive performance was modest: bulk RNA-seq models performed at or near chance level across most cohorts, while scRNA-seq models showed only marginal improvements. Pathway-level analyses revealed sparse and inconsistent biomarker signals across models. Although scRNA-seq-based predictors converged on immune-related programs such as allograft rejection, bulk RNA-seq-based models exhibited little reproducible overlap. PRECISE and NetBio identified the most coherent immune-related themes, whereas IRNet predominantly captured metabolic pathways weakly aligned with ICI biology. Together, these findings demonstrate the limited cross-cohort robustness and biological consistency of current transcriptomic ICI prediction models, underscoring the need for improved domain adaptation, standardised preprocessing, and biologically grounded model design.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript systematically benchmarks nine transcriptomic ICI response predictors (five bulk RNA-seq models: COMPASS, IRNet, NetBio, IKCScore, TNBC-ICI; four scRNA-seq models: PRECISE, DeepGeneX, Tres, scCURE) on publicly available independent datasets. It reports modest overall performance, with bulk models at or near chance level across most cohorts and scRNA-seq models showing only marginal gains, alongside sparse and inconsistent pathway-level biomarker signals (e.g., immune-related themes in PRECISE and NetBio but metabolic focus in IRNet), concluding limited cross-cohort generalisability and calling for better domain adaptation and standardization.
Significance. If the empirical results hold, the work is significant as a comprehensive reference highlighting the robustness challenges facing current transcriptomic predictors in immuno-oncology. The multi-model, multi-cohort design and pathway consistency analysis provide concrete evidence that motivates improved methods; the reliance on public data is a reproducibility strength.
major comments (1)
- [Methods (independent datasets)] Methods section on independent datasets: the central claim that all evaluation cohorts are 'unseen during model development' rests on an assertion without a supporting cross-reference table or appendix that systematically compares the original training cohorts from the nine source papers against the test cohorts (e.g., potential overlaps with GSE78220 or IMvigor210). This verification is load-bearing for interpreting the 'near chance' performance as evidence of limited generalisability rather than an artifact of data leakage.
minor comments (2)
- [Abstract] Abstract: the description of 'modest' performance and 'sparse' signals would be clearer with explicit mention of the number of evaluation cohorts and the primary performance metric (e.g., AUC) used.
- [Results (pathway analyses)] Results (pathway analyses): the criteria for declaring 'inconsistent' or 'coherent' biomarker themes across models are not stated explicitly, making it hard to assess reproducibility of the biological findings.
Simulated Author's Rebuttal
We thank the referee for their constructive feedback and positive evaluation of the study's significance. We address the single major comment below and will incorporate the requested clarification in the revised manuscript.
read point-by-point responses
-
Referee: Methods section on independent datasets: the central claim that all evaluation cohorts are 'unseen during model development' rests on an assertion without a supporting cross-reference table or appendix that systematically compares the original training cohorts from the nine source papers against the test cohorts (e.g., potential overlaps with GSE78220 or IMvigor210). This verification is load-bearing for interpreting the 'near chance' performance as evidence of limited generalisability rather than an artifact of data leakage.
Authors: We agree that an explicit cross-reference table would improve transparency and strengthen the manuscript. Although our evaluation cohorts were chosen after reviewing the training cohort descriptions in each of the nine original publications (COMPASS, IRNet, NetBio, IKCScore, TNBC-ICI, PRECISE, DeepGeneX, Tres, scCURE) and confirming no overlap with the test sets (including GSE78220 and IMvigor210), this verification was not documented in a dedicated table. In the revised version we will add Supplementary Table S1, which will list (i) the exact training cohorts and accession numbers reported in each source paper and (ii) the independent evaluation cohorts used here, with a clear statement of non-overlap. This addition will make the independence claim fully verifiable and support the interpretation of the observed performance as evidence of limited cross-cohort generalisability rather than data leakage. revision: yes
Circularity Check
No circularity: pure empirical benchmarking with no derivations or self-referential equations
full rationale
This is a systematic benchmarking study that evaluates nine pre-existing transcriptomic ICI response predictors on publicly available independent datasets. The abstract and structure contain no equations, fitted parameters, ansatzes, or derivation chains. Performance metrics (AUC, etc.) are computed directly from external test cohorts rather than being defined in terms of the models' own training data or prior outputs. Self-citations to the original model papers are standard and non-load-bearing for the central claim of limited generalisability. The skeptic concern about possible undetected cohort overlap is a validity issue for the evaluation design, not a circularity in any derivation step.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption The selected independent test cohorts are free from batch effects and representative of the target population for ICI response prediction.
Lean theorems connected to this paper
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclearWe systematically benchmark nine state-of-the-art transcriptomic ICI response predictors... using publicly available independent datasets unseen during model development. Overall, predictive performance was modest: bulk RNA-seq models performed at or near chance level across most cohorts
-
IndisputableMonolith/Foundation/AbsoluteFloorClosure.leanreality_from_one_distinction unclearPathway-level analyses revealed sparse and inconsistent biomarker signals across models... PRECISE and NetBio identified the most coherent immune-related themes
Reference graph
Works this paper leans on
-
[1]
Network-based machine learning approach to predict immunotherapy response in cancer patients
Kong J, Ha D, Lee J, Kim I, Park M, Im S-H, et al. Network-based machine learning approach to predict immunotherapy response in cancer patients. Nat Commun 2022;13:3703. https://doi.org/10.1038/s41467-022-31535-6
-
[2]
IRnet: Immunotherapy response prediction using pathway knowledge-informed graph neural network
Jiang Y, Immadi MS, Wang D, Zeng S, On Chan Y, Zhou J, et al. IRnet: Immunotherapy response prediction using pathway knowledge-informed graph neural network. J Adv Res 2024;72:319–31. https://doi.org/10.1016/j.jare.2024.07.036
-
[3]
Generalizable AI predicts immunotherapy outcomes across cancers and treatments
Shen W, Nguyen TH, Li MM, Huang Y, Moon I, Nair N, et al. Generalizable AI predicts immunotherapy outcomes across cancers and treatments. medRxiv 2025:2025.05.01.25326820. https://doi.org/10.1101/2025.05.01.25326820
-
[4]
Pinhasi A, Yizhak K. Uncovering gene and cellular signatures of immune checkpoint response via machine learning and single-cell RNA-seq. Npj Precis Oncol 2025;9:95. https://doi.org/10.1038/s41698-025-00883-z
-
[5]
Deep neural network modeling identifies biomarkers of response to immune-checkpoint therapy
Kang Y, Vijay S, Gujral TS. Deep neural network modeling identifies biomarkers of response to immune-checkpoint therapy. iScience 2022;25. https://doi.org/10.1016/j.isci.2022.104228
-
[6]
A T cell resilience model associated with response to immunotherapy in multiple tumor types
Zhang Y, Vu T, Palmer DC, Kishton RJ, Gong L, Huang J, et al. A T cell resilience model associated with response to immunotherapy in multiple tumor types. Nat Med 2022;28:1421–31. https://doi.org/10.1038/s41591-022-01799-y
-
[7]
Immune checkpoint signaling and cancer immunotherapy
He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res 2020;30:660–9. https://doi.org/10.1038/s41422-020-0343-4
-
[8]
PD-1 and PD-L1 in cancer immunotherapy: clinical implications and future considerations
Jiang Y, Chen M, Nie H, Yuan Y. PD-1 and PD-L1 in cancer immunotherapy: clinical implications and future considerations. Hum Vaccines Immunother 2019;15:1111–22. https://doi.org/10.1080/21645515.2019.1571892
-
[9]
Role of Immunotherapy in the Treatment of Cancer: A Systematic Review
Ling SP, Ming LC, Dhaliwal JS, Gupta M, Ardianto C, Goh KW, et al. Role of Immunotherapy in the Treatment of Cancer: A Systematic Review. Cancers 2022;14:5205. https://doi.org/10.3390/cancers14215205
-
[10]
Adaptive immune resistance: How cancer protects from immune attack
Ribas A. Adaptive immune resistance: How cancer protects from immune attack. Cancer Discov 2015;5:915–9. https://doi.org/10.1158/2159-8290.CD-15-0563
-
[11]
Immunotherapy in Melanoma: Recent Advances and Future Directions
Knight A, Karapetyan L, Kirkwood JM. Immunotherapy in Melanoma: Recent Advances and Future Directions. Cancers 2023;15:1106. https://doi.org/10.3390/cancers15041106
-
[12]
Jin M, Fang J, Peng J, Wang X, Xing P, Jia K, et al. PD-1/PD-L1 immune checkpoint blockade in breast cancer: research insights and sensitization strategies. Mol Cancer 2024;23:266. https://doi.org/10.1186/s12943-024-02176-8
-
[13]
Primary, Adaptive and Acquired Resistance to Cancer Immunotherapy
Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, Adaptive and Acquired Resistance to Cancer Immunotherapy. Cell 2017;168:707. https://doi.org/10.1016/j.cell.2017.01.017
-
[14]
Cell-Intrinsic Barriers of T Cell-Based Immunotherapy
Ghoneim HE, Zamora AE, Thomas PG, Youngblood BA. Cell-Intrinsic Barriers of T Cell-Based Immunotherapy. Trends Mol Med 2016;22:1000–11. https://doi.org/10.1016/j.molmed.2016.10.002
-
[15]
Immune evasion in cancer: mechanisms and cutting-edge therapeutic approaches
Tufail M, Jiang C-H, Li N. Immune evasion in cancer: mechanisms and cutting-edge therapeutic approaches. Signal Transduct Target Ther 2025;10:227. https://doi.org/10.1038/s41392-025-02280-1. 38
-
[16]
Huang R, Chen M, Li H, An X, Xue C, Hu A, et al. Effect of chemotherapy alone or combined with immunotherapy for locally advanced or metastatic genitourinary small cell carcinoma: a real-world retrospective study. BMC Cancer 2023;23:1002. https://doi.org/10.1186/s12885-023-11473-2
-
[17]
The interaction of innate immune and adaptive immune system
Wang R, Lan C, Benlagha K, Camara NOS, Miller H, Kubo M, et al. The interaction of innate immune and adaptive immune system. MedComm 2024;5:e714. https://doi.org/10.1002/mco2.714
-
[18]
Niemeijer AN, Leung D, Huisman MC, Bahce I, Hoekstra OS, van Dongen G a. MS, et al. Whole body PD-1 and PD-L1 positron emission tomography in patients with non- small-cell lung cancer. Nat Commun 2018;9:4664. https://doi.org/10.1038/s41467-018- 07131-y
-
[19]
McGrail DJ, Pilié PG, Rashid NU, Voorwerk L, Slagter M, Kok M, et al. High tumor mutation burden fails to predict immune checkpoint blockade response across all cancer types. Ann Oncol 2021;32:661–72. https://doi.org/10.1016/j.annonc.2021.02.006
-
[20]
Greco L, Rubbino F, Dal Buono A, Laghi L. Microsatellite Instability and Immune Response: From Microenvironment Features to Therapeutic Actionability—Lessons from Colorectal Cancer. Genes 2023;14:1169. https://doi.org/10.3390/genes14061169
-
[21]
Neoantigens: promising targets for cancer therapy
Xie N, Shen G, Gao W, Huang Z, Huang C, Fu L. Neoantigens: promising targets for cancer therapy. Signal Transduct Target Ther 2023;8:9. https://doi.org/10.1038/s41392- 022-01270-x
-
[22]
Immune correlates and mechanisms of TIL therapy efficacy: current insights and knowledge gaps
Navarro Rodrigo B, Ortiz Miranda Y, Corria-Osorio J, Coukos G, Harari A. Immune correlates and mechanisms of TIL therapy efficacy: current insights and knowledge gaps. Trends Cancer 2025;11:993–1004. https://doi.org/10.1016/j.trecan.2025.08.002
-
[23]
Advances and prospects of biomarkers for immune checkpoint inhibitors
Yamaguchi H, Hsu J-M, Sun L, Wang S-C, Hung M-C. Advances and prospects of biomarkers for immune checkpoint inhibitors. Cell Rep Med 2024;5:101621. https://doi.org/10.1016/j.xcrm.2024.101621
-
[24]
Microsatellite- Stable Tumors with High Mutational Burden Benefit from Immunotherapy
Goodman AM, Sokol ES, Frampton GM, Lippman SM, Kurzrock R. Microsatellite- Stable Tumors with High Mutational Burden Benefit from Immunotherapy. Cancer Immunol Res 2019;7:1570–3. https://doi.org/10.1158/2326-6066.CIR-19-0149
-
[25]
Das R, Deb S, Suresh PK. TMB as a predictive biomarker for ICI response in TNBC: current evidence and future directions for augmented anti-tumor responses. Clin Exp Med 2025;26:25. https://doi.org/10.1007/s10238-025-01892-9
-
[26]
Lowe R, Shirley N, Bleackley M, Dolan S, Shafee T. Transcriptomics technologies. PLoS Comput Biol 2017;13:e1005457. https://doi.org/10.1371/journal.pcbi.1005457
-
[27]
Grasso CS, Tsoi J, Onyshchenko M, Abril-Rodriguez G, Ross-Macdonald P, Wind- Rotolo M, et al. Conserved interferon-γ signaling drives clinical response to immune checkpoint blockade therapy in melanoma. Cancer Cell 2020;38:500-515.e3. https://doi.org/10.1016/j.ccell.2020.08.005
-
[28]
Morimoto T, Nakazawa T, Maeoka R, Matsuda R, Nakamura M, Nishimura F, et al. Bulk RNA sequencing reveals the comprehensive genetic characteristics of human cord blood-derived natural killer cells. Regen Ther 2024;25:367–76. https://doi.org/10.1016/j.reth.2024.02.002
-
[29]
Liu H, Sima X, Xiao B, Gulizeba H, Zhao S, Zhou T, et al. Integrated analysis of single- cell and bulk RNA sequencing data reveals a myeloid cell-related regulon predicting neoadjuvant immunotherapy response across cancers. J Transl Med 2024;22:486. https://doi.org/10.1186/s12967-024-05123-9
-
[30]
Ott PA, Bang Y-J, Piha-Paul SA, Razak ARA, Bennouna J, Soria J-C, et al. T-Cell– Inflamed Gene-Expression Profile, Programmed Death Ligand 1 Expression, and Tumor Mutational Burden Predict Efficacy in Patients Treated With Pembrolizumab Across 20 39 Cancers: KEYNOTE-028. J Clin Oncol 2019;37:318–27. https://doi.org/10.1200/JCO.2018.78.2276
-
[31]
Signatures of T cell dysfunction and exclusion predict cancer immunotherapy response
Jiang P, Gu S, Pan D, Fu J, Sahu A, Hu X, et al. Signatures of T cell dysfunction and exclusion predict cancer immunotherapy response. Nat Med 2018;24:1550–8. https://doi.org/10.1038/s41591-018-0136-1
-
[32]
Robust prediction of response to immune checkpoint blockade therapy in metastatic melanoma
Auslander N, Zhang G, Lee JS, Frederick DT, Miao B, Moll T, et al. Robust prediction of response to immune checkpoint blockade therapy in metastatic melanoma. Nat Med 2018;24:1545–9. https://doi.org/10.1038/s41591-018-0157-9
-
[33]
Single-cell RNA sequencing to explore immune cell heterogeneity
Papalexi E, Satija R. Single-cell RNA sequencing to explore immune cell heterogeneity. Nat Rev Immunol 2018;18:35–45. https://doi.org/10.1038/nri.2017.76
-
[34]
Tietscher S, Wagner J, Anzeneder T, Langwieder C, Rees M, Sobottka B, et al. A comprehensive single-cell map of T cell exhaustion-associated immune environments in human breast cancer. Nat Commun 2023;14:98. https://doi.org/10.1038/s41467-022- 35238-w
-
[35]
Huang L, Lou N, Xie T, Tang L, Han X, Shi Y. Identification of an antigen-presenting cells/T/NK cells-related gene signature to predict prognosis and CTSL to predict immunotherapeutic response for lung adenocarcinoma: an integrated analysis of bulk and single-cell RNA sequencing. Cancer Immunol Immunother CII 2023;72:3259–77. https://doi.org/10.1007/s0026...
-
[36]
Wu J, Wang Y, Huang Z, Wu J, Sun H, Zhou R, et al. Tumor microenvironment assessment-based signatures for predicting response to immunotherapy in non-small cell lung cancer. iScience 2024;27:111340. https://doi.org/10.1016/j.isci.2024.111340
-
[37]
Dinstag G, Shulman ED, Elis E, Ben-Zvi DS, Tirosh O, Maimon E, et al. Clinically oriented prediction of patient response to targeted and immunotherapies from the tumor transcriptome. Med 2023;4:15-30.e8. https://doi.org/10.1016/j.medj.2022.11.001
-
[38]
Ensenyat-Mendez M, Orozco JIJ, Llinàs-Arias P, Íñiguez-Muñoz S, Baker JL, Salomon MP, et al. Construction and validation of a gene expression classifier to predict immunotherapy response in primary triple-negative breast cancer. Commun Med 2023;3:93. https://doi.org/10.1038/s43856-023-00311-y
-
[39]
scCURE identifies cell types responding to immunotherapy and enables outcome prediction
Zou X, Liu Y, Wang M, Zou J, Shi Y, Su X, et al. scCURE identifies cell types responding to immunotherapy and enables outcome prediction. Cell Rep Methods 2023;3:100643. https://doi.org/10.1016/j.crmeth.2023.100643
-
[40]
The Cancer Genome Atlas Pan-Cancer analysis project
Weinstein JN, Collisson EA, Mills GB, Shaw KRM, Ozenberger BA, Ellrott K, et al. The Cancer Genome Atlas Pan-Cancer analysis project. Nat Genet 2013;45:1113–20. https://doi.org/10.1038/ng.2764
-
[41]
A Cancer Cell Program Promotes T Cell Exclusion and Resistance to Checkpoint Blockade
Jerby-Arnon L, Shah P, Cuoco MS, Rodman C, Su M-J, Melms JC, et al. A Cancer Cell Program Promotes T Cell Exclusion and Resistance to Checkpoint Blockade. Cell 2018;175:984-997.e24. https://doi.org/10.1016/j.cell.2018.09.006
-
[42]
A TCF4-dependent gene regulatory network confers resistance to immunotherapy in melanoma
Pozniak J, Pedri D, Landeloos E, Herck YV, Antoranz A, Vanwynsberghe L, et al. A TCF4-dependent gene regulatory network confers resistance to immunotherapy in melanoma. Cell 2024;187:166-183.e25. https://doi.org/10.1016/j.cell.2023.11.037
-
[43]
Defining T cell states associated with response to checkpoint immunotherapy in melanoma
Sade-Feldman M, Yizhak K, Bjorgaard SL, Ray JP, de Boer CG, Jenkins RW, et al. Defining T cell states associated with response to checkpoint immunotherapy in melanoma. Cell 2018;175:998-1013.e20. https://doi.org/10.1016/j.cell.2018.10.038
-
[44]
Ahmed YB, Al-Bzour AN, Ababneh OE, Abushukair HM, Saeed A. Genomic and Transcriptomic Predictors of Response to Immune Checkpoint Inhibitors in Melanoma Patients: A Machine Learning Approach. Cancers 2022;14:5605. https://doi.org/10.3390/cancers14225605
-
[45]
Cho J-W, Hong MH, Ha S-J, Kim Y-J, Cho BC, Lee I, et al. Genome-wide identification of differentially methylated promoters and enhancers associated with 40 response to anti-PD-1 therapy in non-small cell lung cancer. Exp Mol Med 2020;52:1550–63. https://doi.org/10.1038/s12276-020-00493-8
-
[46]
Ribas A, Algazi A, Ascierto PA, Butler MO, Chandra S, Gordon M, et al. PD-L1 blockade in combination with inhibition of MAPK oncogenic signaling in patients with advanced melanoma. Nat Commun 2020;11:6262. https://doi.org/10.1038/s41467-020- 19810-w
-
[47]
Poddubskaya E, Suntsova M, Lyadova M, Luppov D, Guryanova A, Lyadov V, et al. Biomarkers of success of anti-PD-(L)1 immunotherapy for non-small cell lung cancer derived from RNA- and whole-exome sequencing: results of a prospective observational study on a cohort of 85 patients. Front Immunol 2024;15. https://doi.org/10.3389/fimmu.2024.1493877
-
[48]
Gondal MN, Cieslik M, Chinnaiyan AM. Integrated cancer cell-specific single-cell RNA-seq datasets of immune checkpoint blockade-treated patients. Sci Data 2025;12:139. https://doi.org/10.1038/s41597-025-04381-6
-
[49]
Franken A, Bila M, Mechels A, Kint S, Dessel JV, Pomella V, et al. CD4+ T cell activation distinguishes response to anti-PD-L1+anti-CTLA4 therapy from anti-PD-L1 monotherapy. Immunity 2024;57:541-558.e7. https://doi.org/10.1016/j.immuni.2024.02.007
-
[50]
Luoma AM, Suo S, Wang Y, Gunasti L, Porter CBM, Nabilsi N, et al. Tissue-resident memory and circulating T cells are early responders to pre-surgical cancer immunotherapy. Cell 2022;185:2918-2935.e29. https://doi.org/10.1016/j.cell.2022.06.018
-
[51]
Reinstein ZZ, Zhang Y, Ospina OE, Nichols MD, Chu VA, de Mingo Pulido A, et al. Pre-existing skin-resident CD8 and γδ T cell circuits mediate immune response in Merkel cell carcinoma and predict immunotherapy efficacy. Cancer Discov 2024;14:1631–52. https://doi.org/10.1158/2159-8290.CD-23-0798
-
[52]
et al.: BEHRT: Transformer for electronic health records
Hołyńska-Iwan I, Szewczyk-Golec K. Analysis of changes in sodium and chloride ion transport in the skin. Sci Rep 2020;10:18094. https://doi.org/10.1038/s41598-020- 75275-3
-
[53]
Despin-Guitard E, Quenec’Hdu R, Nahaboo W, Schwarz N, Leube RE, Chazaud C, et al. Regionally specific levels and patterns of keratin 8 expression in the mouse embryo visceral endoderm emerge upon anterior-posterior axis determination. Front Cell Dev Biol 2022;10. https://doi.org/10.3389/fcell.2022.1037041
-
[54]
NCBI RefSeq: reference sequence standards through 25 years of curation and annotation
Goldfarb T, Kodali VK, Pujar S, Brover V, Robbertse B, Farrell CM, et al. NCBI RefSeq: reference sequence standards through 25 years of curation and annotation. Nucleic Acids Res 2024;53:D243. https://doi.org/10.1093/nar/gkae1038
-
[55]
Wu X, Wang X, Zhao Y, Li K, Yu B, Zhang J. Granzyme family acts as a predict biomarker in cutaneous melanoma and indicates more benefit from anti-PD-1 immunotherapy. Int J Med Sci 2021;18:1657–69. https://doi.org/10.7150/ijms.54747
-
[56]
Nonsense-mediated mRNA decay inhibits TRAF6-dependent anti-tumor immunity in colorectal cancer
Wang Y, Wang Z, Wang C, Wu Y, Li J, Feng L, et al. Nonsense-mediated mRNA decay inhibits TRAF6-dependent anti-tumor immunity in colorectal cancer. Cell Rep Med 2025;6:102463. https://doi.org/10.1016/j.xcrm.2025.102463
-
[57]
Tertiary lymphoid structures and B cells: An intratumoral immunity cycle
Fridman WH, Meylan M, Pupier G, Calvez A, Hernandez I, Sautès-Fridman C. Tertiary lymphoid structures and B cells: An intratumoral immunity cycle. Immunity 2023;56:2254–69. https://doi.org/10.1016/j.immuni.2023.08.009
-
[58]
Shi C, Wang Y, Xue J, Zhou X. Immunotherapy for EGFR-mutant advanced non-small- cell lung cancer: Current status, possible mechanisms and application prospects. Front Immunol 2022;13:940288. https://doi.org/10.3389/fimmu.2022.940288. 41
-
[59]
Hu M, Zheng K, Zhang L, Kan Y, Zhao J, Chen D. Therapeutic Strategies Targeting Aerobic Glycolysis in Cancer and Dynamic Monitoring of Associated Metabolites. Cells 2025;14:1288. https://doi.org/10.3390/cells14161288
-
[60]
Targeting collagen to optimize cancer immunotherapy
Wang Y, Zhang F, Qian Z, Jiang Y, Wu D, Liu L, et al. Targeting collagen to optimize cancer immunotherapy. Exp Hematol Oncol 2025;14:101. https://doi.org/10.1186/s40164-025-00691-y
-
[61]
Cancer Immunity: Lessons From Infectious Diseases
Trinchieri G. Cancer Immunity: Lessons From Infectious Diseases. J Infect Dis 2015;212:S67–73. https://doi.org/10.1093/infdis/jiv070
-
[62]
Metabolic mechanisms of immunotherapy resistance
Cabezón-Gutiérrez L, Palka-Kotlowska M, Custodio-Cabello S, Chacón-Ovejero B, Pacheco-Barcia V. Metabolic mechanisms of immunotherapy resistance. Explor Target Anti-Tumor Ther 2025;6:1002297. https://doi.org/10.37349/etat.2025.1002297
-
[63]
RhoGTPases and inflammasomes: Guardians of effector-triggered immunity
Dufies O, Boyer L. RhoGTPases and inflammasomes: Guardians of effector-triggered immunity. PLoS Pathog 2021;17:e1009504. https://doi.org/10.1371/journal.ppat.1009504
-
[64]
RNA Binding Proteins (RBPs) and their role in DNA Damage and radiation response in cancer
Mehta M, Raguraman R, Ramesh R, Munshi A. RNA Binding Proteins (RBPs) and their role in DNA Damage and radiation response in cancer. Adv Drug Deliv Rev 2022;191:114569. https://doi.org/10.1016/j.addr.2022.114569
-
[65]
Early signaling pathways in virus-infected cells
Bonhomme D, Poirier EZ. Early signaling pathways in virus-infected cells. Curr Opin Virol 2024;66:101411. https://doi.org/10.1016/j.coviro.2024.101411
-
[66]
Immunometabolism – The Role of Branched-Chain Amino Acids
Yahsi B, Gunaydin G. Immunometabolism – The Role of Branched-Chain Amino Acids. Front Immunol 2022;13:886822. https://doi.org/10.3389/fimmu.2022.886822
-
[67]
Karin N. Chemokines in the Landscape of Cancer Immunotherapy: How They and Their Receptors Can Be Used to Turn Cold Tumors into Hot Ones? Cancers 2021;13:6317. https://doi.org/10.3390/cancers13246317
-
[68]
Deciphering the deterministic role of TCR signaling in T cell fate determination
Qin Z, Xu T. Deciphering the deterministic role of TCR signaling in T cell fate determination. Front Immunol 2025;16:1562248. https://doi.org/10.3389/fimmu.2025.1562248
-
[69]
Antigen presentation and tumor immunogenicity in cancer immunotherapy response prediction
Wang S, He Z, Wang X, Li H, Liu X-S. Antigen presentation and tumor immunogenicity in cancer immunotherapy response prediction. eLife n.d.;8:e49020. https://doi.org/10.7554/eLife.49020
-
[70]
NCBI GEO: archive for functional genomics data sets—update
Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, et al. NCBI GEO: archive for functional genomics data sets—update. Nucleic Acids Res 2013;41:D991–5. https://doi.org/10.1093/nar/gks1193
-
[71]
Ajay Nadig, Joseph M Replogle, Angela N Pogson, et al
Mudge JM, Carbonell-Sala S, Diekhans M, Martinez JG, Hunt T, Jungreis I, et al. GENCODE 2025: reference gene annotation for human and mouse. Nucleic Acids Res 2025;53:D966–75. https://doi.org/10.1093/nar/gkae1078
-
[72]
New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1)
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228–47. https://doi.org/10.1016/j.ejca.2008.10.026
-
[73]
Neoadjuvant immune checkpoint blockade in high-risk resectable melanoma
Amaria RN, Reddy SM, Tawbi HA, Davies MA, Ross MI, Glitza IC, et al. Neoadjuvant immune checkpoint blockade in high-risk resectable melanoma. Nat Med 2018;24:1649–54. https://doi.org/10.1038/s41591-018-0197-1
-
[74]
Enrichr: a comprehensive gene set enrichment analysis web server 2016 update
Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res 2016;44:W90–7. https://doi.org/10.1093/nar/gkw377
-
[75]
WikiPathways 2024: next generation pathway database
Agrawal A, Balcı H, Hanspers K, Coort SL, Martens M, Slenter DN, et al. WikiPathways 2024: next generation pathway database. Nucleic Acids Res 2024;52:D679–89. https://doi.org/10.1093/nar/gkad960
-
[76]
KEGG: Kyoto Encyclopedia of Genes and Genomes
Kanehisa M, Goto S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res 2000;28:27–30. https://doi.org/10.1093/nar/28.1.27. 42
-
[77]
The Reactome Pathway Knowledgebase 2024
Milacic M, Beavers D, Conley P, Gong C, Gillespie M, Griss J, et al. The Reactome Pathway Knowledgebase 2024. Nucleic Acids Res 2024;52:D672–8. https://doi.org/10.1093/nar/gkad1025
-
[78]
The Molecular Signatures Database (MSigDB) hallmark gene set collection
Liberzon A, Birger C, Thorvaldsdóttir H, Ghandi M, Mesirov JP, Tamayo P. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst 2015;1:417–25. https://doi.org/10.1016/j.cels.2015.12.004
-
[79]
Enrichment Map: A Network- Based Method for Gene-Set Enrichment Visualization and Interpretation
Merico D, Isserlin R, Stueker O, Emili A, Bader GD. Enrichment Map: A Network- Based Method for Gene-Set Enrichment Visualization and Interpretation. PLOS ONE 2010;5:e13984. https://doi.org/10.1371/journal.pone.0013984. 43 Supplementary Materials Supplementary Table S1. Model inclusion-exclusion criteria. Models were included if they were published betwee...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.