Recognition: 2 theorem links
· Lean TheoremPlatelet plug microstructure and flow modulate fibrin gelation dynamics: Insights from computational simulations
Pith reviewed 2026-05-10 18:14 UTC · model grok-4.3
The pith
Denser platelet plugs accelerate fibrin gelation at their edges but hinder it in the core under flow.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Platelet plug density and flow together control fibrin gelation: higher density speeds overall initiation and raises thrombin levels between platelets but restricts transport so gelation occurs first at the periphery; lower density slows initiation due to less surface area yet permits fibrinogen replenishment deeper in, allowing gelation to start at the vessel wall and spread inward.
What carries the argument
The 2D computational framework that couples a discrete pre-adhered platelet aggregate, a reduced coagulation model generating thrombin on platelet surfaces, and a fibrin polymerization model, simulated at various wall shear rates.
If this is right
- Increasing plug density accelerates gelation initiation but localizes it to the plug periphery.
- Loose platelet configurations support interior gelation through better fibrinogen transport despite slower start.
- Dense plugs show elevated thrombin concentrations between platelets due to restricted outflow.
- Flow conditions modulate the extent of these transport limitations and clotting patterns.
Where Pith is reading between the lines
- If early densification limits interior fibrin, clots may be more susceptible to disruption before full stabilization.
- The tradeoff could inform why certain flow regimes or platelet defects lead to unstable thrombi.
- Extending the model to include platelet contraction dynamics over time would test the proposed sealing-stabilization balance.
- These insights might guide designs for interventions that optimize both rapid sealing and durable fibrin reinforcement.
Load-bearing premise
That the reduced coagulation model correctly predicts thrombin generation rates on platelet surfaces across flow conditions and that the two-dimensional geometry adequately represents transport in actual three-dimensional platelet plugs.
What would settle it
Experimental imaging of fibrin distribution within platelet aggregates of varying densities exposed to controlled shear rates, which would show whether dense plugs indeed exhibit peripheral-only fibrin while loose plugs form fibrin throughout.
Figures







read the original abstract
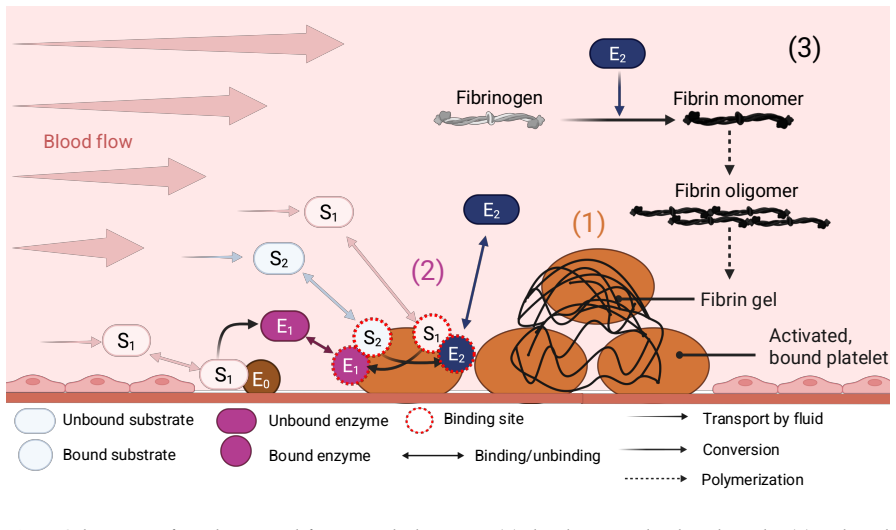
During the formation of a thrombus, the architecture of the growing platelet aggregate is heterogeneous, with areas of dense and loosely packed platelets. The surface of activated platelets facilitate biochemical coagulation reactions that ultimately result in the formation of a fibrin network which stabilizes the thrombus. How platelet-plug microstructure and flow jointly govern the onset and development of fibrin is incompletely understood. We developed a novel 2D computational framework that integrates (1) a pre-adhered, discrete platelet aggregate, (2) a reduced coagulation model that generates thrombin, and (3) a fibrin polymerization model. Three platelet-plug configurations were constructed with prescribed interplatelet gaps and simulations were performed with various wall shear rates. We quantified spatiotemporal clotting metrics, including coagulation factor concentrations, fibrin evolution, and gelation onset. Across geometries, gelation initiation accelerated with increasing plug density. For more dense geometries, gelation emerged first near the plug periphery. As the platelet density increased, intraplug transport was increasingly restricted and the thrombin concentrations in between platelets increased. In contrast, the loose plug supported fibrinogen replenishment deeper into the plug core. Despite slower coagulation initiation due to reduced platelet surface area, monomer generation persisted in the interior, causing gelation to begin at the vessel wall. These results suggest a mechanistic tradeoff: rapid sealing of the injured vessel wall by early platelet contraction, i.e. plug densification, may impede the intraplug fibrin formation needed for durable stabilization. The proposed model provides a basis for studies of platelet-coagulation interactions under flow, including therapeutic developments relevant to prevention of cardiovascular disease.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper develops a novel 2D computational framework integrating a pre-adhered discrete platelet aggregate, a reduced coagulation model for thrombin generation, and a fibrin polymerization model. Simulations of three platelet-plug configurations with prescribed interplatelet gaps at varying wall shear rates quantify spatiotemporal metrics including factor concentrations, fibrin evolution, and gelation onset. Key findings are that gelation accelerates with increasing plug density, emerges first at the periphery in dense geometries due to restricted intraplug transport, and begins at the vessel wall in loose geometries due to better fibrinogen replenishment; this supports a mechanistic tradeoff in which early platelet contraction and densification may impede durable intraplug fibrin stabilization.
Significance. If the model outputs prove robust, the work provides mechanistic insights into how platelet microstructure and flow jointly control fibrin gelation, with potential relevance to therapeutic targeting of thrombus stability in cardiovascular disease. The integrated simulation framework is a strength, enabling exploration of coupled platelet-coagulation dynamics under flow that are challenging to measure directly.
major comments (2)
- [Abstract and Methods (Reduced Coagulation Model)] The central tradeoff claim rests on the spatiotemporal thrombin and fibrin fields generated by the reduced coagulation model (described in the abstract and Methods). This model collapses the cascade to a small set of surface reactions whose rates are prescribed rather than re-derived or benchmarked against measured thrombin generation under the specific shear rates and platelet densities of the simulations; any quantitative mismatch would directly alter the reported gelation onset locations and the mechanistic interpretation.
- [Methods (Geometry and Transport)] The 2D geometry is used to represent transport within the platelet plug, but real plugs are 3D porous structures. This restricts diffusion and advection pathways relative to reality and is load-bearing for the claim that dense plugs impede intraplug fibrin while loose plugs permit core gelation, as the predicted thrombin/fibrinogen distributions would change in 3D.
minor comments (1)
- [Abstract] The abstract states that spatiotemporal clotting metrics were quantified but does not report specific numerical values, time courses, or sensitivity to parameter choices; adding these (e.g., in a new table or figure) would improve clarity without altering the central claim.
Simulated Author's Rebuttal
We thank the referee for their constructive and insightful comments on our manuscript. Below, we provide point-by-point responses to the major comments and describe the revisions made to address them.
read point-by-point responses
-
Referee: [Abstract and Methods (Reduced Coagulation Model)] The central tradeoff claim rests on the spatiotemporal thrombin and fibrin fields generated by the reduced coagulation model (described in the abstract and Methods). This model collapses the cascade to a small set of surface reactions whose rates are prescribed rather than re-derived or benchmarked against measured thrombin generation under the specific shear rates and platelet densities of the simulations; any quantitative mismatch would directly alter the reported gelation onset locations and the mechanistic interpretation.
Authors: We agree that the reduced coagulation model relies on prescribed rate constants drawn from the literature rather than being newly derived or experimentally benchmarked for the precise shear rates and platelet densities in our simulations. The model incorporates established surface-mediated reactions for thrombin generation on activated platelets, as described in the Methods. To address the concern that quantitative mismatches could affect the reported gelation locations, we have added a parameter sensitivity analysis in the revised manuscript. This analysis varies the key reaction rates over physiologically relevant ranges and confirms that the qualitative trends—faster gelation with increasing plug density, peripheral onset in dense plugs, and wall-initiated onset in loose plugs—persist. These results support the mechanistic tradeoff interpretation at the level of qualitative dynamics, which is the primary focus of the work. revision: partial
-
Referee: [Methods (Geometry and Transport)] The 2D geometry is used to represent transport within the platelet plug, but real plugs are 3D porous structures. This restricts diffusion and advection pathways relative to reality and is load-bearing for the claim that dense plugs impede intraplug fibrin while loose plugs permit core gelation, as the predicted thrombin/fibrinogen distributions would change in 3D.
Authors: We acknowledge that the 2D geometry represents a simplification of the three-dimensional porous architecture of real platelet plugs, which could quantitatively alter diffusion and advection pathways. The 2D framework was selected to enable computationally feasible, high-resolution simulations that explicitly resolve discrete platelet positions, prescribed interplatelet gaps, and coupled flow-reaction dynamics. The core transport effects underlying our claims—restricted intraplug access in dense configurations versus enhanced fibrinogen replenishment in loose ones—are governed by local gap sizes and flow patterns, which are expected to produce qualitatively similar directional trends in 3D. We have expanded the Discussion to explicitly note this dimensionality limitation and to outline how the current 2D results can inform future three-dimensional extensions. revision: partial
Circularity Check
Forward simulations produce emergent patterns with no reduction to inputs by construction
full rationale
The paper constructs three prescribed 2D platelet geometries, integrates a reduced coagulation model and fibrin polymerization model with fixed parameters, and runs forward simulations under varying shear rates. All reported quantities (thrombin fields, fibrin evolution, gelation onset locations) are computed outputs of this integrated dynamical system rather than fitted values or quantities defined in terms of themselves. No step equates a prediction to its input by construction, renames a known result, or relies on a self-citation chain whose validity is presupposed; the mechanistic tradeoff is an observed consequence of the transport and reaction dynamics under the stated assumptions.
Axiom & Free-Parameter Ledger
free parameters (3)
- interplatelet gap sizes
- wall shear rates
- coagulation reaction rates
axioms (2)
- domain assumption The 2D computational domain adequately models the 3D flow and diffusion in platelet plugs.
- domain assumption The reduced coagulation model sufficiently represents thrombin generation without needing full biochemical detail.
Lean theorems connected to this paper
-
Cost/FunctionalEquationwashburn_uniqueness_aczel unclear?
unclearRelation between the paper passage and the cited Recognition theorem.
reduced coagulation model that generates thrombin... fibrin polymerization model
-
Foundation/RealityFromDistinctionreality_from_one_distinction unclear?
unclearRelation between the paper passage and the cited Recognition theorem.
Navier-Stokes... discrete platelet aggregate... gelation onset
What do these tags mean?
- matches
- The paper's claim is directly supported by a theorem in the formal canon.
- supports
- The theorem supports part of the paper's argument, but the paper may add assumptions or extra steps.
- extends
- The paper goes beyond the formal theorem; the theorem is a base layer rather than the whole result.
- uses
- The paper appears to rely on the theorem as machinery.
- contradicts
- The paper's claim conflicts with a theorem or certificate in the canon.
- unclear
- Pith found a possible connection, but the passage is too broad, indirect, or ambiguous to say the theorem truly supports the claim.
Reference graph
Works this paper leans on
-
[1]
Mechanisms of thrombus formation
Furie B, Furie BC. Mechanisms of thrombus formation. New England Journal of Medicine. 2008;359(9):938-49. doi:10.1056/NEJMra0801082
-
[2]
A cell-based model of hemostasis
Hoffman M, Monroe III DM. A cell-based model of hemostasis. Thrombosis and Haemostasis. 2001;85(06):958-65. doi:10.1055/s-0037-1615947
-
[3]
Fluid mechanics of blood clot formation
Fogelson AL, Neeves KB. Fluid mechanics of blood clot formation. Annual Review of Fluid Mechanics. 2015;47(1):377-403. doi:10.1146/annurev-fluid-010814-014513
-
[4]
Stalker TJ, Traxler EA, Wu J, Wannemacher KM, Cermignano SL, Voronov R, et al. Hierarchical organization in the hemostatic response and its relationship to the platelet-signaling network. Blood. 2013;121(10):1875-85. doi:10.1182/blood-2012-09-457739
-
[5]
A systems approach to hemostasis: 1
Welsh JD, Stalker TJ, Voronov R, Muthard RW, Tomaiuolo M, Diamond SL, et al. A systems approach to hemostasis: 1. The interdependence of thrombus architecture and agonist movements in the gaps between platelets. Blood. 2014;124(11):1808-15. doi:10.1182/blood-2014-01-550335
-
[6]
A systems approach to hemostasis: 4
Welsh JD, Muthard RW, Stalker TJ, Taliaferro JP , Diamond SL, Brass LF. A systems approach to hemostasis: 4. How hemostatic thrombi limit the loss of plasma-borne molecules from the microvasculature. Blood, The Journal of the American Society of Hematology. 2016;127(12):1598-605. doi:10.1182/blood-2015-09-672188
-
[7]
A systems approach to hemostasis: 3
Stalker TJ, Welsh JD, Tomaiuolo M, Wu J, Colace TV , Diamond SL, et al. A systems approach to hemostasis: 3. Thrombus consolidation regulates intrathrombus solute transport and local thrombin activity. Blood. 2014;124(11):1824-31. doi:10.1182/blood-2014-01-550319
-
[8]
Role of the extrinsic pathway of blood coagulation in hemostasis and thrombosis
Mackman N, Tilley RE, Key NS. Role of the extrinsic pathway of blood coagulation in hemostasis and thrombosis. Arteriosclerosis, Thrombosis, and Vascular biology. 2007;27(8):1687-93. doi:10.1161/ATVBAHA.107.141911
-
[9]
Platelet control of fibrin distribution and microelasticity in thrombus formation under flow
Swieringa F, Baaten CC, Verdoold R, Mastenbroek TG, Rijnveld N, Van Der Laan KO, et al. Platelet control of fibrin distribution and microelasticity in thrombus formation under flow. Arteriosclerosis, thrombosis, and vascular biology. 2016;36(4):692-9. doi:10.1161/ATVBAHA.115.306537
-
[10]
Falati S, Gross P , Merrill-Skoloff G, Furie BC, Furie B. Real-time in vivo imaging of platelets, tissue factor and fibrin during arterial thrombus formation in the mouse. Nature Medicine. 2002 Oct;8(10):1175-80. doi:10.1038/nm782
-
[11]
Kuharsky AL, Fogelson AL. Surface-mediated control of blood coagulation: the role of binding site densities and platelet deposition. Biophysical journal. 2001;80(3):1050-74. doi:10.1016/S0006-3495(01)76085-7. April 10, 2026 50/57
-
[12]
Tissue factor activity under flow
Diamond SL. Tissue factor activity under flow. Thrombosis Research. 2010;125:S29-30. doi:10.1016/j.thromres.2010.01.029
-
[13]
Two-photon intravital imaging of thrombus development
Kamocka MM, Mu J, Liu X, Chen N, Zollman A, Sturonas-Brown B, et al. Two-photon intravital imaging of thrombus development. Journal of Biomedical Optics. 2010;15(1):016020-0. doi:10.1117/1.3322676
-
[14]
Multiscale systems biology and physics of thrombosis under flow
Flamm MH, Diamond S. Multiscale systems biology and physics of thrombosis under flow. Annals of Biomedical Engineering. 2012;40(11):2355-64. doi:10.1007/s10439-012-0557-9
-
[15]
Systems biology of coagulation
Diamond SL. Systems biology of coagulation. Journal of Thrombosis and Haemostasis. 2013;11:224-32. doi:10.1111/jth.12220
-
[16]
An overview of mathematical modeling of thrombus formation under flow
Leiderman K, Fogelson A. An overview of mathematical modeling of thrombus formation under flow. Thrombosis Research. 2014;133:S12-4. doi:10.1016/j.thromres.2014.03.005
-
[17]
Recent advances in computational modeling of fibrin clot formation: A review
Yesudasan S, Averett RD. Recent advances in computational modeling of fibrin clot formation: A review. Computational Biology and Chemistry. 2019;83:107148. doi:10.1016/j.compbiolchem.2019.107148
-
[18]
Mathematical models of fibrin polymerization: past, present, and future
Nelson AC, Kelley MA, Haynes LM, Leiderman K. Mathematical models of fibrin polymerization: past, present, and future. Current Opinion in Biomedical Engineering. 2021;20:100350. doi:https://doi.org/10.1016/j.cobme.2021.100350
-
[19]
Mathematical models of coagulation—are we there yet? Journal of Thrombosis and Haemostasis
Owen MJ, Wright JR, Tuddenham EG, King JR, Goodall AH, Dunster JL. Mathematical models of coagulation—are we there yet? Journal of Thrombosis and Haemostasis. 2024;22(6):1689-703. doi:10.1016/j.jtha.2024.03.009
-
[20]
Membrane Binding-site Density Can Modulate Activation Thresholds in Enzyme Systems
Fogelson AL, Kuharsky AL. Membrane Binding-site Density Can Modulate Activation Thresholds in Enzyme Systems. Journal of Theoretical Biology. 1998;193(1):1-18. doi:https://doi.org/10.1006/jtbi.1998.0670
-
[21]
Blood Clot Formation under Flow: The Importance of Factor XI Depends Strongly on Platelet Count
Fogelson AL, Hussain YH, Leiderman K. Blood Clot Formation under Flow: The Importance of Factor XI Depends Strongly on Platelet Count. Biophysical Journal. 2012;102(1):10-8. doi:https://doi.org/10.1016/j.bpj.2011.10.048
-
[22]
Grow with the flow: a spatial–temporal model of platelet deposition and blood coagulation under flow
Leiderman K, Fogelson AL. Grow with the flow: a spatial–temporal model of platelet deposition and blood coagulation under flow. Mathematical Medicine and Biology. 2011;28(1):47-84. doi:10.1093/imammb/dqq005
-
[23]
Inhibition of platelet-surface-bound proteins during coagulation under flow I: TFPI
Miyazawa K, Fogelson AL, Leiderman K. Inhibition of platelet-surface-bound proteins during coagulation under flow I: TFPI. Biophysical Journal. 2023;122(1):99-113. doi:10.1016/j.bpj.2022.11.023. April 10, 2026 51/57
-
[24]
Fogelson AL, Tania N. Coagulation under Flow: The Influence of Flow-Mediated Transport on the Initiation and Inhibition of Coagulation. Pathophysiology of Haemostasis and Thrombosis. 2006 01;34(2-3):91-108. doi:10.1159/000089930
-
[25]
A multiscale model of thrombus development
Xu Z, Chen N, Kamocka MM, Rosen ED, Alber M. A multiscale model of thrombus development. Journal of the Royal Society Interface. 2008;5(24):705-22. doi:10.1098/rsif.2007.1202
-
[26]
Modelling of platelet–fibrin clot formation in flow with a DPD–PDE method
Tosenberger A, Ataullakhanov F, Bessonov N, Panteleev M, Tokarev A, Volpert V . Modelling of platelet–fibrin clot formation in flow with a DPD–PDE method. Journal of Mathematical Biology. 2016;72(3):649-81. doi:10.1007/s00285-015-0891-2
-
[27]
Xu S, Xu Z, Kim OV , Litvinov RI, Weisel JW, Alber M. Model predictions of deformation, embolization and permeability of partially obstructive blood clots under variable shear flow. Journal of The Royal Society Interface. 2017;14(136):20170441. doi:10.1098/rsif.2017.0441
-
[28]
Fibrin networks regulate protein transport during thrombus development
Kim OV , Xu Z, Rosen ED, Alber MS. Fibrin networks regulate protein transport during thrombus development. PLOS Computational Biology. 2013;9(6):e1003095. doi:10.1371/journal.pcbi.1003095
-
[29]
House C, Huang Z, Shankar KN, Young SJ, Roberts ME, Diamond SL, et al. From imaging to computational domains for physics-driven molecular biology simulations: Hindered diffusion in platelet masses. PLOS Computational Biology. 2025;21(7):e1012853. doi:10.1371/journal.pcbi.1012853
-
[31]
A systems approach to hemostasis: 2
Tomaiuolo M, Stalker TJ, Welsh JD, Diamond SL, Sinno T, Brass LF. A systems approach to hemostasis: 2. Computational analysis of molecular transport in the thrombus microenvironment. Blood. 2014;124(11):1816-23. doi:10.1182/blood-2014-01-550343
-
[32]
Shankar KN, Zhang Y, Sinno T, Diamond SL. A three-dimensional multiscale model for the prediction of thrombus growth under flow with single-platelet resolution. PLOS Computational Biology. 2022;18(1):e1009850. doi:10.1371/journal.pcbi.1009850
-
[33]
Development of a parallel multiscale 3D model for thrombus growth under flow
Shankar KN, Diamond SL, Sinno T. Development of a parallel multiscale 3D model for thrombus growth under flow. Frontiers in Physics. 2023;Volume 11 - 2023. Available from:https: //www.frontiersin.org/journals/physics/articles/10.3389/fphy.2023.1256462. doi:10.3389/fphy.2023.1256462. April 10, 2026 52/57
-
[34]
clotFoam: An open-source framework to simulate blood clot formation under arterial flow
Montgomery D, Municchi F, Leiderman K. clotFoam: An open-source framework to simulate blood clot formation under arterial flow. SoftwareX. 2023;23:101483. doi:10.1016/j.softx.2023.101483
-
[35]
Toward an understanding of fibrin branching structure
Fogelson AL, Keener JP . Toward an understanding of fibrin branching structure. Physical Review E. 2010;81(5):051922. doi:10.1103/PhysRevE.81.051922
-
[36]
Development of fibrin branch structure before and after gelation
Fogelson AL, Nelson AC, Zapata-Allegro C, Keener JP . Development of fibrin branch structure before and after gelation. SIAM journal on applied mathematics. 2022;82(1):267-93. doi:10.1137/21m1401024
-
[37]
Clot permeability, agonist transport, and platelet binding kinetics in arterial thrombosis
Du J, Kim D, Alhawael G, Ku DN, Fogelson AL. Clot permeability, agonist transport, and platelet binding kinetics in arterial thrombosis. Biophysical journal. 2020;119(10):2102-15. doi:10.1016/j.bpj.2020.08.041
-
[38]
Blood Rheology and Hemodynamics
Baskurt OKM Herbert J. Blood Rheology and Hemodynamics. Seminars in Thrombosis and Hemostasis. 2003 Nov;29(05):435-50. doi:10.1055/s-2003-44551
-
[39]
Handbook of hemorheology and hemodynamics
Baskurt OK, Hardeman MR, Rampling MW. Handbook of hemorheology and hemodynamics. vol. 69. IOS press; 2007
2007
-
[40]
Structural origins of fibrin clot rheology
Ryan EA, Mockros LF, Weisel JW, Lorand L. Structural origins of fibrin clot rheology. Biophysical journal. 1999;77(5):2813-26. doi:10.1016/S0006-3495(99)77113-4
-
[41]
Hantgan RR, Hermans J. Assembly of fibrin. A light scattering study. Journal of Biological Chemistry. 1979;254(22):11272-81. doi:10.1016/S0021-9258(19)86481-5
-
[42]
Naski M, Shafer J. A kinetic model for the alpha-thrombin-catalyzed conversion of plasma levels of fibrinogen to fibrin in the presence of antithrombin III. Journal of Biological Chemistry. 1991;266(20):13003-10. doi:10.1016/S0021-9258(18)98795-8
-
[43]
Miyazawa K, Fogelson AL, Leiderman K. Inhibition of platelet-surface-bound proteins during coagulation under flow II: Antithrombin and heparin. Biophysical Journal. 2023;122(1):230-40. doi:https://doi.org/10.1016/j.bpj.2022.10.038
-
[44]
Olson S, Björk I, Sheffer R, Craig P , Shore J, Choay J. Role of the antithrombin-binding pentasaccharide in heparin acceleration of antithrombin-proteinase reactions. Resolution of the antithrombin conformational change contribution to heparin rate enhancement. Journal of Biological Chemistry. 1992;267(18):12528-38. doi:10.1016/S0021-9258(18)42309-5
-
[45]
Fibrin formation, structure and properties
Weisel JW, Litvinov RI. Fibrin formation, structure and properties. Fibrous proteins: structures and mechanisms. 2017:405-56
2017
-
[46]
Platelet–coagulant protein interaction
Walsh PN. Platelet–coagulant protein interaction. In: Colman R, Hirsh J, Marder V , Salzman E, editors. Hemostasis and Thrombosis: Basic Principles and Clinical Practice. 3rd ed. Philadelphia, PA: J.B. Lippincott Company; 1994. p. 629-51. April 10, 2026 53/57
1994
-
[47]
Platelet coagulation-protein interactions
Walsh PN. Platelet coagulation-protein interactions. Seminars in Thrombosis and Hemostasis. 2004;30(4):461-71. doi:10.1055/s-2004-833481
-
[48]
Prothrombin and thrombin
Mann KG. Prothrombin and thrombin. In: Colman R, Hirsh J, Marder V , Salzman E, editors. Hemostasis and Thrombosis: Basic Principles and Clinical Practice. 3rd ed. Philadelphia, PA: J.B. Lippincott Company; 1994. p. 184-99
1994
-
[49]
Towards understanding the effect of fibrinogen interactions on fibrin gel structure
Nelson AC, Fogelson AL. Towards understanding the effect of fibrinogen interactions on fibrin gel structure. Physical Review E. 2023;107(2):024413. doi:10.1103/PhysRevE.107.024413
-
[50]
Contact pathway function during human whole blood clotting on procoagulant surfaces
Zhu S, Herbig BA, Yu X, Chen J, Diamond SL. Contact pathway function during human whole blood clotting on procoagulant surfaces. Frontiers in Medicine. 2018;5:209. doi:10.3389/fmed.2018.00209
-
[51]
Chen J, Diamond SL. Reduced model to predict thrombin and fibrin during thrombosis on collagen/tissue factor under venous flow: Roles of γ’-fibrin and factor XIa. PLoS Computational Biology. 2019;15(8):e1007266. doi:10.1371/journal.pcbi.1007266e
-
[52]
Blood clot contraction: mechanisms, pathophysiology, and disease
Litvinov RI, Weisel JW. Blood clot contraction: mechanisms, pathophysiology, and disease. Research and Practice in Thrombosis and Haemostasis. 2023;7(1):100023. doi:10.1016/j.rpth.2022.100023
-
[53]
Platelet packing density is an independent regulator of the hemostatic response to injury
Mirramezani M, Herbig BA, Stalker TJ, Nettey L, Cooper M, Weisel JW, et al. Platelet packing density is an independent regulator of the hemostatic response to injury. Journal of Thrombosis and Haemostasis. 2018;16(5):973-83. doi:10.1111/jth.13986
-
[54]
Thrombin generation and fibrin formation under flow on biomimetic tissue factor-rich surfaces
Onasoga-Jarvis A, Puls T, O’Brien S, Kuang L, Liang H, Neeves K. Thrombin generation and fibrin formation under flow on biomimetic tissue factor-rich surfaces. Journal of Thrombosis and Haemostasis. 2014;12(3):373-82. doi:10.1111/jth.12491
-
[55]
Ballard-Kordeliski A, Lee RH, O’Shaughnessy EC, Kim PY, Jones SR, Pawlinski R, et al. 4D intravital imaging studies identify platelets as the predominant cellular procoagulant surface in a mouse hemostasis model. Blood. 2024;144(10):1116-26. doi:10.1182/blood.2023022608
-
[56]
Contact activation of blood coagulation on a defined kaolin/collagen surface in a microfluidic assay
Zhu S, Diamond SL. Contact activation of blood coagulation on a defined kaolin/collagen surface in a microfluidic assay. Thrombosis Research. 2014;134(6):1335-43. doi:10.1016/j.thromres.2014.09.030
-
[57]
Campbell RA, Overmyer KA, Bagnell CR, Wolberg AS. Cellular procoagulant activity dictates clot structure and stability as a function of distance from the cell surface. Arteriosclerosis, Thrombosis, and Vascular Biology. 2008;28(12):2247-54. doi:10.1161/ATVBAHA.108.176008. April 10, 2026 54/57
-
[58]
Campbell RA, Overmyer KA, Selzman CH, Sheridan BC, Wolberg AS. Contributions of extravascular and intravascular cells to fibrin network formation, structure, and stability. Blood. 2009;114(23):4886-96. doi:10.1182/blood-2009-06-228940
-
[59]
Mechanisms of thrombin inhibition by protein S and the TFPIα-fVshort-protein S complex; 2025
Ginsberg AG, Ahnström J, Crawley JTB, Leiderman K, Monroe DM, Neeves KB, et al.. Mechanisms of thrombin inhibition by protein S and the TFPIα-fVshort-protein S complex; 2025. arXiv:arXiv:2512.11495
-
[60]
Synergy between tissue factor and exogenous factor XIa in initiating coagulation
Leiderman K, Chang WC, Ovanesov M, Fogelson AL. Synergy between tissue factor and exogenous factor XIa in initiating coagulation. Arteriosclerosis, Thrombosis, and Vascular Biology. 2016;36(12):2334-45. doi:10.1161/ATVBAHA.116.308186
-
[61]
Ziff RM, Stell G. Kinetics of polymer gelation. The Journal of Chemical Physics. 1980;73(7):3492-9. doi:10.1063/1.440502
-
[62]
Glycoprotein VI interplay with fibrin (ogen) in thrombosis
Mangin PH, Gardiner EE, Ariëns RA, Jandrot-Perrus M. Glycoprotein VI interplay with fibrin (ogen) in thrombosis. Journal of Thrombosis and Haemostasis. 2023;21(7):1703-13. doi:10.1016/j.jtha.2023.03.022
-
[63]
Okorie UM, Denney WS, Chatterjee MS, Neeves KB, Diamond SL. Determination of surface tissue factor thresholds that trigger coagulation at venous and arterial shear rates: amplification of 100 fM circulating tissue factor requires flow. Blood. 2008;111(7):3507-13. doi:10.1182/blood-2007-08-106229
-
[64]
Perspective: A Method for Uniform Reporting of Grid Refinement Studies
Roache PJ. Perspective: A Method for Uniform Reporting of Grid Refinement Studies. Journal of Fluids Engineering. 1994 Sep;116(3):405-13. doi:10.1115/1.2910291. April 10, 2026 55/57 T able 1.Parameters and chemical species (with their corresponding variables) in the discrete platelet model, the reduced model of coagulation, and the fibrin polymerization m...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.