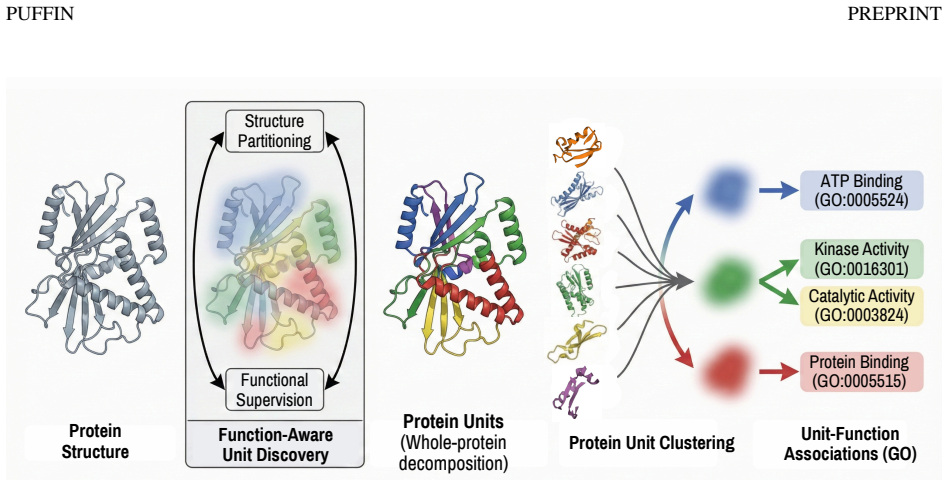
A putative, computationally stable structure of homotrimeric BP180/collagen XVII
Molecular dynamics shows the AI-predicted structure of the skin adhesion protein remains mostly stable over 500 ns with domain-specific flex
abstract
click to expand
Background: BP180, also known as collagen XVII and BPAG2 (bullous pemphigoid antigen 2), is a 180-kDa transmembrane protein within the hemidesmosomal plaque complex, and which is known to be a major antigen in bullous pemphigoid, gestational pemphigoid, cicatricial (mucous membrane) pemphigoid, and linear IgA bullous disease.
Objective: At present, the 3D structure of BP180 is not known. The goal is to predict a reasonable structure for BP180 through machine learning and molecular dynamics.
Methods: In this work, we use the recent Boltz-2 model to predict a putative structure for the intracellular, transmembrane, and proximal extracellular domains, including the NC16A antigenic region and a portion of its first extracellular collagenous domain, Col-15. We computationally embed BP180 in a simple phospholipid bilayer, demonstrate that the putative structure is stable using molecular dynamics, and analyze its allosteric properties.
Results: The structures presented satisfy symmetry and secondary structure properties which are expected from homology modelling. Over three 500 ns trajectories, there is minor instability of the predicted globular head domain, but the homotrimer otherwise stays mostly folded. The putative NC16A domain is stiff, whereas the truncated Col-15 domain is highly flexible. There does not appear to be a nearby stable conformation distinct from the initial state.
Conclusion: The structure presented is a useful starting point for targeting BP180 pharmacologically, for further experimental characterization of BP180, and for generating hypotheses regarding the relevant epitopes contributing to bullous disease. Diffusion models such as Boltz-2 and AlphaFold3 are useful, but their results must be evaluated carefully.
 full image
full image