Recognition: unknown
Unity and Diversity of Intracellular pH Maintenance Mechanisms
Pith reviewed 2026-05-10 09:18 UTC · model grok-4.3
The pith
A thermodynamic lower bound on the power cells must expend to hold ionic gradients equals exactly the rate of free-energy loss through passive ion leakage.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
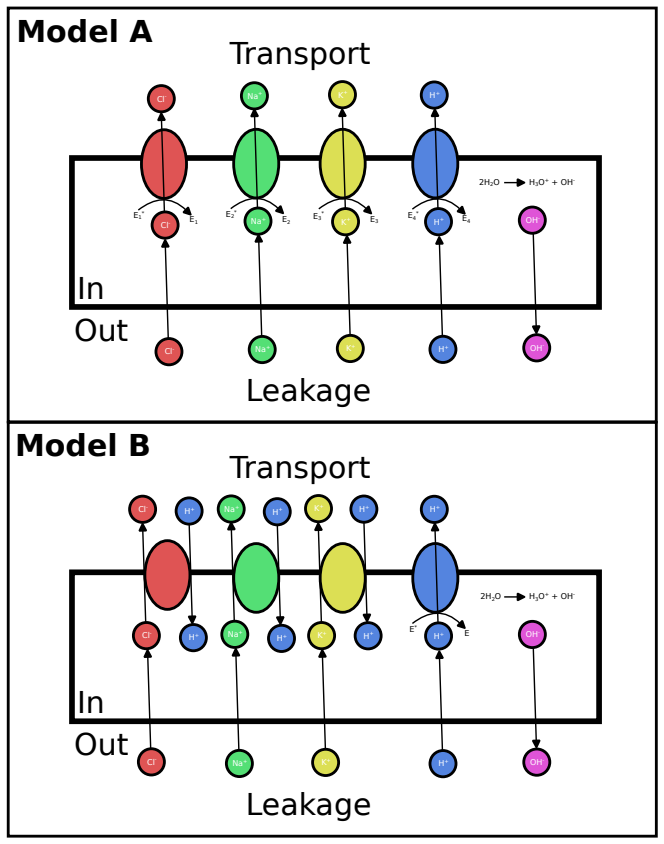
The minimum power required to sustain ionic motive forces at steady state equals the free-energy dissipation rate due to passive ion leakage. This lower bound is independent of organism, energy source, and transporter architecture within a broad class of electrophysiological models. Cost minimization under the bound yields the universal cytoplasmic ion asymmetry from permeability differences alone, while environmental extremes raise the bound and imperfect efficiency plus variability select for multiple transporter architectures each optimal in its own regime.
What carries the argument
The model-independent thermodynamic equality between steady-state maintenance power and ion-leakage dissipation rate.
If this is right
- Asymmetric membrane permeabilities alone suffice to produce the K+-rich, Na+-poor cytoplasm observed across all taxa.
- Extremophiles incur higher maintenance costs under extreme pH, salinity, or temperature.
- When a large proton motive force becomes prohibitively expensive, cells shift to metabolic modes compatible with smaller PMF values.
- Diversity of transport architectures arises because each is optimal only within a discrete environmental regime once efficiency is imperfect.
Where Pith is reading between the lines
- The bound supplies a quantitative limit that could be used to predict when environmental change will force metabolic reconfiguration in natural or synthetic cells.
- Because the bound depends only on leakage rates, it offers a way to compare energetic overhead across different membrane compositions without knowing the full transporter inventory.
- Variability in external conditions would further widen the range of viable architectures, suggesting that stable environments might support fewer transporter types.
Load-bearing premise
Cells operate at steady state where active transport exactly balances passive ion leakage.
What would settle it
Measure the actual metabolic power allocated to ion pumping and the free-energy dissipation rate from measured ion leaks in the same cell; the bound is falsified if pumping power falls below leakage dissipation under any steady-state condition.
Figures















read the original abstract
All cells must sustain ionic motive forces (IMFs) -- the electrochemical gradients of permeant ions, together with the membrane potential they produce -- to regulate intracellular pH, drive secondary transport, and power ATP synthesis. Because membranes are imperfectly impermeable, IMFs continuously dissipate through passive leakage, and active transport must compensate at an energetic cost that competes with growth and biosynthesis. How environmental conditions set this cost, and why cells across the tree of life share a common ionic logic yet deploy strikingly diverse transporter repertoires, has lacked a unifying quantitative account. Here we derive a thermodynamic lower bound on the power required to maintain IMFs at steady state. The bound equals the rate of free-energy dissipation by ion leakage, holds across a broad family of electrophysiological models, and is independent of organism, energy source, or transporter architecture. Cost minimization recovers, from first principles, the universal K+-rich, Na+-poor cytoplasm observed across taxa: asymmetric membrane permeabilities alone are sufficient to explain it. The same framework predicts that extremophiles face higher maintenance costs under extreme pH, salinity, and temperature, and that when sustaining a large proton motive force becomes prohibitive, cells should shift to metabolic regimes compatible with smaller PMF, providing a thermodynamic rationale for stress-induced metabolic reconfiguration. Finally, we show that perfect energetic efficiency is unattainable in practice, and that this very imperfection, combined with environmental variability, selects for the diversity of transport architectures observed in nature: each architecture is optimal within a discrete regime of environmental constraints.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript derives a thermodynamic lower bound on the power required to maintain ionic motive forces (IMFs) at steady state. This bound equals the rate of free-energy dissipation by passive ion leakage, holds across a broad family of electrophysiological models, and is independent of organism, energy source, or transporter architecture. The framework recovers the conserved K+-rich, Na+-poor cytoplasm from first principles via asymmetric permeabilities, predicts elevated maintenance costs for extremophiles under extreme conditions, and attributes observed diversity in transport architectures to imperfect efficiency combined with environmental variability.
Significance. If the central derivation holds, the result supplies a parameter-free, architecture-independent thermodynamic account of the minimal energetic cost of pH and ion homeostasis. This unifies disparate observations across taxa, supplies falsifiable predictions for extremophiles and stress-induced metabolic shifts, and offers a quantitative basis for why cells cannot achieve perfect efficiency. The explicit grounding in net-flux=0 energy balance and the breadth of the model family are particular strengths.
minor comments (3)
- [Abstract] The abstract states that the bound 'equals the rate of free-energy dissipation by ion leakage' but does not display the explicit expression; adding the mathematical form (e.g., as a sum over ionic species of J_i * Δμ_i) would improve immediate accessibility.
- [Introduction] The claim that cost minimization recovers the universal ionic composition is presented as a direct consequence of asymmetric permeabilities; a brief derivation sketch or reference to the relevant equation in the main text would strengthen this section.
- [Results] Figure captions for any plots of maintenance cost versus environmental parameters should explicitly note the fixed parameters (e.g., membrane potential, temperature) used in the numerical examples.
Simulated Author's Rebuttal
We thank the referee for the positive summary, the recognition of the architecture-independent thermodynamic bound, and the recommendation for minor revision. No specific major comments were enumerated in the report.
Circularity Check
No significant circularity in thermodynamic bound derivation
full rationale
The paper derives a lower bound on maintenance power equaling leakage dissipation at steady state. This follows directly from net ion flux = 0 enforcing energy balance, independent of transporter details or fitted parameters, across a general family of electrophysiological models. No self-definitional steps, renamed predictions, or load-bearing self-citations appear; the result is a standard first-principles consequence of steady-state thermodynamics and is self-contained.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Cells maintain steady-state ionic motive forces by balancing active transport against continuous passive leakage
- standard math Thermodynamic free-energy dissipation applies directly to ion leakage across imperfect membranes
Reference graph
Works this paper leans on
-
[1]
On the pH Dependence of Protein Stability
An-Suei Yang and Barry Honig. On the pH Dependence of Protein Stability. Journal of Molecular Biology, 231(2):459 – 474, 1993
1993
-
[2]
Krulwich, George Sachs, and Etana Padan
Terry A. Krulwich, George Sachs, and Etana Padan. Molecular aspects of bacterial pH sensing and homeostasis.Nature Reviews Microbiology, 9(5):330–343, May 2011
2011
-
[3]
Importance of pH homeosta- sis in metabolic health and diseases: crucial role of membrane proton 21 transport.BioMed research international, 2014:598986–598986, 2014
Wataru Aoi and Yoshinori Marunaka. Importance of pH homeosta- sis in metabolic health and diseases: crucial role of membrane proton 21 transport.BioMed research international, 2014:598986–598986, 2014. PMC4180894[pmcid]
2014
-
[4]
Structural investigation of the proton-coupled secondary trans- porters.Current Opinion in Structural Biology, 23(4):483–491, 2013
Nieng Yan. Structural investigation of the proton-coupled secondary trans- porters.Current Opinion in Structural Biology, 23(4):483–491, 2013
2013
-
[5]
D. C. Tosteson and J. F. Hoffman. Regulation of Cell Volume by Active Cation Transport in High and Low Potassium Sheep Red Cells.Journal of General Physiology, 44(1):169–194, 09 1960
1960
-
[6]
Alan R. Kay. How Cells Can Control Their Size by Pumping Ions.Frontiers in Cell and Developmental Biology, 5, 2017
2017
-
[7]
Cambridge Texts in Biomedical Engineering
DanielA.BeardandHongQian.Chemical Biophysics: Quantitative Analysis of Cellular Systems. Cambridge Texts in Biomedical Engineering. Cambridge University Press, 2008
2008
-
[8]
Garlid, Andrew D
Keith D. Garlid, Andrew D. Beavis, and Signe K. Ratkje. On the na- ture of ion leaks in energy-transducing membranes.Biochim Biophy Acta, 976(2):109 – 120, 1989
1989
-
[9]
Swain, and Teuta Pilizota
Guillaume Terradot, Ekaterina Krasnopeeva, Peter S. Swain, and Teuta Pilizota. Escherichia coli Maintains pH via the Membrane Potential.PRX Life, 2:043015, Nov 2024
2024
-
[10]
Ion transport and osmotic adjustment in escherichia coli in response to ionic and non-ionic osmotica.Environmental Microbiology, 11(1):137–148, 2009
Lana Shabala, John Bowman, Janelle Brown, Tom Ross, Tom McMeekin, and Sergey Shabala. Ion transport and osmotic adjustment in escherichia coli in response to ionic and non-ionic osmotica.Environmental Microbiology, 11(1):137–148, 2009
2009
-
[11]
Regu- lation of intracellular level of Na+, K+ and glycerol in Saccharomyces cerevisiae under osmotic stress.Molecular and Cellular Biochemistry, 158(2):121–124, May 1996
Sham Sunder, Arvinder Jit Singh, Sukhdeep Gill, and Balwant Singh. Regu- lation of intracellular level of Na+, K+ and glycerol in Saccharomyces cerevisiae under osmotic stress.Molecular and Cellular Biochemistry, 158(2):121–124, May 1996
1996
-
[12]
Energy flux and osmoregula- tion of Saccharomyces cerevisiae grown in chemostats under NaCl stress
R Olz, K Larsson, L Adler, and L Gustafsson. Energy flux and osmoregula- tion of Saccharomyces cerevisiae grown in chemostats under NaCl stress. Journal of Bacteriology, 175(8):2205–2213, April 1993
1993
-
[13]
The Regulation of Potassium Fluxes for the Adjustment and Maintenance of Potassium Levels in Escherichia coli
Jean MEURY and Adam KEPES. The Regulation of Potassium Fluxes for the Adjustment and Maintenance of Potassium Levels in Escherichia coli. European Journal of Biochemistry, 119(1):165–170, September 1981
1981
-
[14]
Wolfgang Epstein and Stanley G. Schultz. Cation transport in escherichia coli.The Journal of General Physiology, 49(3):469–481, January 1966
1966
-
[15]
Schultz, Norman L
Stanley G. Schultz, Norman L. Wilson, and Wolfgang Epstein. Cation Transport in Escherichia coli.The Journal of General Physiology, 46(1):159– 166, 1962. 22
1962
-
[16]
Schultz and A
Stanley G. Schultz and A. K. Solomon. Cation Transport in Escherichia coli.The Journal of General Physiology, 45(2):355–369, 1961
1961
-
[17]
Yoshinori Toyoshima and T. E. Thompson. Chloride flux in bilayer mem- branes. Chloride permeability in aqueous dispersions of single-walled, bilayer vesicles.Biochemistry, 14(7):1525–1531, Apr 1975
1975
-
[18]
Driessen, Jack L.C.M
Arnold J.M. Driessen, Jack L.C.M. van de Vossenberg, and Wil N. Konings. Membrane composition and ion-permeability in extremophiles.FEMS Microbiology Reviews, 18(2–3):139–148, May 1996
1996
-
[19]
Williamson, and Terence Hwa
Markus Basan, Sheng Hui, Hiroyuki Okano, Zhongge Zhang, Yang Shen, James R. Williamson, and Terence Hwa. Overflow metabolism in Escherichia coli results from efficient proteome allocation.Nature, 528(7580):99–104, Dec 2015. Article
2015
-
[20]
Ancient Systems of Sodium/Potassium Homeostasis as Predecessors of Membrane Bioenergetics.Biochemistry (Mosc), 80(5):495–516, May 2015
D V Dibrova, M Y Galperin, E V Koonin, and A Y Mulkidjanian. Ancient Systems of Sodium/Potassium Homeostasis as Predecessors of Membrane Bioenergetics.Biochemistry (Mosc), 80(5):495–516, May 2015
2015
-
[21]
Zhao, and K
Wei Bai, K.S. Zhao, and K. Asami. Dielectric properties of E. coli cell as simulated by the three-shell spheroidal model.Biophy Chem, 122(2):136 – 142, 2006
2006
-
[22]
Inflating bacterial cells by increased protein synthesis.Molecular Systems Biology, 11(10):MSB156178, Oct 2015
Markus Basan, Manlu Zhu, Xiongfeng Dai, Mya Warren, Daniel Sévin, Yi-Ping Wang, and Terence Hwa. Inflating bacterial cells by increased protein synthesis.Molecular Systems Biology, 11(10):MSB156178, Oct 2015
2015
-
[23]
Dynamics of Escherichia coli’s passive response to a sudden decrease in external osmolarity.Proc Nat Acad Sci USA, 113(40):E5838–E5846, 2016
Renata Buda, Yunxiao Liu, Jin Yang, Smitha Hegde, Keiran Stevenson, Fan Bai, and Teuta Pilizota. Dynamics of Escherichia coli’s passive response to a sudden decrease in external osmolarity.Proc Nat Acad Sci USA, 113(40):E5838–E5846, 2016
2016
-
[24]
eQuilibra- tor—the biochemical thermodynamics calculator.Nucleic Acids Research, 40(D1):D770–D775, 01 2012
Avi Flamholz, Elad Noor, Arren Bar-Even, and Ron Milo. eQuilibra- tor—the biochemical thermodynamics calculator.Nucleic Acids Research, 40(D1):D770–D775, 01 2012
2012
-
[25]
Determination of ionic permeability coefficients of the plasma membrane of Xenopus laevis oocytes under voltage clamp.J Physiol, 413(1):199–211, 1989
P F Costa, M G Emilio, P L Fernandes, H G Ferreira, and K G Ferreira. Determination of ionic permeability coefficients of the plasma membrane of Xenopus laevis oocytes under voltage clamp.J Physiol, 413(1):199–211, 1989
1989
-
[26]
Robertson.The lively membranes
R.N. Robertson.The lively membranes. Cambridge University Press, 2022
2022
-
[27]
Dynamics of Escherichia coli s passive response to a sudden decrease in external osmolarity.Proc
Renata Buda, Yunxiao Liu, Jin Yang, Smitha Hegde, Keiran Stevenson, Fan Bai, and Teuta Pilizota. Dynamics of Escherichia coli s passive response to a sudden decrease in external osmolarity.Proc. Natl. Acad. Sci. USA, 113(40):E5838–E5846, oct 2016. 23
2016
-
[28]
Sauls, Norbert S
Sattar Taheri-Araghi, Serena Bradde, John T. Sauls, Norbert S. Hill, Pe- tra Anne Levin, Johan Paulsson, Massimo Vergassola, and Suckjoon Jun. Cell-sizecontrol andhomeostasis inbacteria.Current Biology, 25(3):385–391, 2015
2015
-
[29]
Keener and J
J. Keener and J. Sneyd.Mathematical Physiology: I. Springer, New York, New York, 2009. A Constants and Parameters The following tables collect the constant parameters used for calculations and numerical simulations. Values refer toE. coliat T = 298K unless otherwise noted. Name Symbol Units Value Faraday constant F C/mol 96485 Gas constant R J/mol/K 8.31 ...
2009
-
[30]
First we solve Eq.(33) in the two dimensionsαY ∈{−2,−1, 0, +1, +2} and∆ GC+∈[−500, 0]mV for which the approximation was evaluated to be robust in [9]
-
[31]
We then rearrange Eq.(38) as to express the SMF (Sodium Motive Force) as a function of the KMF (Potassium Motive Force): ∆G Na+ =η−1ln (1 2 1 αNa+ ·eη(∆G C+−∆ψ)−αK+ αNa+ ·eη(∆G K+−∆ψ) ) + ∆ψ (40)
-
[32]
and to ensure fine grained sampling of the SMF and KMF we also express the KMF as a function of the SMF: ∆G K+ =η−1ln (1 2 1 αK+ ·eη(∆G C+−∆ψ)−αNa+ αK+ ·eη(∆G Na+−∆ψ) ) + ∆ψ (41) and introducing the extracellular sodium to extracellular potassium ratio: β=αNa+ αK+ (42) we transform Eqs. (40) and (41) into: ∆G Na+ =η−1ln [( 1 β+ 1 ) ·eη(∆G C+−∆ψ)−1 β·eη(∆G...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.