Recognition: unknown
Effect of antibiotic spectrum on the abundance of resistant bacteria in multispecies communities
Pith reviewed 2026-05-10 03:10 UTC · model grok-4.3
The pith
A mathematical net-effect measure from microbial interaction networks predicts how antibiotic spectrum affects the abundance of resistant bacteria.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
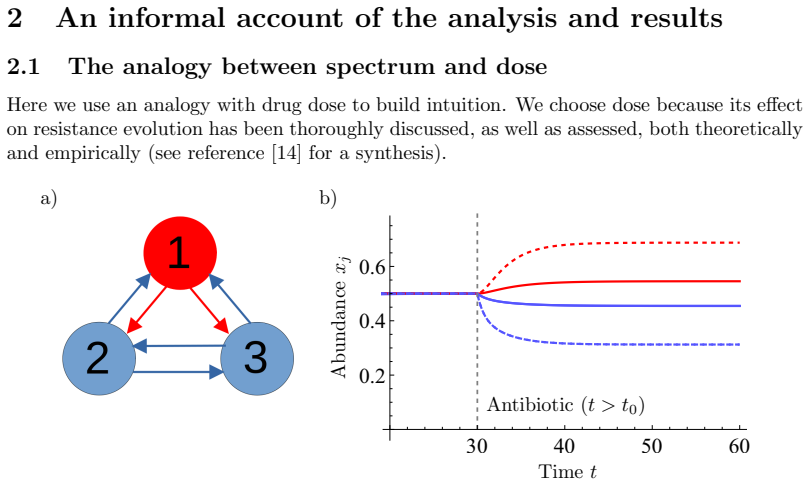
By analysing established community ecology theory, the authors construct a net-effect measure that sums all direct and indirect influences one taxon exerts on another within the interaction network. Applying this measure to antibiotic exposure shows that the spectrum of the drug (the set of species it suppresses) determines the expected change in abundance of resistant taxa, thereby providing a formal link between antibiotic choice and resistance dynamics in multispecies communities.
What carries the argument
The net-effect measure, which aggregates direct and indirect interactions across the entire community network to quantify one taxon's total influence on another's abundance.
If this is right
- Different antibiotic spectra produce predictable, network-dependent changes in resistant taxon abundance rather than uniform selection.
- Narrow-spectrum antibiotics can sometimes increase resistant taxa more than broad-spectrum ones when indirect positive effects dominate the network.
- The framework supplies a formal theoretical basis for designing experiments that test optimal antibiotic choice in complex communities.
- Resistance management strategies should incorporate the structure of the resident microbial interaction network rather than species identity alone.
Where Pith is reading between the lines
- The same net-effect logic could be applied to other community perturbations such as phage therapy or dietary changes that alter taxon abundances.
- If interaction networks can be inferred from metagenomic data, the measure might allow in silico screening of antibiotic spectra for patient-specific microbiomes.
- The approach highlights a possible route to test whether resistance dynamics in the gut or soil follow the same net-effect rules derived from the abstract model.
Load-bearing premise
The net-effect measure derived from the interaction network fully captures the relevant dynamics of resistance emergence and spread under antibiotic exposure in real multispecies communities.
What would settle it
A controlled experiment that assembles a microbial community with a known interaction network, applies antibiotics of contrasting spectra, and measures whether the observed shifts in resistant taxon abundance match the directions and relative magnitudes predicted by the net-effect measure.
Figures


read the original abstract
Antibiotic resistance is a major threat to global health. It emerges in multispecies microbial communities under antibiotic exposure. This makes antibiotic spectrum -- a drug's distribution of effects across species -- a potential key parameter in resistance management. However, we currently lack evolutionary theory for resistance dynamics in a multispecies setting. Analysing established community ecology theory, we develop a simple mathematical measure for how one taxon (strain or species) affects another taxon through all direct and indirect interactions in a complex interaction network. Using this, we derive the expected effects of different antibiotic spectra on the abundance of resistant taxa in microbial communities. This furthers our understanding of microbial evolutionary ecology in multispecies communities, and provides a formal theoretical basis for empirical work on optimal antibiotic choice.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper analyzes established community ecology theory to construct a simple mathematical measure of net effects between taxa (via all direct and indirect paths in an interaction network). It then uses this measure to derive the expected abundance responses of resistant taxa to narrow- versus broad-spectrum antibiotics in multispecies microbial communities, with the goal of providing a formal theoretical basis for resistance management.
Significance. If the derivation is internally consistent and the modeling assumptions are justified, the work would offer a useful theoretical bridge between network ecology and antibiotic resistance, potentially informing empirical studies on spectrum choice. The approach of repurposing net-effect calculations from interaction matrices is a clear strength when the mapping to resistance abundance is made explicit and falsifiable.
major comments (1)
- The central derivation models abundance shifts under a fixed interaction network with resistance treated as a pre-existing trait. However, the abstract claims to address 'evolutionary theory for resistance dynamics' and 'resistance emergence and spread.' The net-effect measure does not appear to incorporate spectrum-dependent selection (differential killing rates on sensitive vs. resistant cells), mutation supply, or horizontal gene transfer that could alter effective interactions under treatment. This assumption is load-bearing for the claim that the measure predicts effects on resistant taxa abundance under antibiotic exposure.
minor comments (1)
- The abstract asserts that a derivation exists but supplies no equations, proof outline, or validation against simulations or data; the full manuscript should include these in the methods or results to allow direct assessment of the net-effect construction.
Simulated Author's Rebuttal
We thank the referee for their positive assessment of the manuscript's potential contribution and for the detailed comment. We address the major point below, clarifying the model's scope and making targeted revisions to avoid overstatement.
read point-by-point responses
-
Referee: The central derivation models abundance shifts under a fixed interaction network with resistance treated as a pre-existing trait. However, the abstract claims to address 'evolutionary theory for resistance dynamics' and 'resistance emergence and spread.' The net-effect measure does not appear to incorporate spectrum-dependent selection (differential killing rates on sensitive vs. resistant cells), mutation supply, or horizontal gene transfer that could alter effective interactions under treatment. This assumption is load-bearing for the claim that the measure predicts effects on resistant taxa abundance under antibiotic exposure.
Authors: We agree that the model treats resistance as a pre-existing trait within a fixed interaction network and derives abundance responses to antibiotic spectra via net effects (direct plus indirect paths). It does not incorporate mutation supply, horizontal gene transfer, or explicit spectrum-dependent selection dynamics beyond the assumption that resistant taxa are unaffected while sensitive taxa experience reduced growth. The abstract's reference to 'evolutionary theory for resistance dynamics' and 'resistance emergence and spread' is therefore imprecise and risks overstating the scope. The contribution is an ecological measure, repurposed from community ecology, that predicts how antibiotic spectrum modulates the relative abundance of already-resistant taxa through community interactions; this provides a formal basis for understanding resistance in multispecies settings but does not model the evolutionary processes themselves. We have revised the abstract and the opening paragraphs of the introduction to state the scope more precisely as an ecological framework that can inform resistance management and dynamics, without claiming to derive evolutionary mechanisms. This change preserves the central derivation and results while addressing the referee's concern directly. revision: yes
Circularity Check
Derivation applies established community ecology measure to new context without reduction to inputs
full rationale
The paper states it analyzes established community ecology theory to develop a net-effect measure across direct and indirect interactions in a network, then applies this measure to derive expected abundance shifts for resistant taxa under different antibiotic spectra. No equations or steps are shown that define the measure in terms of the target antibiotic-resistance outcome or that rename a fitted parameter as a prediction. The abstract presents the measure as an independent analytical tool drawn from prior theory, with the resistance application as a downstream use rather than a self-referential construction. No self-citations are invoked as load-bearing uniqueness theorems. This is a self-contained derivation with independent content.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Microbial communities can be represented as a network of direct and indirect interaction effects whose net impact on each taxon can be summarized by a single scalar measure.
Reference graph
Works this paper leans on
-
[1]
Fact Sheet
World Health Organisation.Antimicrobial resistance. Fact Sheet. Accessed = 2025- 02-18. 2023.url:https : / / www . who . int / news - room / fact - sheets / detail / antimicrobial-resistance
2025
-
[2]
Population biological principles of drug-resistance evolution in infectious diseases
Pia Abel zur Wiesch et al. “Population biological principles of drug-resistance evolution in infectious diseases”. In:The Lancet Infectious Diseases11 (3 2011), pp. 236–247.doi: doi:10.1016/S1473- 3099(10)70264- 4.url:https://doi.org/10.1016/S1473- 3099(10)70264-4
-
[3]
O.J. Dyar et al. “ESCMID generic competencies in antimicrobial prescribing and stew- ardship: towards a European consensus”. In:Clinical Microbiology and Infection25.1 (2019).doi:https://doi.org/10.1016/j.cmi.2018.09.022
-
[4]
A practical toolkit
World Health Organization.Antimicrobial stewardship programmes in health-care fa- cilities in low- and middle-income countries. A practical toolkit. 2019
2019
-
[5]
accessed 2025-02-21
European Commission.European Health Union: EU steps up the fight against an- timicrobial resistance. accessed 2025-02-21. 2023.url:https : / / ec . europa . eu / commission/presscorner/detail/en/ip_23_3187
2025
-
[6]
accessed 2025-2-21
World Health Organization.AWaRe classification of antibiotics for evaluation and monitoring of use, 2023. accessed 2025-2-21. 2023.url:https : / / www . who . int / publications/i/item/WHO-MHP-HPS-EML-2023.04
2023
-
[7]
Unrealized targets in the discovery of antibiotics for Gram-negative bacterial infections
Ursula Theuretzbacher et al. “Unrealized targets in the discovery of antibiotics for Gram-negative bacterial infections”. In:Nature Reviews Drug Discovery22 (12 2023), pp. 957–975.url:https://doi.org/10.1038/s41573-023-00791-6
-
[8]
Bystander Selection for Antimicrobial Resistance: Implications for Patient Health
Valerie J. Morley, Robert J. Woods, and Andrew F. Read. “Bystander Selection for Antimicrobial Resistance: Implications for Patient Health”. In:Trends in Microbiology 27 (10 2019), pp. 864–877.url:https://doi.org/10.1016/j.tim.2019.06.004. 17
-
[9]
2017.url:https://eur- lex.europa.eu/legal- content/EN/ALL/?uri= CELEX:52017XC0701(01)
European Commission.EU Guidelines for the prudent use of antimicrobials in human health. 2017.url:https://eur- lex.europa.eu/legal- content/EN/ALL/?uri= CELEX:52017XC0701(01)
2017
-
[10]
acessed 2025-02-21
World Health Organization.In the face of slow progress, WHO offers a new tool and sets a target to accelerate action against antimicrobial resistance. acessed 2025-02-21. 2019.url:https://www.who.int/news/item/18-06-2019-in-the-face-of-slow- progress-who-offers-a-new-tool-and-sets-a-target-to-accelerate-action- against-antimicrobial-resistance
2025
-
[11]
Christine Tedijanto et al. “Estimating the proportion of bystander selection for an- tibiotic resistance among potentially pathogenic bacterial flora”. In:Proceedings of the National Academy of Sciences115.51 (2018), E11988–E11995.url:https : / / www . pnas.org/doi/abs/10.1073/pnas.1810840115
-
[12]
Bacterial species rarely work together
Jacob D. Palmer and Kevin R. Foster. “Bacterial species rarely work together”. In: Science376.6593 (2022), pp. 581–582.url:https://www.science.org/doi/abs/10. 1126/science.abn5093
2022
-
[13]
Is selection relevant in the evolution- ary emergence of drug resistance?
Troy Day, Silvie Huijben, and Andrew F. Read. “Is selection relevant in the evolution- ary emergence of drug resistance?” In:Trends in Microbiology23 (3 2015), pp. 126– 133.url:https://doi.org/10.1016/j.tim.2015.01.005
-
[14]
Does High-Dose Antimicrobial Chemotherapy Prevent the Evolution of Resistance?
Troy Day and Andrew F. Read. “Does High-Dose Antimicrobial Chemotherapy Prevent the Evolution of Resistance?” In:PLOS Computational Biology12 (Jan. 2016), pp. 1– 20.doi:10 . 1371 / journal . pcbi . 1004689.url:https : / / doi . org / 10 . 1371 / journal.pcbi.1004689
2016
-
[15]
Methods of quantifying interactions among populations using Lotka-Volterra models
Jacob D Davis et al. “Methods of quantifying interactions among populations using Lotka-Volterra models”. In:Frontiers in Systems Biology2 (2022), p. 1021897
2022
-
[16]
Microbial communities as dynamical systems
Didier Gonze et al. “Microbial communities as dynamical systems”. In:Current Opin- ion in Microbiology44 (2018). Microbiota, pp. 41–49.issn: 1369-5274.doi:https: //doi.org/10.1016/j.mib.2018.07.004.url:https://www.sciencedirect.com/ science/article/pii/S1369527418300092
work page doi:10.1016/j.mib.2018.07.004.url:https://www.sciencedirect.com/ 2018
-
[17]
Defining the benefits of antibiotic resistance in commensals and the scope for resistance optimization
Kristofer Wollein Waldetoft et al. “Defining the benefits of antibiotic resistance in commensals and the scope for resistance optimization”. In:Mbio14.1 (2023), e01349– 22
2023
-
[18]
The ecology of the micro- biome: Networks, competition, and stability
Katharine Z. Coyte, Jonas Schluter, and Kevin R. Foster. “The ecology of the micro- biome: Networks, competition, and stability”. In:Science350.6261 (2015), pp. 663– 666.doi:10.1126/science.aad2602.url:https://www.science.org/doi/abs/ 10.1126/science.aad2602
work page doi:10.1126/science.aad2602.url:https://www.science.org/doi/abs/ 2015
-
[19]
The Long-Term Stability of the Human Gut Microbiota
Jeremiah J. Faith et al. “The Long-Term Stability of the Human Gut Microbiota”. In: Science341.6141 (2013), p. 1237439.doi:10.1126/science.1237439.url:https: //www.science.org/doi/abs/10.1126/science.1237439
-
[20]
Pablo Lech´ on-Alonso et al. “Robust coexistence in competitive ecological communi- ties”. In:Nat. Commun.17.2637 (2026).doi:10.1038/s41467- 026- 69151- 3.url: https://doi.org/10.1038/s41467-026-69151-3. 18
-
[21]
Strange attractors in Volterra equations for species in competition
A. Arneodo et al. “Strange attractors in Volterra equations for species in competition”. In:J. Math. Biol.14.2 (1982), pp. 153–157.issn: 0303-6812,1432-1416.doi:10.1007/ BF01832841.url:https://doi.org/10.1007/BF01832841
-
[22]
A. Arneodo, P. Coullet, and C. Tresser. “Occurrence of strange attractors in three- dimensional Volterra equations”. In:Phys. Lett. A79.4 (1980), pp. 259–263.issn: 0375-9601,1873-2429.doi:10.1016/0375- 9601(80)90342- 4.url:https://doi. org/10.1016/0375-9601(80)90342-4
-
[23]
Chaos in low-dimensional Lotka-Volterra models of competition
J. A. Vano et al. “Chaos in low-dimensional Lotka-Volterra models of competition”. In: Nonlinearity19.10 (2006), pp. 2391–2404.issn: 0951-7715,1361-6544.doi:10.1088/ 0951-7715/19/10/006.url:https://doi.org/10.1088/0951-7715/19/10/006
-
[24]
Morris W. Hirsch. “Systems of differential equations that are competitive or coop- erative. II. Convergence almost everywhere”. In:SIAM J. Math. Anal.16.3 (1985), pp. 423–439.issn: 0036-1410.doi:10.1137/0516030.url:https://doi.org/10. 1137/0516030
-
[25]
Systems of differential equations which are competitive or coop- erative. III. Competing species
Morris W. Hirsch. “Systems of differential equations which are competitive or coop- erative. III. Competing species”. In:Nonlinearity1.1 (1988), pp. 51–71.issn: 0951- 7715,1361-6544.url:http://stacks.iop.org/0951-7715/1/51
1988
-
[26]
On the differential equations of species in competition
S. Smale. “On the differential equations of species in competition”. In:J. Math. Biol. 3.1 (1976), pp. 5–7.issn: 0303-6812,1432-1416.doi:10 . 1007 / BF00307854.url: https://doi.org/10.1007/BF00307854
-
[27]
The dynamics of the H´ enon map
Michael Benedicks and Lennart Carleson. “The dynamics of the H´ enon map”. In:Ann. of Math. (2)133.1 (1991), pp. 73–169.issn: 0003-486X,1939-8980.doi:10 . 2307 / 2944326.url:https://doi.org/10.2307/2944326
-
[28]
A rigorous ODE solver and Smale’s 14th problem
Warwick Tucker. “A rigorous ODE solver and Smale’s 14th problem”. In:Found. Comput. Math.2.1 (2002), pp. 53–117.issn: 1615-3375,1615-3383.doi:10 . 1007 / s002080010018.url:https://doi.org/10.1007/s002080010018
-
[29]
Complex systems in ecology: a guided tour with large Lotka–Volterra models and random matrices
Imane Akjouj et al. “Complex systems in ecology: a guided tour with large Lotka–Volterra models and random matrices”. In:Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences480.2285 (Mar. 2024), p. 20230284.issn: 1364-5021. doi:10.1098/rspa.2023.0284. eprint:https://royalsocietypublishing.org/ rspa/article-pdf/doi/10.1098/rs...
-
[30]
Equilibrium and surviving species in a large Lotka-Volterra system of differential equations
Maxime Clenet, Fran¸ cois Massol, and Jamal Najim. “Equilibrium and surviving species in a large Lotka-Volterra system of differential equations”. In:J. Math. Biol.87.1 (2023), Paper No. 13, 32.issn: 0303-6812,1432-1416.doi:10 . 1007 / s00285 - 023 - 01939-z.url:https://doi.org/10.1007/s00285-023-01939-z
-
[31]
Positive solutions for large random linear systems
Pierre Bizeul and Jamal Najim. “Positive solutions for large random linear systems”. In:Proc. Amer. Math. Soc.149.6 (2021), pp. 2333–2348.issn: 0002-9939,1088-6826. doi:10.1090/proc/15383.url:https://doi.org/10.1090/proc/15383
work page doi:10.1090/proc/15383.url:https://doi.org/10.1090/proc/15383 2021
-
[32]
Global Stability in Many-Species Systems
B. S. Goh. “Global Stability in Many-Species Systems”. In:The American Naturalist 111.977 (1977), pp. 135–143.issn: 00030147, 15375323.url:http://www.jstor.org/ stable/2459985(visited on 12/15/2025). 19
-
[33]
The stability of gen- eralized Volterra equations
Yasuhiro Takeuchi, Norihiko Adachi, and Hidekatsu Tokumaru. “The stability of gen- eralized Volterra equations”. In:J. Math. Anal. Appl.62.3 (1978), pp. 453–473.issn: 0022-247X.doi:10 . 1016 / 0022 - 247X(78 ) 90139 - 7.url:https : / / doi . org / 10 . 1016/0022-247X(78)90139-7
1978
-
[34]
Global stability of ecosystems of the generalized Volterra type
Yasuhiro Takeuchi, Norihiko Adachi, and Hidekatsu Tokumaru. “Global stability of ecosystems of the generalized Volterra type”. In:Math. Biosci.42.1-2 (1978), pp. 119– 136.issn: 0025-5564,1879-3134.doi:10.1016/0025-5564(78)90010-X.url:https: //doi.org/10.1016/0025-5564(78)90010-X
-
[35]
The existence of globally stable equilibria of ecosystems of the generalized Volterra type
Yasuhiro Takeuchi and Norihiko Adachi. “The existence of globally stable equilibria of ecosystems of the generalized Volterra type”. In:J. Math. Biol.10.4 (1980), pp. 401– 415.issn: 0303-6812,1432-1416.doi:10.1007/BF00276098.url:https://doi.org/ 10.1007/BF00276098
-
[37]
Universal classe s of hash functions
Yasuhiro Takeuchi and Norihiko Adachi. “Stable equilibrium of systems of generalized Volterra type”. In:J. Math. Anal. Appl.88.1 (1982), pp. 157–169.issn: 0022-247X. doi:10.1016/0022- 247X(82)90183- 4.url:https://doi.org/10.1016/0022- 247X(82)90183-4
-
[38]
Stable equilibria in the Lotka-Volterra equations
Magnus Aspenberg, Erik A. Martens, and Kristofer Wollein Waldetoft. “Necessary condition for stable equilibria in the Lotka-Volterra equations”. In:arXiv preprint, arXiv:2512.13347(2025)
work page internal anchor Pith review Pith/arXiv arXiv 2025
-
[39]
Commensal antimicrobial resistance mediates microbiome resilience to antibiotic disruption
Shakti K Bhattarai et al. “Commensal antimicrobial resistance mediates microbiome resilience to antibiotic disruption”. In:Science translational medicine16.730 (2024), eadi9711
2024
-
[40]
Equilibrium in a large Lotka- Volterra system with pairwise correlated interactions
Maxime Clenet, Hafedh El Ferchichi, and Jamal Najim. “Equilibrium in a large Lotka- Volterra system with pairwise correlated interactions”. In:Stochastic Process. Appl. 153 (2022), pp. 423–444.issn: 0304-4149,1879-209X.doi:10.1016/j.spa.2022.08. 004.url:https://doi.org/10.1016/j.spa.2022.08.004. Appendix A Numerical frequency sampling of attractors We bri...
-
[41]
For each realization, we com- puted the trajectories using initial conditions sampled i.i.d
We randomly draw interactions with|α ij| ≤1 (i.i.d.). For each realization, we com- puted the trajectories using initial conditions sampled i.i.d. uniformly from the positive domainx i(0)∈[0, L] for alli= 1, . . . , n. Note that trajectories with such initial con- ditions remain forever positive. We sampledn i initial conditions forn r realization of the ...
-
[42]
We compute trajectories for a time ofTunits
The asymptotic behavior of each trajectory is classified into one of three (main) distinct behaviors: (stable) equilibrium, oscillatory behavior, divergence (i.e.,x k(t)→ ∞for somek). We compute trajectories for a time ofTunits. Transient behavior is assumed to subside afterT t time units, and fort∈ T:= [0.5T, T] we measured the ’amplitude’ A:= max tT ||x...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.