Recognition: unknown
Distinct Structural Dynamics of the Semiquinone State Define a Signalling Pathway in Avian Cryptochrome
Pith reviewed 2026-05-10 00:46 UTC · model grok-4.3
The pith
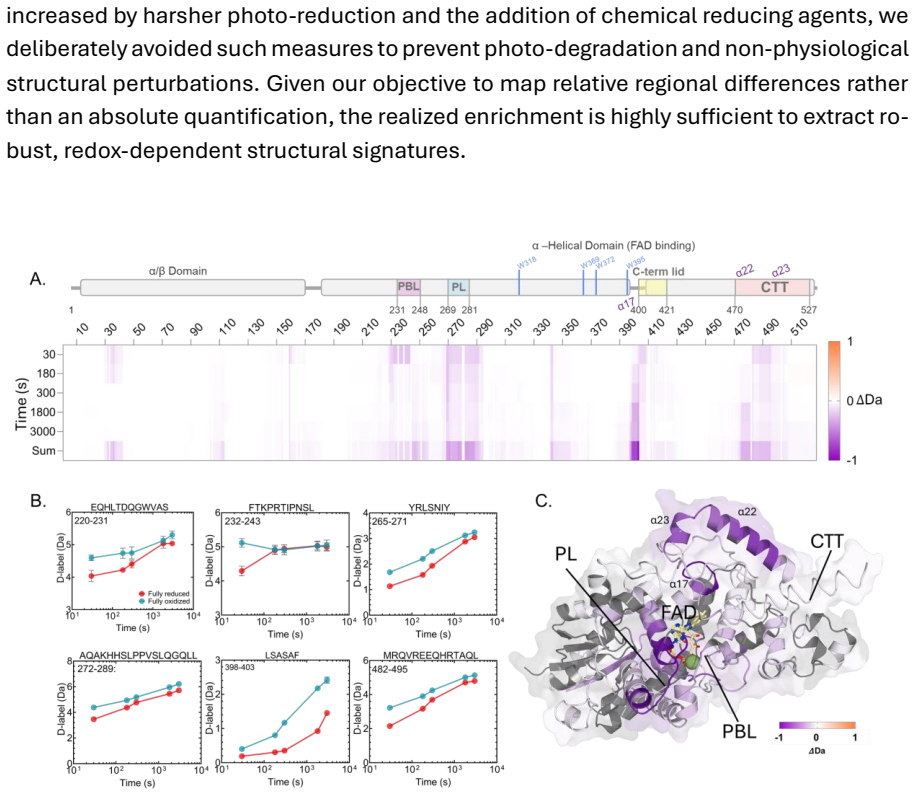
The semiquinone state in European robin cryptochrome 4a shows a distinct non-monotonic conformational signature with transient destabilization of the phosphate-binding and protrusion loops.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Photochemical reduction drives allosteric structural transitions across key functional nodes including the phosphate-binding loop, protrusion loop, FAD-proximal helix alpha17, and the C-terminal alpha22/alpha23 network. The transient semiquinone exhibits a distinct, non-monotonic conformational signature characterized by transient destabilization of the PBL and PL, contrasting sharply with the global rigidification observed in the fully reduced state. These findings establish the semiquinone as a structurally unique and functionally competent biological entity.
What carries the argument
Redox state-resolved hydrogen/deuterium-exchange mass spectrometry (HDX-MS), which reports local flexibility by measuring how readily backbone hydrogens exchange with deuterium in each redox state and thereby isolates the structural fingerprint of the transient semiquinone.
Where Pith is reading between the lines
- Mutations that selectively stabilize or destabilize the PBL or PL could be used to test whether the semiquinone conformation is necessary and sufficient for downstream signaling.
- The non-monotonic structural trajectory suggests the protein may use a ratchet-like mechanism that holds the semiquinone in a signaling-ready shape before progressing to the fully reduced state.
- Parallel HDX-MS measurements on cryptochromes from non-migratory species could reveal whether the same semiquinone signature is conserved only in animals that rely on magnetic compasses.
- The work points toward the possibility of designing small-molecule stabilizers that lock the semiquinone conformation to probe its interaction with putative signaling partners.
Load-bearing premise
The HDX-MS differences measured specifically in the semiquinone state represent the conformation that initiates the cellular signaling cascade for magnetic sensing rather than an off-pathway or non-signaling species.
What would settle it
A site-directed mutation that removes the semiquinone-specific destabilization of the PBL and PL while still permitting semiquinone formation would eliminate magnetic orientation behavior in the bird while leaving other light responses intact.
Figures

read the original abstract
The light-dependent magnetic compass of night-migratory songbirds is widely hypothesized to rely on the radical pair mechanism within retinal cryptochrome. However, bridging the mechanistic gap between microsecond quantum spin dynamics and the long-lived, global protein conformational changes required for cellular signalling remains a formidable challenge. Here, we apply redox state-resolved hydrogen/deuterium-exchange mass spectrometry (HDX-MS) to map the conformational landscape of European robin cryptochrome 4a (ErCry4a) across its photocycle. We reveal that photochemical reduction drives robust, allosteric structural transitions across key functional nodes, including the phosphate-binding loop (PBL), protrusion loop (PL), FAD-proximal helix {\alpha}17, and the C-terminal {\alpha}22/{\alpha}23 network. Crucially, we isolate the structural fingerprint of the transient semiquinone, the presumed signalling species. Rather than acting as a linear structural stepping-stone, the semiquinone exhibits a distinct, non-monotonic conformational signature characterized by a transient destabilization of the PBL and PL, contrasting sharply with the global rigidification observed in the fully reduced state. These findings establish the semiquinone as a structurally unique and functionally competent biological entity. Our results provide direct biophysical evidence for a dedicated, high-fidelity structural signalling cascade, detailing how localized quantum-level photochemistry is translated into the precise conformational dynamics required for animal navigation.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript applies redox-state-resolved HDX-MS to European robin cryptochrome 4a (ErCry4a) to map conformational changes across the photocycle. It reports that photochemical reduction induces allosteric transitions at nodes including the PBL, PL, α17, and C-terminal α22/α23 network. The central result is that the transient semiquinone state displays a distinct non-monotonic signature of PBL/PL destabilization, in contrast to global rigidification in the fully reduced state; this is interpreted as establishing the semiquinone as the unique, functionally competent signaling species that bridges radical-pair photochemistry to cellular signaling for magnetic sensing.
Significance. If the structural interpretation is validated, the work supplies direct experimental mapping of how local redox changes propagate to specific loops and helices, addressing a key mechanistic gap between microsecond spin dynamics and longer-timescale conformational signaling in avian cryptochrome. The non-monotonic character of the semiquinone dynamics is a potentially important observation that could refine models of the photocycle.
major comments (2)
- [Abstract] Abstract and main claims: The assertion that the HDX-MS data 'establish the semiquinone as a structurally unique and functionally competent biological entity' and provide 'direct biophysical evidence for a dedicated, high-fidelity structural signalling cascade' is load-bearing for the central thesis. The measurements demonstrate differential deuterium uptake between redox states, but the manuscript does not supply orthogonal validation (state-specific cross-linking, signaling-deficient mutants, or direct comparison to downstream effectors) to confirm that the observed PBL/PL destabilization occurs on the radical-pair-active population and is sufficient to initiate cellular signaling.
- [Results] Results/Methods (HDX-MS data presentation): The abstract and summary provide no raw uptake values, error bars, replicate numbers, statistical tests, or exclusion criteria for the semiquinone versus oxidized/reduced comparisons. Without these, the robustness of the claimed 'transient destabilization' and 'non-monotonic' signature cannot be evaluated, weakening the ability to judge whether the differences are specific to the signaling-competent semiquinone or could arise from off-pathway species.
minor comments (2)
- [Abstract] The abstract contains LaTeX artifacts (e.g., {α}17) that should be rendered as proper Greek symbols in the final manuscript.
- [Abstract] The final sentence of the abstract is long and compound; splitting it would improve readability of the key conclusion.
Simulated Author's Rebuttal
We thank the referee for the constructive feedback and positive assessment of the work's significance. We have revised the manuscript to moderate the abstract claims and to provide fuller details on HDX-MS data processing, replicates, and statistics. These changes improve clarity and transparency while preserving the core interpretation of the non-monotonic semiquinone signature.
read point-by-point responses
-
Referee: [Abstract] Abstract and main claims: The assertion that the HDX-MS data 'establish the semiquinone as a structurally unique and functionally competent biological entity' and provide 'direct biophysical evidence for a dedicated, high-fidelity structural signalling cascade' is load-bearing for the central thesis. The measurements demonstrate differential deuterium uptake between redox states, but the manuscript does not supply orthogonal validation (state-specific cross-linking, signaling-deficient mutants, or direct comparison to downstream effectors) to confirm that the observed PBL/PL destabilization occurs on the radical-pair-active population and is sufficient to initiate cellular signaling.
Authors: We agree that the original abstract language was overly definitive and that orthogonal validation (e.g., mutants or cross-linking) would further strengthen functional assignment. The HDX-MS data provide the first direct structural map distinguishing the semiquinone from both oxidized and fully reduced states, revealing its unique non-monotonic PBL/PL destabilization. We have revised the abstract to replace 'establish' with 'support' and 'provide direct biophysical evidence' with 'offer structural evidence consistent with', while adding a sentence noting that downstream functional validation lies beyond the present biophysical scope. This preserves the central observation without overstating its immediate implications for cellular signaling. revision: partial
-
Referee: [Results] Results/Methods (HDX-MS data presentation): The abstract and summary provide no raw uptake values, error bars, replicate numbers, statistical tests, or exclusion criteria for the semiquinone versus oxidized/reduced comparisons. Without these, the robustness of the claimed 'transient destabilization' and 'non-monotonic' signature cannot be evaluated, weakening the ability to judge whether the differences are specific to the signaling-competent semiquinone or could arise from off-pathway species.
Authors: We apologize for the insufficient detail in the initial presentation. The full manuscript contains uptake plots with error bars (SD from n=3 biological replicates) and peptide-level comparisons, but these were not explicitly summarized in the main text. We have now added a dedicated paragraph in the Results section reporting replicate numbers (n=3 independent HDX-MS experiments), statistical testing (two-tailed unpaired t-tests, p<0.05 threshold after Benjamini-Hochberg correction), and peptide exclusion criteria (minimum 3 overlapping peptides per region, >70% sequence coverage). Representative raw uptake values and full statistical tables have been moved to Supplementary Information. These additions allow direct evaluation of the non-monotonic signature and confirm that the PBL/PL destabilization is statistically significant and specific to the semiquinone population under the controlled illumination conditions used. revision: yes
Circularity Check
No circularity: purely experimental HDX-MS conformational mapping
full rationale
The paper reports experimental HDX-MS measurements of deuterium uptake differences across redox states of ErCry4a, identifying non-monotonic changes in the semiquinone state versus the fully reduced state. No equations, fitted parameters, predictions derived from subsets of the same data, or self-citations are used to derive the central claims. The interpretation that the semiquinone signature is functionally competent is presented as a biophysical observation rather than a self-referential derivation that reduces to its own inputs by construction. The work is self-contained as direct experimental mapping with no load-bearing circular steps.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption HDX-MS deuterium uptake rates reflect local protein flexibility, hydrogen bonding, and solvent accessibility
Reference graph
Works this paper leans on
-
[1]
Johnsen, S. and K.J. Lohmann, The physics and neurobiology of magnetoreception. Nature Reviews Neuroscience, 2005. 6(9): p. 703-712
2005
-
[2]
Nießner, and W
Wiltschko, R., C. Nießner, and W. Wiltschko, The Magnetic Compass of Birds: The Role of Cryptochrome. Frontiers in Physiology, 2021. 12
2021
-
[3]
Wiltschko, R. and W. Wiltschko, Magnetoreception in birds. Journal of The Royal Society Interface, 2019. 16(158): p. 20190295
2019
-
[4]
Hore, P .J. and H. Mouritsen, The radical-pair mechanism of magnetoreception. Annual Review of Biophysics, 2016. 45: p. 299-344
2016
-
[5]
Proceedings of the National Academy of Sciences, 2012
Maeda, K., et al., Magnetically sensitive light-induced reactions in cryptochrome are consistent with its proposed role as a magnetoreceptor. Proceedings of the National Academy of Sciences, 2012. 109(13): p. 4774-4779
2012
-
[6]
Nature Chemistry, 2016
Kattnig, D.R., et al., Chemical amplification of magnetic field effects relevant to avian magnetoreception. Nature Chemistry, 2016. 8(4): p. 384-391
2016
-
[7]
Scientific reports, 2017
Sheppard, D.M., et al., Millitesla magnetic field effects on the photocycle of an animal cryptochrome. Scientific reports, 2017. 7(1): p. 42228
2017
-
[8]
Nature, 2021
Xu, J., et al., Magnetic sensitivity of cryptochrome 4 from a migratory songbird. Nature, 2021. 594(7864): p. 535-540
2021
-
[9]
Wiltschko, W. and F . Merkel, Orientierung zugunruhiger Rotkehlchen im statischen Magnetfeld. Verhandlungen der Deutschen Zoologischen Gesellschaft, 1966. 59: p. 362-367
1966
-
[10]
Journal of the Royal Society Interface, 2014
Lee, A.A., et al., Alternative radical pairs for cryptochrome-based magnetoreception. Journal of the Royal Society Interface, 2014. 11(95): p. 20131063. 19
2014
-
[11]
Journal of The Royal Society Interface, 2016
Wiltschko, R., et al., Light-dependent magnetoreception in birds: The crucial step occurs in the dark. Journal of The Royal Society Interface, 2016. 13(118): p. 20151010
2016
-
[12]
Proceedings of the National Academy of Sciences, 2019
Zoltowski, B.D., et al., Chemical and structural analysis of a photoactive vertebrate cryptochrome from pigeon. Proceedings of the National Academy of Sciences, 2019. 116(39): p. 19449
2019
-
[13]
and P .J
Player, T.C. and P .J. Hore, Viability of superoxide-containing radical pairs as magnetoreceptors. The Journal of Chemical Physics, 2019. 151(22)
2019
-
[14]
Science Advances, 2020
Hochstoeger, T., et al., The biophysical, molecular, and anatomical landscape of pigeon CRY4: A candidate light-based quantal magnetosensor. Science Advances, 2020. 6(33): p. eabb9110
2020
-
[15]
Scientific Reports, 2020
Wu, H., et al., Protein-protein interaction of the putative magnetoreceptor cryptochrome 4 expressed in the avian retina. Scientific Reports, 2020. 10(1): p. 7364
2020
-
[16]
Frontiers in Molecular Neuroscience, 2023
Yee, C., et al., Kinetics of cone specific G-protein signaling in avian photoreceptor cells. Frontiers in Molecular Neuroscience, 2023. 16
2023
-
[17]
Journal of Biological Chemistry, 2012
Watari, R., et al., Light-dependent structural change of chicken retinal cryptochrome4. Journal of Biological Chemistry, 2012. 287(51): p. 42634-42641
2012
-
[18]
Sci Adv, 2025
Maestre-Reyna, M., et al., Capturing structural intermediates in an animal-like cryptochrome photoreceptor by time-resolved crystallography. Sci Adv, 2025. 11(20): p. eadu7247
2025
-
[19]
Solov'yov, and P .J
Kattnig, D.R., I.A. Solov'yov, and P .J. Hore, Electron spin relaxation in cryptochrome-based magnetoreception. Physical Chemistry Chemical Physics,
-
[20]
12443-12456
18(18): p. 12443-12456
-
[21]
J Am Chem Soc, 2022
Gruning, G., et al., Effects of Dynamical Degrees of Freedom on Magnetic Compass Sensitivity: A Comparison of Plant and Avian Cryptochromes. J Am Chem Soc, 2022. 144(50): p. 22902-22914
2022
-
[22]
PLOS ONE, 2007
Liedvogel, M., et al., Chemical Magnetoreception: Bird Cryptochrome 1a Is Excited by Blue Light and Forms Long-Lived Radical-Pairs. PLOS ONE, 2007. 2(10): p. e1106
2007
-
[23]
Scientific Reports, 2017
Kutta, R.J., et al., Vertebrate Cryptochromes are Vestigial Flavoproteins. Scientific Reports, 2017. 7(1): p. 44906
2017
-
[24]
Journal of Neurochemistry, 2006
Kubo, Y ., et al., Molecular cloning, mRNA expression, and immunocytochemical localization of a putative blue-light photoreceptor CRY4 in the chicken pineal gland. Journal of Neurochemistry, 2006. 97(4): p. 1155-1165
2006
-
[25]
Chronobiology international, 2006
Helfer, G., et al., Molecular analysis of clock gene expression in the avian brain. Chronobiology international, 2006. 23(1-2): p. 113-127
2006
-
[26]
Rani, and V
Singh, D., S. Rani, and V . Kumar, Daily Expression of Six Clock Genes in Central and Peripheral Tissues of a Night-Migratory SongBird: Evidence for Tissue- Specific Circadian Timing. Chronobiology International, 2013. 30(10): p. 1208- 1217
2013
-
[27]
Wang, Q. and C. Lin, Mechanisms of Cryptochrome-Mediated Photoresponses in Plants. Annu Rev Plant Biol., 2020. 71: p. 103-129
2020
-
[28]
Nature Structural & Molecular Biology, 2020
Shao, K., et al., The oligomeric structures of plant cryptochromes. Nature Structural & Molecular Biology, 2020. 27(5): p. 480-488. 20
2020
-
[29]
Plant Cell, 2005
Sang, Y ., et al., N-terminal domain-mediated homodimerization is required for photoreceptor activity of Arabidopsis CRYPTOCHROME 1. Plant Cell, 2005. 17(5): p. 1569-84
2005
-
[30]
Proceedings of the National Academy of Sciences of the United States of America, 2013
Vaidya, A.T., et al., Flavin reduction activates Drosophila cryptochrome. Proceedings of the National Academy of Sciences of the United States of America, 2013. 110(51): p. 20455-20460
2013
-
[31]
Commun Biol,
Chandrasekaran, S., et al., Tuning flavin environment to detect and control light- induced conformational switching in Drosophila cryptochrome. Commun Biol,
-
[32]
Structure, 2025
Chandler, S.A., et al., Light-induced conformational switching and magnetic sensitivity of Drosophila cryptochrome. Structure, 2025. 33(11): p. 1930-1943.e4
2025
-
[33]
Nat Commun, 2017
Lim, X.X., et al., Conformational changes in intact dengue virus reveal serotype- specific expansion. Nat Commun, 2017. 8: p. 14339
2017
-
[34]
Chem Rev,
James, E.I., et al., Advances in Hydrogen/Deuterium Exchange Mass Spectrometry and the Pursuit of Challenging Biological Systems. Chem Rev,
-
[35]
7562-7623
122(8): p. 7562-7623
-
[36]
Analytical Chemistry, 2023
Kish, M., et al., Online Fully Automated System for Hydrogen/Deuterium- Exchange Mass Spectrometry with Millisecond Time Resolution. Analytical Chemistry, 2023. 95(11): p. 5000–5008
2023
-
[37]
Anal Chem, 2019
Svejdal, R.R., et al., Thiol-ene Microfluidic Chip for Performing Hydrogen/Deuterium Exchange of Proteins at Subsecond Time Scales. Anal Chem, 2019. 91(2): p. 1309-1317
2019
-
[38]
Keppel, T.R. and D.D. Weis, Mapping Residual Structure in Intrinsically Disordered Proteins at Residue Resolution Using Millisecond Hydrogen/Deuterium Exchange and Residue Averaging. Journal of the American Society for Mass Spectrometry, 2015. 26(4): p. 547-554
2015
-
[39]
Analytical Chemistry, 2022
Seetaloo, N., et al., Millisecond Hydrogen/Deuterium-Exchange Mass Spectrometry Approach to Correlate Local Structure and Aggregation in α- Synuclein. Analytical Chemistry, 2022. 94(48): p. 16711-16719
2022
-
[40]
Anal Chem, 2020
Zheng, J., et al., Comparative Analysis of Cleavage Specificities of Immobilized Porcine Pepsin and Nepenthesin II under Hydrogen/Deuterium Exchange Conditions. Anal Chem, 2020. 92(16): p. 11018-11028
2020
-
[41]
J Am Chem Soc, 2008
Zehl, M., et al., Electron transfer dissociation facilitates the measurement of deuterium incorporation into selectively labeled peptides with single residue resolution. J Am Chem Soc, 2008. 130(51): p. 17453-9
2008
-
[42]
J Am Chem Soc, 2008
Rand, K.D., et al., Electron capture dissociation proceeds with a low degree of intramolecular migration of peptide amide hydrogens. J Am Chem Soc, 2008. 130(4): p. 1341-9
2008
-
[43]
Anal Chem, 2018
Brodie, N.I., et al., Top-Down Hydrogen-Deuterium Exchange Analysis of Protein Structures Using Ultraviolet Photodissociation. Anal Chem, 2018. 90(5): p. 3079- 3082
2018
-
[44]
Journal of Molecular Biology, 2024
Saft, M., et al., One More for Light-triggered Conformational Changes in Cryptochromes: CryP from Phaeodactylum tricornutum. Journal of Molecular Biology, 2024. 436(5): p. 168408
2024
-
[45]
Scientific Reports,
Franz-Badur, S., et al., Structural changes within the bifunctional cryptochrome/photolyase CraCRY upon blue light excitation. Scientific Reports,
-
[46]
The Journal of Physical Chemistry B, 2023
Hanić, M., et al., Dimerization of European Robin Cryptochrome 4a. The Journal of Physical Chemistry B, 2023. 127(28): p. 6251-6264
2023
-
[47]
J Am Chem Soc, 2023
Timmer, D., et al., Tracking the Electron Transfer Cascade in European Robin Cryptochrome 4 Mutants. J Am Chem Soc, 2023. 145(21): p. 11566-11578
2023
-
[48]
Hageman, T.S. and D.D. Weis, Reliable Identification of Significant Differences in Differential Hydrogen Exchange-Mass Spectrometry Measurements Using a Hybrid Significance Testing Approach. Analytical Chemistry, 2019. 91(13): p. 8008-8016
2019
-
[49]
Nat Commun, 2024
Denton, M.C.J., et al., Magnetosensitivity of tightly bound radical pairs in cryptochrome is enabled by the quantum Zeno effect. Nat Commun, 2024. 15(1): p. 10823
2024
-
[50]
Computational and Structural Biotechnology Journal, 2024
Ramsay, J.L., et al., Cryptochrome magnetoreception: time course of photoactivation from non-equilibrium coarse-grained molecular dynamics. Computational and Structural Biotechnology Journal, 2024. 26: p. 58-69
2024
-
[51]
Kattnig, and I.A
Schuhmann, F ., D.R. Kattnig, and I.A. Solov'yov, Exploring post-activation conformational changes in pigeon cryptochrome 4. Journal of Physical Chemistry B, 2021. 125(34): p. 9652-9659
2021
-
[52]
The Journal of Physical Chemistry B, 2024
Schuhmann, F ., et al., Structural Rearrangements of Pigeon Cryptochrome 4 Undergoing a Complete Redox Cycle. The Journal of Physical Chemistry B, 2024. 128(16): p. 3844-3855
2024
-
[53]
Nielsen, and I.A
Kattnig, D.R., C. Nielsen, and I.A. Solov'yov, Molecular dynamics simulations disclose early stages of the photo-activation of cryptochrome 4. New Journal of Physics, 2018. 20: p. 083018
2018
-
[54]
Nature, 2023
Bradlaugh, A.A., et al., Essential elements of radical pair magnetosensitivity in Drosophila. Nature, 2023. 615(7950): p. 111-116
2023
-
[55]
Kish, M. and J.J. Phillips, HydroBot: Software for Interactive Hydrogen/Deuterium- Exchange Mass Spectrometry Multistate Analysis. J Am Soc Mass Spectrom,
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.