Recognition: unknown
Mitochondrial mechanics nucleates axonal jamming and swelling
Pith reviewed 2026-05-08 12:52 UTC · model grok-4.3
The pith
Mitochondrial shape and rigidity control whether they jam in axons and cause swelling.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Mitochondrial traffic jams emerge from a force balance between active propulsion and steric interactions, and their severity is governed by organelle shape and mechanical properties. Elongated, mechanically rigid mitochondria remain aligned and are transported rapidly, whereas flexible, low-aspect-ratio mitochondria are prone to jamming and accumulation. Incorporating fission and fusion dynamics reveals that fission amplifies transport disruption by generating collision-prone populations, while fusion restores transport by producing anisotropic structures that navigate crowded environments more efficiently. Sustained jamming generates mechanical stress on the axonal membrane, leading to变形和sw
What carries the argument
Agent-based model coupling mitochondrial motility, morphology and lifecycle dynamics to a deformable axonal boundary, through force balance between active propulsion and steric interactions.
If this is right
- Elongated rigid mitochondria align and move rapidly through axons without jamming.
- Flexible low-aspect-ratio mitochondria jam and accumulate at higher rates.
- Increased fission generates more collision-prone mitochondria and worsens jams.
- Increased fusion produces elongated shapes that reduce jamming and restore flow.
- Persistent jams produce membrane stress that deforms and swells the axon.
Where Pith is reading between the lines
- Dysregulated fission-fusion balance could accelerate axonal structural damage in disease through this jamming route.
- The model supplies concrete, testable links between organelle population statistics and measurable axon diameter changes.
- Similar shape-dependent jamming may occur among other crowded organelles inside narrow cellular processes.
Load-bearing premise
The model assumes that force balance between active propulsion and steric interactions, together with mitochondrial shape and mechanical properties, are the dominant factors governing jamming and axonal deformation.
What would settle it
Live-cell imaging that finds no membrane deformation or swelling at sites of mitochondrial accumulation, regardless of shape or fission-fusion state, would falsify the proposed stress mechanism.
Figures
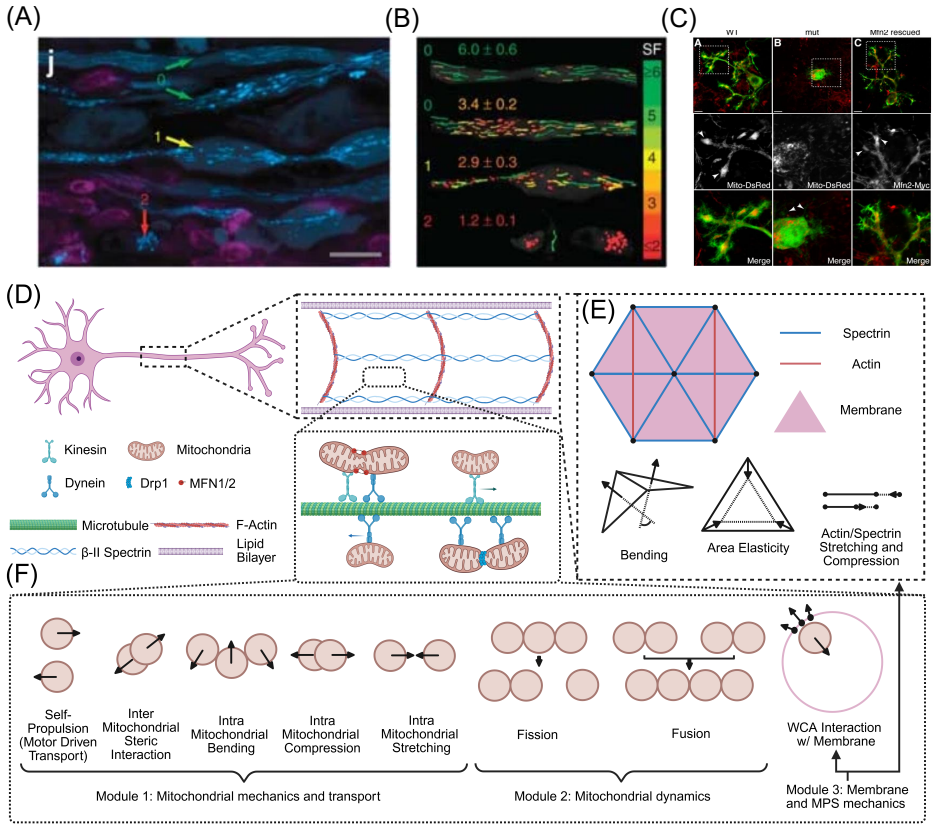

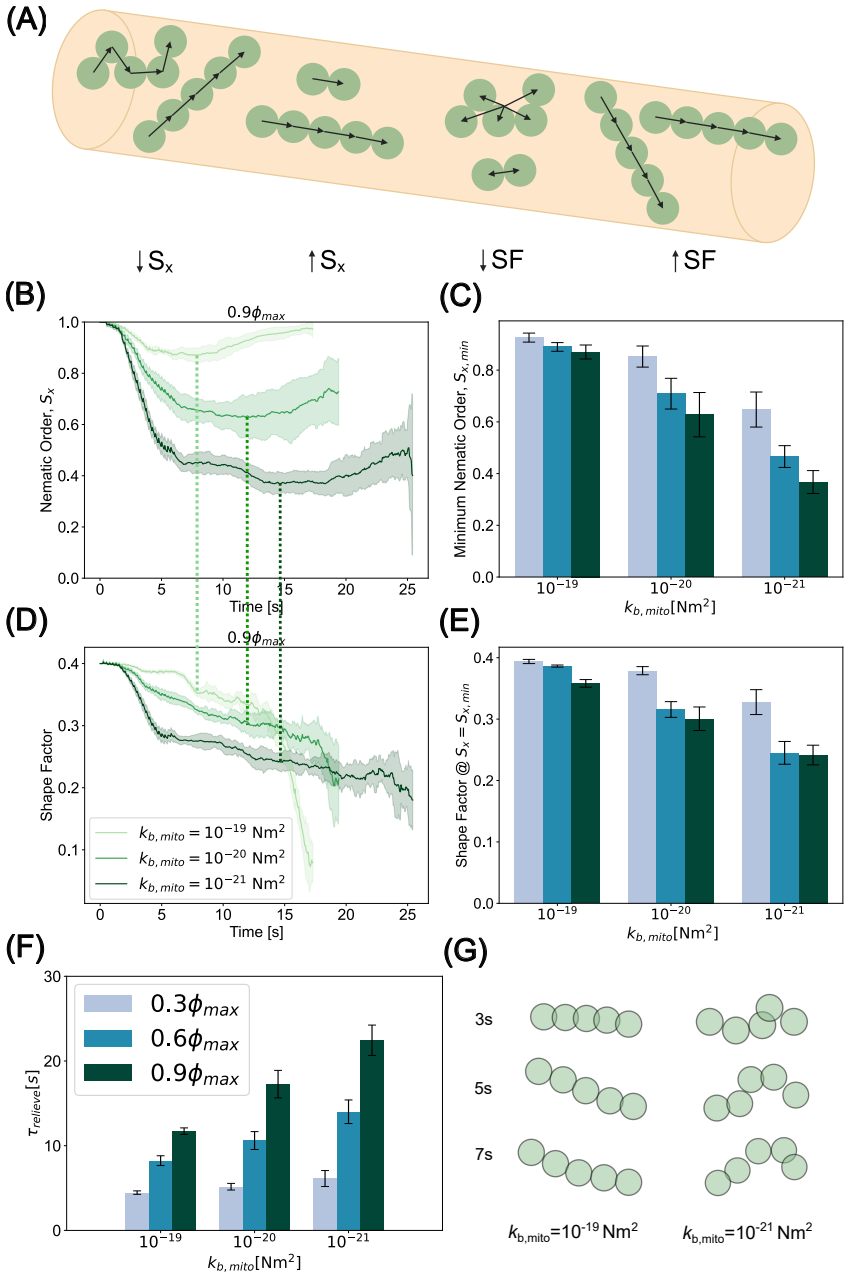





read the original abstract
Neuronal function requires precise spatial organization of mitochondria to meet localized energetic demand. However, the physical constraints governing mitochondrial transport in axons remain poorly defined. Bidirectional motor-driven trafficking inherently introduces the potential for collisions, but the implications of these interactions for transport failure and structural damage are not understood. Here, we develop an agent-based model that couples mitochondrial motility, morphology, and lifecycle dynamics to a deformable axonal boundary. We show that mitochondrial traffic jams emerge from a force balance between active propulsion and steric interactions, and that their severity is governed by organelle shape and mechanical properties. Elongated, mechanically rigid mitochondria remain aligned and are transported rapidly, whereas flexible, low-aspect-ratio mitochondria are prone to jamming and accumulation. Incorporating fission and fusion dynamics reveals that fission amplifies transport disruption by generating collision-prone populations, while fusion restores transport by producing anisotropic structures that navigate crowded environments more efficiently. Importantly, we find that sustained jamming generates mechanical stress on the axonal membrane, leading to deformation and swelling. Together, these results establish a physical framework linking mitochondrial dynamics to axonal integrity and provide testable predictions for how dysregulated fission-fusion balance can drive transport failure and structural pathology in neurons.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper introduces an agent-based model that couples mitochondrial motility, morphology, and fission-fusion dynamics to a deformable axonal boundary. It claims that mitochondrial jamming emerges from the force balance between active motor-driven propulsion and steric interactions, modulated by organelle shape and mechanical properties. Elongated, rigid mitochondria are transported efficiently, while flexible, low-aspect-ratio ones are prone to jamming. Fission increases disruption, fusion restores transport, and sustained jamming produces mechanical stress leading to axonal swelling and deformation.
Significance. If the model's assumptions hold, this establishes a physical link between mitochondrial dynamics and axonal pathology, with implications for understanding neurodegenerative conditions involving transport defects. The model generates specific, testable predictions regarding the effects of fission-fusion imbalance. Strengths include the integration of morphology, mechanics, and boundary deformation in a single framework. However, the results are simulation-based and would benefit from direct experimental validation to confirm the dominance of the modeled interactions.
major comments (1)
- [Model description and results on membrane deformation] Model description and results on membrane deformation: The claim that sustained jamming generates mechanical stress on the axonal membrane, leading to deformation and swelling, depends on the untested assumption that steric and mitochondrial forces dominate over other cytoskeletal elements such as microtubules, actin cortex, and neurofilaments. No quantitative bounds, sensitivity analysis, or comparison showing that inclusion of these elements would not alter the deformation threshold are provided. This assumption is load-bearing for the headline result.
minor comments (1)
- [Abstract] The abstract mentions 'testable predictions' but does not explicitly state them; including one or two key predictions would improve clarity for readers.
Simulated Author's Rebuttal
We thank the referee for their constructive review and for acknowledging the potential significance of our agent-based model in connecting mitochondrial dynamics to axonal pathology. We address the single major comment below and will revise the manuscript accordingly to strengthen the presentation of our assumptions and results.
read point-by-point responses
-
Referee: The claim that sustained jamming generates mechanical stress on the axonal membrane, leading to deformation and swelling, depends on the untested assumption that steric and mitochondrial forces dominate over other cytoskeletal elements such as microtubules, actin cortex, and neurofilaments. No quantitative bounds, sensitivity analysis, or comparison showing that inclusion of these elements would not alter the deformation threshold are provided. This assumption is load-bearing for the headline result.
Authors: We appreciate the referee identifying this foundational assumption in our model of axonal deformation. The framework is constructed to isolate the mechanical consequences of mitochondrial jamming and steric interactions with a deformable boundary, without explicit representation of microtubules, actin cortex, or neurofilaments. We agree that these cytoskeletal elements contribute to axonal mechanics in vivo and could modulate deformation thresholds. Incorporating them fully would require a substantially more complex multi-component biomechanical model beyond the current scope. In the revised manuscript we will add an explicit limitations subsection that states the assumption clearly, supplies order-of-magnitude estimates comparing typical mitochondrial propulsion forces to reported cytoskeletal stiffnesses, and includes new sensitivity simulations in which boundary stiffness is varied over a physiologically plausible range. These additions will provide quantitative bounds on the deformation threshold and clarify the conditions under which mitochondrial forces remain dominant. revision: yes
Circularity Check
Model simulation produces emergent jamming and swelling from explicit force-balance rules
full rationale
The paper constructs an agent-based model with explicit rules for mitochondrial motility, morphology, fission/fusion, and force balance between active propulsion and steric interactions acting on a deformable axonal boundary. Reported outcomes (jamming severity governed by shape/mechanics, sustained jamming producing membrane stress and swelling) are simulation results under those rules rather than quantities fitted to or defined by the target phenomenon. No self-definitional equations, fitted-input predictions, or load-bearing self-citations that reduce the central claim to its own inputs appear in the abstract or model description. The derivation is therefore self-contained as a forward simulation.
Axiom & Free-Parameter Ledger
free parameters (1)
- mitochondrial aspect ratio and rigidity parameters
axioms (1)
- domain assumption Force balance between active propulsion and steric interactions governs mitochondrial movement and jamming.
Reference graph
Works this paper leans on
-
[1]
D., Sokoloff, L
Clarke, D. D., Sokoloff, L. & Siegel, G. Basic neurochemistry: molecular, cellular and medical aspects. Lippincott-Raven, Philadelphia(1999)
1999
-
[2]
A., Carruthers, A
Simpson, I. A., Carruthers, A. & Vannucci, S. J. Supply and demand in cerebral energy metabolism: the role of nutrient transporters.Journal of Cerebral Blood Flow & Metabolism27,1766–1791 (2007)
2007
-
[3]
& Hilliard, M
Donato, A., Kagias, K., Zhang, Y . & Hilliard, M. A. Neuronal sub-compartmentalization: a strategy to optimize neuronal function.Biological Reviews94,1023–1037 (2019)
2019
-
[4]
J., Jolivet, R
Harris, J. J., Jolivet, R. & Attwell, D. Synaptic energy use and supply.Neuron75,762–777 (2012)
2012
-
[5]
& Sheng, Z.-H
Li, S. & Sheng, Z.-H. Energy matters: presynaptic metabolism and the maintenance of synaptic trans- mission.Nature Reviews Neuroscience23,4–22 (2022)
2022
-
[6]
H.et al.Action potential generation requires a high sodium channel density in the axon initial segment.Nature neuroscience11,178–186 (2008)
Kole, M. H.et al.Action potential generation requires a high sodium channel density in the axon initial segment.Nature neuroscience11,178–186 (2008)
2008
-
[7]
Bender, K. J. & Trussell, L. O. Axon initial segment Ca2+ channels influence action potential genera- tion and timing.Neuron61,259–271 (2009)
2009
-
[8]
E., Best, T
Sheffield, M. E., Best, T. K., Mensh, B. D., Kath, W. L. & Spruston, N. Slow integration leads to persistent action potential firing in distal axons of coupled interneurons.Nature neuroscience14,200– 207 (2011)
2011
-
[9]
& Ikegaya, Y
Sasaki, T., Matsuki, N. & Ikegaya, Y . Action-potential modulation during axonal conduction.Science 331,599–601 (2011)
2011
-
[10]
J.et al.Tracking axonal action potential propagation on a high-density microelectrode array across hundreds of sites.Nature communications4,2181 (2013)
Bakkum, D. J.et al.Tracking axonal action potential propagation on a high-density microelectrode array across hundreds of sites.Nature communications4,2181 (2013)
2013
-
[11]
The metabolism of the central nervous system in vivo.Handbook of physiology, section I, neurophysiology3,1843–1864 (1960)
Sokoloff, L. The metabolism of the central nervous system in vivo.Handbook of physiology, section I, neurophysiology3,1843–1864 (1960)
1960
-
[12]
Schwarz, T. L. Mitochondrial trafficking in neurons.Cold Spring Harbor perspectives in biology5, a011304 (2013)
2013
-
[13]
& Sheng, Z.-H
Lin, M.-Y . & Sheng, Z.-H. Regulation of mitochondrial transport in neurons.Experimental cell re- search334,35–44 (2015)
2015
-
[14]
& Wang, X
Pekkurnaz, G. & Wang, X. Mitochondrial heterogeneity and homeostasis through the lens of a neuron. Nature metabolism4,802–812 (2022)
2022
-
[15]
V ., Ciampi, D
Duarte, F. V ., Ciampi, D. & Duarte, C. B. Mitochondria as central hubs in synaptic modulation.Cellu- lar and Molecular Life Sciences80,173 (2023)
2023
-
[16]
& Okabe, S
Obashi, K. & Okabe, S. Regulation of mitochondrial dynamics and distribution by synapse position and neuronal activity in the axon.European Journal of Neuroscience38,2350–2363 (2013)
2013
-
[17]
& Ryan, T
Rangaraju, V ., Calloway, N. & Ryan, T. A. Activity-driven local ATP synthesis is required for synaptic function.Cell156,825–835 (2014)
2014
-
[18]
& Rangaraju, V
Ghosh, I., Fan, R., Shah, M., Bapat, O. & Rangaraju, V . Synapses drive local mitochondrial ATP synthesis to fuel plasticity.bioRxiv,2025–04 (2025)
2025
-
[19]
& Schuman, E
Rangaraju, V ., Lauterbach, M. & Schuman, E. M. Spatially stable mitochondrial compartments fuel local translation during plasticity.Cell176,73–84 (2019)
2019
-
[20]
Saxton, W. M. & Hollenbeck, P. J. The axonal transport of mitochondria.Journal of cell science125, 2095–2104 (2012). 22
2095
-
[21]
Kruppa, A. J. & Buss, F. Motor proteins at the mitochondria–cytoskeleton interface.Journal of cell science134,jcs226084 (2021)
2021
-
[22]
T., Schroeder, C
Bhabha, G., Johnson, G. T., Schroeder, C. M. & Vale, R. D. How dynein moves along microtubules. Trends in biochemical sciences41,94–105 (2016)
2016
-
[23]
Deguchi, T.et al.Direct observation of motor protein stepping in living cells using MINFLUX.Science 379,1010–1015 (2023)
2023
-
[24]
R., Landers, J
Heidemann, S. R., Landers, J. M. & Hamborg, M. A. Polarity orientation of axonal microtubules.The Journal of cell biology91,661–665 (1981)
1981
-
[25]
Perlson, E., Maday, S., Fu, M.-m., Moughamian, A. J. & Holzbaur, E. L. Retrograde axonal transport: pathways to cell death?Trends in neurosciences33,335–344 (2010)
2010
-
[26]
J., Grierson, A
De V os, K. J., Grierson, A. J., Ackerley, S. & Miller, C. C. Role of axonal transport in neurodegenera- tive diseases.Annu. Rev. Neurosci.31,151–173 (2008)
2008
-
[27]
Marinkovi ´c, P.et al.Axonal transport deficits and degeneration can evolve independently in mouse models of amyotrophic lateral sclerosis.Proceedings of the National Academy of Sciences109,4296– 4301 (2012)
2012
-
[28]
Puls, I.et al.Mutant dynactin in motor neuron disease.Nature genetics33,455–456 (2003)
2003
-
[29]
Hafezparast, M.et al.Mutations in dynein link motor neuron degeneration to defects in retrograde transport.Science300,808–812 (2003)
2003
-
[30]
B.et al.Axonopathy and transport deficits early in the pathogenesis of Alzheimer’s disease
Stokin, G. B.et al.Axonopathy and transport deficits early in the pathogenesis of Alzheimer’s disease. Science307,1282–1288 (2005)
2005
-
[31]
J.et al.Familial amyotrophic lateral sclerosis-linked SOD1 mutants perturb fast axonal transport to reduce axonal mitochondria content.Human molecular genetics16,2720–2728 (2007)
De V os, K. J.et al.Familial amyotrophic lateral sclerosis-linked SOD1 mutants perturb fast axonal transport to reduce axonal mitochondria content.Human molecular genetics16,2720–2728 (2007)
2007
-
[32]
Niki ´c, I.et al.A reversible form of axon damage in experimental autoimmune encephalomyelitis and multiple sclerosis.Nature medicine17,495–499 (2011)
2011
-
[33]
& Stevens, J
Greenberg, M., Leitao, C., Trogadis, J. & Stevens, J. Irregular geometries in normal unmyelinated axons: a 3D serial EM analysis.Journal of neurocytology19,978–988 (1990)
1990
-
[34]
& Baloh, R
Misko, A., Jiang, S., Wegorzewska, I., Milbrandt, J. & Baloh, R. H. Mitofusin 2 is necessary for transport of axonal mitochondria and interacts with the Miro/Milton complex.Journal of Neuroscience 30,4232–4240 (2010)
2010
-
[35]
Berthet, A.et al.Loss of mitochondrial fission depletes axonal mitochondria in midbrain dopamine neurons.Journal of Neuroscience34,14304–14317 (2014)
2014
-
[36]
Chen, H., McCaffery, J. M. & Chan, D. C. Mitochondrial fusion protects against neurodegeneration in the cerebellum.Cell130,548–562 (2007)
2007
-
[37]
Fukumitsu, K.et al.Mitochondrial fission protein Drp1 regulates mitochondrial transport and dendritic arborization in cerebellar Purkinje cells.Molecular and Cellular Neuroscience71,56–65 (2016)
2016
-
[38]
J.et al.Dendrite architecture determines mitochondrial distribution patterns in vivo.Cell reports43(2024)
Donovan, E. J.et al.Dendrite architecture determines mitochondrial distribution patterns in vivo.Cell reports43(2024)
2024
-
[39]
& Avramenko, A
Kuznetsov, A. & Avramenko, A. A macroscopic model of traffic jams in axons.Mathematical Bio- sciences218,142–152 (2009)
2009
-
[40]
B., Brigante, A
Fern ´andez Casafuz, A. B., Brigante, A. M., De Rossi, M. C., Monastra, A. G. & Bruno, L. Deciphering the intracellular forces shaping mitochondrial motion.Scientific Reports14,23914 (2024). 23
2024
-
[41]
Zhang, Y .et al.Modeling of the axon membrane skeleton structure and implications for its mechanical properties.PLoS computational biology13,e1005407 (2017)
2017
-
[42]
M.et al.Membrane mechanics dictate axonal pearls-on-a-string morphology and func- tion.Nature neuroscience28,49–61 (2025)
Griswold, J. M.et al.Membrane mechanics dictate axonal pearls-on-a-string morphology and func- tion.Nature neuroscience28,49–61 (2025)
2025
-
[43]
Ligon, L. A. & Steward, O. Movement of mitochondria in the axons and dendrites of cultured hip- pocampal neurons.Journal of Comparative Neurology427,340–350 (2000)
2000
-
[44]
Hollenbeck, P. J. The distribution, abundance and subcellular localization of kinesin.The Journal of cell biology108,2335–2342 (1989)
1989
-
[45]
M., Spooner, B
Yamada, K. M., Spooner, B. S. & Wessells, N. K. Ultrastructure and function of growth cones and axons of cultured nerve cells.The Journal of cell biology49,614–635 (1971)
1971
-
[46]
& Sch ¨oneberg, J
Arkfeld, E., Wang, Z., Hakozaki, H. & Sch ¨oneberg, J. Whole-cell particle-based digital twin simula- tions from 4D lattice light-sheet microscopy data.bioRxiv,2025–04 (2025)
2025
-
[47]
D., Dash, P
Fischer, T. D., Dash, P. K., Liu, J. & Waxham, M. N. Morphology of mitochondria in spatially restricted axons revealed by cryo-electron tomography.PLoS biology16,e2006169 (2018)
2018
-
[48]
Twist and turn: elastohydrodynamics of microscale active fibres.The European Physical Journal Special Topics233,2775–2808 (2024)
Lisicki, M. Twist and turn: elastohydrodynamics of microscale active fibres.The European Physical Journal Special Topics233,2775–2808 (2024)
2024
-
[49]
& Surrey, T
Rickman, J., N ´ed´elec, F. & Surrey, T. Effects of spatial dimensionality and steric interactions on microtubule-motor self-organization.Physical Biology16,046004 (2019)
2019
-
[50]
N., Bertrand, M., Harden, J
Shendruk, T. N., Bertrand, M., Harden, J. L., Slater, G. W. & de Haan, H. W. Coarse-grained molec- ular dynamics simulations of depletion-induced interactions for soft matter systems.The Journal of chemical physics141(2014)
2014
-
[51]
M., Shen, Q
Van der Bliek, A. M., Shen, Q. & Kawajiri, S. Mechanisms of mitochondrial fission and fusion.Cold Spring Harbor perspectives in biology5,a011072 (2013)
2013
-
[52]
& Zhuang, X
Xu, K., Zhong, G. & Zhuang, X. Actin, spectrin, and associated proteins form a periodic cytoskeletal structure in axons.Science339,452–456 (2013)
2013
-
[53]
& Grinspun, E
Wardetzky, M., Bergou, M., Harmon, D., Zorin, D. & Grinspun, E. Discrete quadratic curvature ener- gies.Computer Aided Geometric Design24,499–518 (2007)
2007
-
[54]
& Iber, D
Runser, S., Vetter, R. & Iber, D. SimuCell3D: three-dimensional simulation of tissue mechanics with cell polarization.Nature Computational Science4,299–309 (2024)
2024
-
[55]
A., Niven, J
Perge, J. A., Niven, J. E., Mugnaini, E., Balasubramanian, V . & Sterling, P. Why do axons differ in caliber?Journal of Neuroscience32,626–638 (2012)
2012
-
[56]
Teixeira, P., Galland, R. & Chevrollier, A.Super-resolution microscopies, technological breakthrough to decipher mitochondrial structure and dynamic.inSeminars in Cell & Developmental Biology159 (2024), 38–51
2024
-
[57]
Kang, J.-S.et al.Docking of axonal mitochondria by syntaphilin controls their mobility and affects short-term facilitation.Cell132,137–148 (2008)
2008
-
[58]
& Monticelli, L
Bochicchio, D. & Monticelli, L. The membrane bending modulus in experiments and simulations: a puzzling picture.Advances in Biomembranes and Lipid Self-Assembly23,117–143 (2016)
2016
-
[59]
Simson, R.et al.Membrane bending modulus and adhesion energy of wild-type and mutant cells of Dictyostelium lacking talin or cortexillins.Biophysical journal74,514–522 (1998)
1998
-
[60]
E., Waters, S
Lang, G. E., Waters, S. L., Vella, D. & Goriely, A. Axonal buckling following stretch injury.Journal of Elasticity129,239–256 (2017). 24
2017
-
[61]
Hossain, M. N. B. & Adnan, A. Mechanical characterization of spectrin at the molecular level.Scien- tific Reports14,16631 (2024)
2024
-
[62]
A formal proof of the Kepler conjectureinForum of mathematics, Pi5(2017), e2
Hales, T.et al. A formal proof of the Kepler conjectureinForum of mathematics, Pi5(2017), e2
2017
-
[63]
PatrickNoerr.RangamaniLabUCSD/Mitochondrial-Transport-in-Axons: Mitochondrial mechanics de- termines axonal jamming and swelling v1version v1.0.0. Apr. 2026.https://doi.org/10. 5281/zenodo.19657215
2026
-
[64]
J., Manczak, M., Mao, P., Shirendeb, U
Calkins, M. J., Manczak, M., Mao, P., Shirendeb, U. & Reddy, P. H. Impaired mitochondrial bio- genesis, defective axonal transport of mitochondria, abnormal mitochondrial dynamics and synaptic degeneration in a mouse model of Alzheimer’s disease.Human molecular genetics20,4515–4529 (2011)
2011
-
[65]
& Suki, B
Bartol ´ak-Suki, E., Imsirovic, J., Nishibori, Y ., Krishnan, R. & Suki, B. Regulation of mitochondrial structure and dynamics by the cytoskeleton and mechanical factors.International journal of molecular sciences18,1812 (2017)
2017
-
[66]
R.et al.Actin chromobody imaging reveals sub-organellar actin dynamics.Nature meth- ods17,917–921 (2020)
Schiavon, C. R.et al.Actin chromobody imaging reveals sub-organellar actin dynamics.Nature meth- ods17,917–921 (2020)
2020
-
[67]
& Kornmann, B
Feng, Q. & Kornmann, B. Mechanical forces on cellular organelles.Journal of cell science131, jcs218479 (2018)
2018
-
[68]
B., De Rossi, M
Fern ´andez Casafuz, A. B., De Rossi, M. C. & Bruno, L. Morphological fluctuations of individual mitochondria in living cells.Journal of Physics: Condensed Matter34,094005 (2022)
2022
-
[69]
Memarian, F. L.et al.Active nematic order and dynamic lane formation of microtubules driven by membrane-bound diffusing motors.Proceedings of the National Academy of Sciences118,e2117107118 (2021)
2021
-
[70]
Mendelsohn, R.et al.Morphological principles of neuronal mitochondria.Journal of Comparative Neurology530,886–902 (2022)
2022
-
[71]
C., Gupta, K., Bartol, T
Garcia, G. C., Gupta, K., Bartol, T. M., Sejnowski, T. J. & Rangamani, P. Mitochondrial morphology governs ATP production rate.Journal of General Physiology155,e202213263 (2023)
2023
-
[72]
Venkatraman, K.et al.Cristae formation is a mechanical buckling event controlled by the inner mito- chondrial membrane lipidome.The EMBO journal42,EMBJ2023114054 (2023)
2023
-
[73]
M., Drocco, J
Westrate, L. M., Drocco, J. A., Martin, K. R., Hlavacek, W. S. & MacKeigan, J. P. Mitochondrial morphological features are associated with fission and fusion events.PloS one9,e95265 (2014)
2014
-
[74]
Mitochondrial fusion and fission in cell life and death.Nature reviews Molecular cell biology11,872–884 (2010)
Westermann, B. Mitochondrial fusion and fission in cell life and death.Nature reviews Molecular cell biology11,872–884 (2010)
2010
-
[75]
bioRxiv,2025–10 (2025)
Khalilimeybodi, A.et al.Systems modeling of mitochondrial dynamics in different exercise regimes. bioRxiv,2025–10 (2025)
2025
-
[76]
Cagalinec, M.et al.Principles of the mitochondrial fusion and fission cycle in neurons.Journal of cell science126,2187–2197 (2013)
2013
-
[77]
Chen, H.et al.Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development.The Journal of cell biology160,189–200 (2003)
2003
-
[78]
R., Pinto-Costa, R., Sousa, S
Costa, A. R., Pinto-Costa, R., Sousa, S. C. & Sousa, M. M. The regulation of axon diameter: from axonal circumferential contractility to activity-dependent axon swelling.Frontiers in Molecular Neu- roscience11,319 (2018). 25
2018
-
[79]
Bernal, R., Pullarkat, P. A. & Melo, F. Mechanical properties of axons.Physical review letters99, 018301 (2007)
2007
-
[80]
Gesztelyi, R.et al.The Hill equation and the origin of quantitative pharmacology.Archive for history of exact sciences66,427–438 (2012)
2012
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.