Recognition: unknown
A CMUT-Based Transcranial Focused Ultrasound Platform for Blood-Brain Barrier Opening in Small Animal Models
Pith reviewed 2026-05-08 08:48 UTC · model grok-4.3
The pith
A CMUT-based ultrasound system opens the blood-brain barrier in rats while monitoring microbubble activity.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
The authors developed a CMUT-based transcranial focused ultrasound system using a geometrically focused half-ring array with five transmitters and one receiving element. Phase-inversion during transmission suppresses CMUT-generated harmonics and enhances broadband detection of microbubble emissions. In rat models, the system achieves spatially localized blood-brain barrier opening confirmed by T1-weighted MRI, with Ktrans permeability maps scaling with applied pressure. Time-resolved acoustic spectra capture microbubble arrival and decay kinetics.
What carries the argument
The geometrically focused half-ring CMUT array with phase-inversion transmission technique, which enables both focused ultrasound delivery for BBB opening and enhanced acoustic monitoring of microbubbles.
Load-bearing premise
That the phase-inversion method and half-ring geometry will preserve therapeutic precision and avoid undetected off-target effects from skull interactions in living subjects.
What would settle it
An in-vivo experiment revealing significant harmonic artifacts or microbubble activation outside the target region that is not reflected in the Ktrans MRI permeability maps.
Figures


read the original abstract
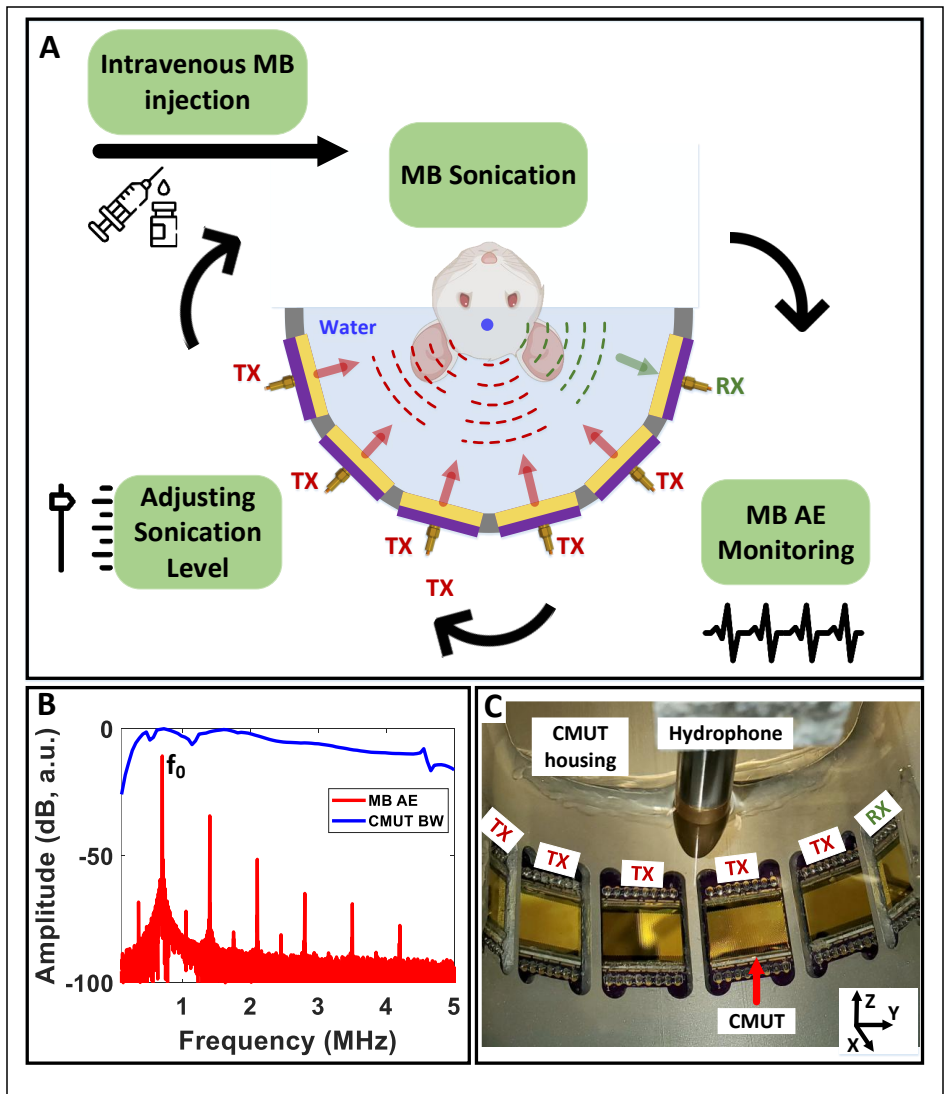
Drug delivery to the brain is limited by the blood-brain barrier (BBB). We developed a capacitive micromachined ultrasonic transducer (CMUT)-based transcranial focused ultrasound system capable of both delivering therapy via BBB opening and monitoring microbubble activity across a broad frequency range. The performance of the geometrically focused half-ring array consisting of five transmitters and one receiving element was first assessed through simulations and in-vitro acoustic measurements with microbubbles. Use of phase-inversion (PI) during transmission effectively suppressed CMUT-generated harmonics and enhanced broadband detection of microbubble emissions. In rats, the same system achieved spatially localized BBB opening, confirmed by T1-weighted magnetic resonance imaging. BBB permeability mapping using dynamic contrast-enhanced magnetic resonance imaging (Ktrans) scaled with pressure. Time-resolved acoustic spectra captured microbubble arrival and decay kinetics, and 7-20dB enhancement in the effective dynamic range is observed with PI processing of acoustic emission signals. Together, these findings establish an integrated CMUT platform for combined therapeutic and sensing applications for BBB opening in small animal models, providing a foundation for future real-time, frequency-agile, closed-loop control of ultrasound-mediated drug delivery to the brain.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript presents the development of a CMUT-based transcranial focused ultrasound system using a half-ring array (5 transmitters, 1 receiver) for blood-brain barrier opening in small animal models. It includes simulations and in-vitro tests demonstrating phase-inversion for harmonic suppression and 7-20 dB dynamic range improvement in microbubble emission detection. In-vivo rat experiments show localized BBB opening via T1-weighted MRI, Ktrans scaling with pressure from DCE-MRI, and time-resolved acoustic spectra for microbubble kinetics. The authors conclude this establishes an integrated platform for therapeutic and sensing applications, foundational for closed-loop control.
Significance. If validated, this represents a meaningful advance in integrated ultrasound devices for preclinical BBB opening studies. The CMUT platform's dual use for therapy and broadband sensing, combined with the phase-inversion technique, could enable more precise, real-time monitoring of microbubble activity during drug delivery. The in-vivo demonstration in rats with MRI confirmation is a strength, providing direct evidence for the system's feasibility in small animal models.
major comments (2)
- [Abstract] Abstract and system design: The central claim that the platform provides a foundation for closed-loop control is load-bearing but not fully supported, as the single receiving element in the half-ring geometry cannot spatially resolve microbubble emissions, and no quantification of skull-induced aberrations or off-target microbubble activation is provided despite reliance on Ktrans maps for localization claims.
- [In-vivo experiments] In-vivo results: The reported scaling of Ktrans with pressure lacks error bars, statistical tests, or a complete parameter table (pressures, frequencies, durations), which undermines the robustness of the therapeutic efficacy and permeability mapping claims central to validating the integrated platform.
minor comments (3)
- [Figures] Figures: MRI images should include clear scale bars, focal region annotations, and error indicators to better demonstrate spatial localization and Ktrans variations.
- [Methods] Methods: Expand details on CMUT array dimensions, exact operating frequencies, simulation parameters, and microbubble concentrations for improved reproducibility.
- [Discussion] Discussion: Add citations to prior transcranial ultrasound and CMUT sensing literature to better contextualize the dynamic range improvements and sensing approach.
Simulated Author's Rebuttal
We thank the referee for their constructive and detailed review. The comments highlight important aspects of clarity and robustness that we will address in the revision. Below we provide point-by-point responses to the major comments.
read point-by-point responses
-
Referee: [Abstract] Abstract and system design: The central claim that the platform provides a foundation for closed-loop control is load-bearing but not fully supported, as the single receiving element in the half-ring geometry cannot spatially resolve microbubble emissions, and no quantification of skull-induced aberrations or off-target microbubble activation is provided despite reliance on Ktrans maps for localization claims.
Authors: We acknowledge that a single receiving element inherently cannot provide spatial resolution of microbubble emissions, which limits the current platform's capability for spatially resolved closed-loop feedback. The phase-inversion processing improves broadband detection sensitivity (7-20 dB) for monitoring microbubble kinetics via time-resolved spectra, but does not add spatial information. Localization of BBB opening is validated by T1-weighted and DCE-MRI Ktrans maps, which serve as the established gold standard in preclinical studies. We did not quantify skull-induced aberrations or off-target activation in this work, as the study focused on demonstrating integrated therapy and sensing feasibility with MRI-confirmed localized effects. The statement regarding a 'foundation for closed-loop control' is forward-looking and refers to the combined therapeutic delivery and real-time acoustic monitoring of kinetics (including frequency content), which could support future control strategies. We will revise the abstract to clarify this scope and avoid implying current spatial resolution for closed-loop applications. revision: partial
-
Referee: [In-vivo experiments] In-vivo results: The reported scaling of Ktrans with pressure lacks error bars, statistical tests, or a complete parameter table (pressures, frequencies, durations), which undermines the robustness of the therapeutic efficacy and permeability mapping claims central to validating the integrated platform.
Authors: We agree that the in-vivo results section would benefit from greater statistical rigor and transparency. The Ktrans scaling with pressure was observed consistently across animals, but the manuscript summarized the trend without full supporting details. In the revised manuscript we will add error bars to the relevant figure, include statistical analysis (e.g., linear regression or ANOVA with post-hoc tests) to support the scaling relationship, and provide a complete parameter table (or supplementary table) listing acoustic pressures, frequencies, pulse durations, repetition rates, total sonication times, and animal numbers per condition. These additions will strengthen the presentation of the therapeutic efficacy and permeability mapping data. revision: yes
Circularity Check
No significant circularity; all claims rest on direct experimental measurements
full rationale
The manuscript reports simulations, in-vitro acoustic tests, and in-vivo rat experiments with MRI (T1-weighted and DCE-MRI Ktrans) plus time-resolved acoustic spectra. No mathematical derivation, fitted-parameter predictions, or self-citation load-bearing steps appear in the provided text. Claims of localized BBB opening and dynamic-range improvement are presented as outcomes of direct measurements rather than reductions to prior inputs. This matches the reader's assessment of zero circularity burden.
Axiom & Free-Parameter Ledger
axioms (2)
- standard math Standard linear and nonlinear acoustic propagation through skull and brain tissue
- domain assumption Microbubble response produces detectable broadband acoustic emissions under focused ultrasound
Reference graph
Works this paper leans on
-
[1]
Proc Natl Acad Sci U S A, 2020
Rezai, A.R., et al., Noninvasive hippocampal blood-brain barrier opening in Alzheimer's disease with focused ultrasound. Proc Natl Acad Sci U S A, 2020. 117(17): p. 9180-9182
2020
-
[2]
Nat Commun, 2021
Gasca-Salas, C., et al., Blood-brain barrier opening with focused ultrasound in Parkinson's disease dementia. Nat Commun, 2021. 12(1): p. 779
2021
-
[3]
Ferraro, and R.K
Arvanitis, C.D., G.B. Ferraro, and R.K. Jain, The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat Rev Cancer, 2020. 20(1): p. 26-41
2020
-
[4]
Hynynen, and N
Meng, Y., K. Hynynen, and N. Lipsman, Applications of focused ultrasound in the brain: from thermoablation to drug delivery. Nat Rev Neurol, 2021. 17(1): p. 7-22
2021
-
[5]
Nat Commun, 2018
Lipsman, N., et al., Blood-brain barrier opening in Alzheimer's disease using MR-guided focused ultrasound. Nat Commun, 2018. 9(1): p. 2336
2018
-
[6]
Adv Drug Deliv Rev, 2022
Schoen, S., Jr., et al., Towards controlled drug delivery in brain tumors with microbubble-enhanced focused ultrasound. Adv Drug Deliv Rev, 2022. 180: p. 114043
2022
-
[7]
Proc Natl Acad Sci U S A, 2021
Anastasiadis, P., et al., Localized blood-brain barrier opening in infiltrating gliomas with MRI-guided acoustic emissions-controlled focused ultrasound. Proc Natl Acad Sci U S A, 2021. 118(37)
2021
-
[8]
Sci Transl Med, 2025
Wu, C.C., et al., Blood-brain barrier opening with neuronavigation-guided focused ultrasound in pediatric patients with diffuse midline glioma. Sci Transl Med, 2025. 17(824): p. eadq6645
2025
-
[9]
Sci Adv, 2021
Chen, K.T., et al., Neuronavigation-guided focused ultrasound for transcranial blood-brain barrier opening and immunostimulation in brain tumors. Sci Adv, 2021. 7(6)
2021
-
[10]
Lancet Oncol,
Sonabend, A.M., et al., Repeated blood-brain barrier opening with an implantable ultrasound device for delivery of albumin-bound paclitaxel in patients with recurrent glioblastoma: a phase 1 trial. Lancet Oncol,
-
[11]
Alzheimers Res Ther, 2022
Epelbaum, S., et al., Pilot study of repeated blood-brain barrier disruption in patients with mild Alzheimer's disease with an implantable ultrasound device. Alzheimers Res Ther, 2022. 14(1): p. 40
2022
-
[12]
IEEE Trans Biomed Eng, 2021
Adams, C., et al., Implementation of a Skull-Conformal Phased Array for Transcranial Focused Ultrasound Therapy. IEEE Trans Biomed Eng, 2021. 68(11): p. 3457-3468
2021
-
[13]
Science Advances, 2022
Lee, H., et al., Spatially targeted brain cancer immunotherapy with closed-loop controlled focused ultrasound and immune checkpoint blockade. Science Advances, 2022. 8(46)
2022
-
[14]
Scientific Reports, 2021
Pouliopoulos, A.N., et al., Safety evaluation of a clinical focused ultrasound system for neuronavigation guided blood-brain barrier opening in non-human primates. Scientific Reports, 2021. 11(1)
2021
-
[15]
Theranostics, 2018
Jones, R.M., et al., Three-dimensional transcranial microbubble imaging for guiding volumetric ultrasound- mediated blood-brain barrier opening. Theranostics, 2018. 8(11): p. 2909-2926
2018
-
[16]
IEEE Trans Biomed Eng, 2023
Bae, S., et al., Real-Time Passive Acoustic Mapping With Enhanced Spatial Resolution in Neuronavigation- Guided Focused Ultrasound for Blood-Brain Barrier Opening. IEEE Trans Biomed Eng, 2023. 70(10): p. 2874-2885
2023
-
[17]
Phys Med Biol, 2024
Fletcher, S.P., et al., The impact of pulse repetition frequency on microbubble activity and drug delivery during focused ultrasound-mediated blood-brain barrier opening. Phys Med Biol, 2024. 69(14)
2024
-
[18]
Device, 2025
Woodworth, G.F., et al., Acoustic emissions dose and spatial control of blood-brain barrier opening with focused ultrasound. Device, 2025. 3(10)
2025
-
[19]
Journal of the Acoustical Society of America, 2020
Dauba, A., et al., Evaluation of capacitive micromachined ultrasonic transducers for passive monitoring of microbubble-assisted ultrasound therapies. Journal of the Acoustical Society of America, 2020. 148(4): p. 2248-2255
2020
-
[20]
Phys Med Biol, 2024
Cornu, C., et al., CMUT for ultrafast passive cavitation detection during ultrasound-induced blood-brain barrier disruption: proof of concept study. Phys Med Biol, 2024. 69(20)
2024
-
[21]
49(11): p
Oralkan, O., et al., Capacitive micromachined ultrasonic transducers: next-generation arrays for acoustic imaging? IEEE Trans Ultrason Ferroelectr Freq Control, 2002. 49(11): p. 1596-610
2002
-
[22]
Ultrasonics, 2018
Pekar, M., et al., Quantitative imaging performance of frequency-tunable capacitive micromachined ultrasonic transducer array designed for intracardiac application: Phantom study. Ultrasonics, 2018. 84: p. 421-429
2018
-
[23]
Oralkan, and B.T
Park, K.K., Ö. Oralkan, and B.T. Khuri-Yakub, A Comparison Between Conventional and Collapse-Mode Capacitive Micromachined Ultrasonic Transducers in 10-MHz 1-D Arrays. Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control, 2013. 60(6): p. 1245-1255
2013
-
[24]
Hasler, and F.L
Gurun, G., P. Hasler, and F.L. Degertekin, Front-End Receiver Electronics for High-Frequency Monolithic CMUT-on-CMOS Imaging Arrays. Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control,
-
[25]
IEEE Trans Ultrason Ferroelectr Freq Control, 2014
Gurun, G., et al., Single-chip CMUT-on-CMOS front-end system for real-time volumetric IVUS and ICE imaging. IEEE Trans Ultrason Ferroelectr Freq Control, 2014. 61(2): p. 239-50
2014
-
[26]
IEEE Trans Ultrason Ferroelectr Freq Control, 2011
Zahorian, J., et al., Monolithic CMUT-on-CMOS integration for intravascular ultrasound applications. IEEE Trans Ultrason Ferroelectr Freq Control, 2011. 58(12): p. 2659-67
2011
-
[27]
IEEE Trans Ultrason Ferroelectr Freq Control, 2024
Pakdaman Zangabad, R., et al., A High Sensitivity CMUT-Based Passive Cavitation Detector for Monitoring Microbubble Dynamics During Focused Ultrasound Interventions. IEEE Trans Ultrason Ferroelectr Freq Control, 2024. 71(9): p. 1087-1096
2024
-
[28]
Lohfink, A. and P.C. Eccardt, Linear and nonlinear equivalent circuit modeling of CMUTs. IEEE Trans Ultrason Ferroelectr Freq Control, 2005. 52(12): p. 2163-72
2005
-
[29]
Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control, 2024
Kilinc, M.S., et al., CMUT as a Transmitter for Microbubble-Assisted Blood-Brain Barrier Opening. Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control, 2024. 71(8): p. 1042-1050
2024
-
[30]
2024 Ieee Ultrasonics, Ferroelectrics, and Frequency Control Joint Symposium, Uffc- Js 2024, 2024
Kilinc, M.S., et al., CMUT-Based Focused Ultrasound Transmit Array for Blood-Brain Barrier Opening in Small Animal Models. 2024 Ieee Ultrasonics, Ferroelectrics, and Frequency Control Joint Symposium, Uffc- Js 2024, 2024
2024
-
[31]
Medical & Biological Engineering & Computing, 1997
Jensen, J.A., Field: A program for simulating ultrasound systems. Medical & Biological Engineering & Computing, 1997. 34(sup. 1): p. 351-353
1997
-
[32]
Ultrasound Med Biol, 2015
Daeichin, V., et al., Subharmonic, non-linear fundamental and ultraharmonic imaging of microbubble contrast at high frequencies. Ultrasound Med Biol, 2015. 41(2): p. 486-97
2015
-
[33]
O'Reilly, M.A. and K. Hynynen, Blood-brain barrier: real-time feedback-controlled focused ultrasound disruption by using an acoustic emissions-based controller. Radiology, 2012. 263(1): p. 96-106
2012
-
[34]
Adv Sci (Weinh), 2026: p
Lee, H., et al., Data-Driven Feedback Identifies Focused Ultrasound Exposure Regimens for Improved Nanotheranostic Targeting of the Brain. Adv Sci (Weinh), 2026: p. e17834
2026
-
[35]
Satir, S. and F.L. Degertekin, Phase and Amplitude Modulation Methods for Nonlinear Ultrasound Imaging With CMUTs. IEEE Trans Ultrason Ferroelectr Freq Control, 2016. 63(8): p. 1086-92
2016
-
[36]
Rowley, and V.M
Carroll, T.J., H.A. Rowley, and V.M. Haughton, Automatic calculation of the arterial input function for cerebral perfusion imaging with MR imaging. Radiology, 2003. 227(2): p. 593-600
2003
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.