Recognition: unknown
OpenMRF: A Modular, Vendor-Neutral Open-Source Framework for Reproducible Magnetic Resonance Fingerprinting using Pulseq
Pith reviewed 2026-05-08 08:54 UTC · model grok-4.3
The pith
OpenMRF supplies a modular Pulseq-based framework for consistent magnetic resonance fingerprinting across MRI vendors and field strengths.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
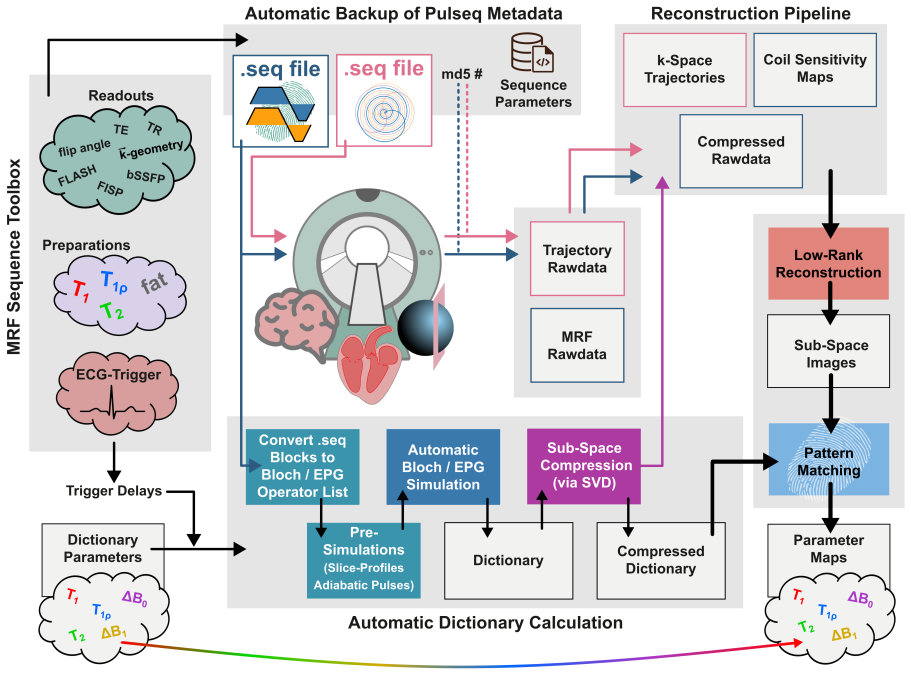
OpenMRF integrates modular Pulseq-based sequence design, Bloch-simulation-based dictionary creation directly from .seq files, and iterative low-rank subspace reconstruction to enable consistent, reproducible, and transferable MRF across vendors, sites, and field strengths, as demonstrated by accurate simulations and multi-site phantom and in vivo results.
What carries the argument
The OpenMRF framework, which uses Pulseq for sequence implementation, performs physics-accurate Bloch simulations from .seq files for dictionary generation, and applies iterative low-rank subspace reconstruction for parameter mapping.
If this is right
- Digital phantom simulations produce T1 deviations of 0.03+/-0.32% and T2 deviations of 0.12+/-1.94%.
- Multi-site phantom measurements remain consistent with references at all tested field strengths and vendors, with mean deviations of -0.1+/-2.9% for T1, -1.5+/-8.7% for T2, and -4.0+/-7.2% for T1rho.
- In vivo liver, myocardium, and brain acquisitions generate high-quality parameter maps on platforms spanning 0.55 T to 3 T.
- The unified platform supports direct method development, comparison, and multi-site validation for quantitative MRI.
Where Pith is reading between the lines
- Groups running multi-center studies could adopt OpenMRF to reduce protocol variability across sites.
- The modular structure may support quick extension to new MRF variants or related quantitative techniques such as diffusion mapping.
- Wider testing on additional scanner models would further test the claim of full vendor neutrality.
Load-bearing premise
The modular Pulseq-based design and simulations will yield accurate and transferable results on real scanners without needing substantial vendor-specific adjustments or calibrations.
What would settle it
Repeating the multi-site phantom protocol on an untested MRI vendor or field strength and finding T1 or T2 deviations exceeding 10% from reference values would undermine the transferability claim.
Figures









read the original abstract
Purpose: Widespread adoption and methodological advancement of Magnetic Resonance Fingerprinting (MRF) are limited by the lack of unified, reproducible implementation frameworks and fragmented open-source tools. To address these barriers, we introduce OpenMRF - a comprehensive Pulseq-based solution - designed to enable consistent, reproducible, and transferable MRF research across vendors, sites, and field strengths. Methods: OpenMRF integrates modular Pulseq-based sequence design, Bloch-simulation-based dictionary creation directly from .seq files, and iterative low-rank subspace reconstruction. The framework was evaluated through digital phantom simulations, a multi-site ISMRM/NIST phantom study on Siemens MRI systems at 0.55 T, 1.5 T, and 3 T as well as GE and United Imaging 3 T platforms, and representative in vivo acquisitions in the liver (0.55 T), myocardium (1.5 T), and brain (3 T). Results: Simulations demonstrated high mapping accuracy in an ISMRM/NIST-like digital phantom, with low-rank reconstruction yielding deviations of 0.03+/-0.32 % (T1) and 0.12+/-1.94 % (T2). The multi-site phantom study yielded relaxation times consistent with reference values at all field strengths, with mean deviations of -0.1+/-2.9 % (T1), -1.5+/-8.7 % (T2), and -4.0+/-7.2 % (T1rho). In vivo acquisitions produced high-quality parameter maps across platforms and field strengths. Conclusion: OpenMRF provides a robust, open-source, end-to-end Pulseq-based solution for MRF that enables reproducible sequence implementation, physics-accurate dictionary simulation, and advanced reconstruction across vendors and field strengths. By providing a unified platform for method development, comparison, and multi-site validation, OpenMRF aims to accelerate reproducible and harmonized quantitative MRI research within the community.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper introduces OpenMRF, a modular, vendor-neutral open-source framework for Magnetic Resonance Fingerprinting (MRF) using Pulseq. It integrates sequence design, Bloch-simulation-based dictionary creation directly from .seq files, and iterative low-rank subspace reconstruction. Validation consists of digital phantom simulations (deviations of 0.03±0.32% T1 and 0.12±1.94% T2), a multi-site ISMRM/NIST phantom study across Siemens (0.55 T, 1.5 T, 3 T), GE, and United Imaging 3 T systems (mean deviations -0.1±2.9% T1, -1.5±8.7% T2, -4.0±7.2% T1ρ), and in vivo acquisitions in liver (0.55 T), myocardium (1.5 T), and brain (3 T), with claims of high mapping accuracy and robustness.
Significance. If the results hold, this work offers a meaningful contribution to quantitative MRI by supplying a unified, reproducible platform that addresses tool fragmentation in MRF. The Pulseq-based modular design and open-source release could facilitate method development, cross-vendor comparisons, and multi-site harmonization, particularly valuable for advancing standardized quantitative imaging protocols.
major comments (1)
- [§2.2] §2.2: The Bloch simulation pipeline from Pulseq .seq files is presented as enabling physics-accurate dictionary generation. Standard Bloch solvers typically omit scanner-specific effects including gradient delays, eddy currents, slice-profile imperfections, and B1 inhomogeneity unless explicitly modeled. Table 3 reports low mean deviations in the multi-site phantom study, but the manuscript provides no direct comparison of simulated versus measured signal evolutions on each platform or vendor, leaving open the possibility that reported consistency arises from post-acquisition scaling rather than intrinsic simulation fidelity.
minor comments (3)
- [Abstract] Abstract: The purpose and conclusion paragraphs are repetitive; the conclusion could emphasize implications for community adoption and harmonization rather than restating framework features.
- [Methods] Methods/Results: Additional details on error propagation, statistical tests, and any data exclusion criteria for the phantom and in vivo evaluations would strengthen assessment of the reported deviation metrics, even with code availability.
- General: Figure captions for parameter maps should specify color scales, units, and any masking applied to improve interpretability across field strengths.
Simulated Author's Rebuttal
We thank the referee for the constructive comment and positive overall assessment of OpenMRF. We address the point raised in §2.2 below and will revise the manuscript accordingly.
read point-by-point responses
-
Referee: [§2.2] §2.2: The Bloch simulation pipeline from Pulseq .seq files is presented as enabling physics-accurate dictionary generation. Standard Bloch solvers typically omit scanner-specific effects including gradient delays, eddy currents, slice-profile imperfections, and B1 inhomogeneity unless explicitly modeled. Table 3 reports low mean deviations in the multi-site phantom study, but the manuscript provides no direct comparison of simulated versus measured signal evolutions on each platform or vendor, leaving open the possibility that reported consistency arises from post-acquisition scaling rather than intrinsic simulation fidelity.
Authors: We thank the referee for this observation. OpenMRF's dictionary generation applies a standard Bloch solver to the Pulseq .seq file without explicit modeling of scanner-specific effects (gradient delays, eddy currents, slice-profile imperfections, or B1 inhomogeneity). The multi-site phantom results show good agreement with reference values, but we agree that the absence of a direct simulated-versus-measured signal comparison leaves the intrinsic fidelity of the simulation less clearly demonstrated. In the revised manuscript we will add a direct comparison: we will extract representative signal evolutions from phantom acquisitions on one or more platforms and overlay them with the corresponding simulated signals generated by the OpenMRF Bloch pipeline. This addition will clarify the contribution of the simulation step to the observed mapping accuracy. revision: yes
Circularity Check
No significant circularity; framework claims rest on independent experimental validations
full rationale
The paper describes OpenMRF as an integrated framework for sequence design, Bloch simulation from .seq files, and low-rank reconstruction, with all accuracy and reproducibility claims supported by direct experimental results: digital phantom simulations reporting specific deviation percentages, multi-site phantom studies on Siemens/GE/United Imaging systems at multiple field strengths showing mean deviations against reference values, and in vivo acquisitions. No load-bearing steps in the presented methods or results reduce by construction to self-definitions, fitted inputs renamed as predictions, or self-citation chains; the derivation chain is self-contained because outcomes are benchmarked externally rather than internally redefined.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Bloch equations accurately model MR signal evolution when driven by Pulseq-defined RF and gradient waveforms
- domain assumption Iterative low-rank subspace reconstruction recovers accurate T1, T2, and T1rho maps from MRF data
Reference graph
Works this paper leans on
-
[1]
Gulani and N
V. Gulani and N. Seiberlich. Quantitative mri: rationale and challenges. In N. Seiberlich, V. Gulani, F. Calamante, A. Campbell-Washburn, M. Doneva, H. H. Hu, and S. Sourbron, editors,Quantitative Magnetic Resonance Imaging, volume 1, pages xxxvii–li. Academic Press, 2020
2020
-
[2]
Hockings, N
P. Hockings, N. Saeed, R. Simms, N. Smith, M. G. Hall, J. C. Waterton, and S. Sour- bron. Mri biomarkers. In N. Seiberlich, V. Gulani, F. Calamante, A. Campbell-Washburn, M. Doneva, H. H. Hu, and S. Sourbron, editors,Quantitative Magnetic Resonance Imaging, volume 1, pages liii–lxxxvi. Academic Press, 2020
2020
-
[3]
K. E. Keenan, M. Ainslie, A. J. Barker, M. A. Boss, K. M. Cecil, C. Charles, T. L. Chen- evert, L. Clarke, J. L. Evelhoch, P. Finn, D. Gembris, J. L. Gunter, D. L. G. Hill, Jr. Jack, C. R., E. F. Jackson, G. Liu, S. E. Russek, S. D. Sharma, M. Steckner, K. F. Stupic, J. D. Trzasko, C. Yuan, and J. Zheng. Quantitative magnetic resonance imaging phantoms: A...
2018
-
[4]
D. R. Messroghli, J. C. Moon, V. M. Ferreira, L. Grosse-Wortmann, T. He, P. Kellman, J. Mascherbauer, R. Nezafat, M. Salerno, E. B. Schelbert, A. J. Taylor, R. Thompson, M. Ugander, R. B. van Heeswijk, and M. G. Friedrich. Clinical recommendations for cardiovascular magnetic resonance mapping of t1, t2, t2* and extracellular volume: A consensus statement ...
2017
-
[5]
Shukla-Dave, N
A. Shukla-Dave, N. A. Obuchowski, T. L. Chenevert, S. Jambawalikar, L. H. Schwartz, D. Malyarenko, W. Huang, S. M. Noworolski, R. J. Young, M. S. Shiroishi, H. Kim, C. Coolens, H. Laue, C. Chung, M. Rosen, M. Boss, and E. F. Jackson. Quantitative imag- ing biomarkers alliance (qiba) recommendations for improved precision of dwi and dce-mri derived biomark...
2019
-
[6]
D. Ma, V. Gulani, N. Seiberlich, K. Liu, J. L. Sunshine, J. L. Duerk, and M. A. Griswold. Magnetic resonance fingerprinting.Nature, 495(7440):187–192, March 2013
2013
-
[7]
S. Gaur, A. Panda, J. E. Fajardo, J. Hamilton, Y. Jiang, and V. Gulani. Magnetic resonance fingerprinting: A review of clinical applications.Invest Radiol, 58(8):561–577, August 2023
2023
-
[8]
K. J. Layton, S. Kroboth, F. Jia, S. Littin, H. Yu, J. Leupold, J. F. Nielsen, T. Stöcker, and M. Zaitsev. Pulseq: A rapid and hardware-independent pulse sequence prototyping framework.Magn Reson Med, 77(4):1544–1552, April 2017
2017
-
[9]
K. S. Ravi, S. Geethanath, and J. T. Vaughan. Pypulseq: A python package for mri pulse sequence design.J Open Source Softw, 4(42):1725, 2019
2019
-
[10]
J. F. Nielsen and D. C. Noll. Toppe: A framework for rapid prototyping of mr pulse sequences.Magn Reson Med, 79(6):3128–3134, June 2018
2018
-
[11]
T. H. M. Roos, E. Versteeg, M. Gosselink, H. Hoogduin, K. M. Nam, N. Boulant, V. Gras, F. Mauconduit, D. W. J. Klomp, J. C. W. Siero, and J. P. Wijnen. ptx-pulseq in hybrid sequences: Accessible and advanced hybrid open-source mri sequences on philips scanners. Magn Reson Med, 94(5):1946–1962, November 2025. 23
1946
-
[12]
M.Gram, Z.Zhu, T.Griesler, S.Kaplan, P.M.Jakob, P.Nordbeck, N.Seiberlich, P.Martin, and Q. Liu. Initial tests of cardiac magnetic resonance fingerprinting at 5t on a whole-body mri scanner using pulseq. InProceedings of the ISMRM 2026 Annual Meeting, Capetown, May 2026. Abstract #02134
2026
-
[13]
A. S. Gaspar, N. A. Silva, A. N. Price, A. M. Ferreira, and R. G. Nunes. Open-source myocardial t1 mapping with simultaneous multi-slice acceleration: Combining an auto- calibrated blipped-bssfp readout with verse-mb pulses.Magn Reson Med, 90(2):539–551, August 2023
2023
-
[14]
Q. Liu, L. Ning, I. A. Shaik, C. Liao, B. Gagoski, B. Bilgic, W. Grissom, J. F. Nielsen, M.Zaitsev, andY.Rathi. Reducedcross-scannervariabilityusingvendor-agnosticsequences for single-shell diffusion mri.Magn Reson Med, 92(1):246–256, July 2024
2024
-
[15]
K. Herz, S. Mueller, O. Perlman, M. Zaitsev, L. Knutsson, P. Z. Sun, J. Zhou, P. van Zijl, K. Heinecke, P. Schuenke, C. T. Farrar, M. Schmidt, A. Dörfler, K. Scheffler, and M. Zaiss. Pulseq-cest: Towards multi-site multi-vendor compatibility and reproducibility of cest experiments using an open-source sequence standard.Magn Reson Med, 86(4):1845– 1858, Oc...
2021
-
[16]
D. Ma, S. Coppo, Y. Chen, D. F. McGivney, Y. Jiang, S. Pahwa, V. Gulani, and M. A. Griswold. Slice profile and b1 corrections in 2d magnetic resonance fingerprinting.Magn Reson Med, 78(5):1781–1789, November 2017
2017
-
[17]
Loktyushin, K
A. Loktyushin, K. Herz, N. Dang, F. Glang, A. Deshmane, S. Weinmüller, A. Doerfler, B. Schölkopf, K. Scheffler, and M. Zaiss. Mrzero - automated discovery of mri sequences using supervised learning.Magn Reson Med, 86(2):709–724, August 2021
2021
-
[18]
Stöcker, K
T. Stöcker, K. Vahedipour, D. Pflugfelder, and N. J. Shah. High-performance computing mri simulations.Magn Reson Med, 64(1):186–193, July 2010
2010
-
[19]
Castillo-Passi, R
C. Castillo-Passi, R. Coronado, G. Varela-Mattatall, C. Alberola-López, R. Botnar, and P. Irarrazaval. Komamri.jl: An open-source framework for general mri simulations with gpu acceleration.Magn Reson Med, 90(1):329–342, July 2023
2023
-
[20]
Schuenke, C
P. Schuenke, C. Redshaw Kranich, M. Lutz, J. Schattenfroh, M. Anders, P. Reisdorf, J. Schulz-Menger, I. Sack, J. Hamilton, N. Seiberlich, and C. Kolbitsch. Open-source cardiac magnetic resonance fingerprinting.MAGMA, 38(4):665–677, August 2025
2025
-
[21]
J. H. Lee, B. A. Hargreaves, B. S. Hu, and D. G. Nishimura. Fast 3d imaging using variable-density spiral trajectories with applications to limb perfusion.Magn Reson Med, 50(6):1276–1285, December 2003
2003
-
[22]
R. K. Robison, Z. Li, D. Wang, M. B. Ooi, and J. G. Pipe. Correction of b0 eddy current effects in spiral mri.Magn Reson Med, 81(4):2501–2513, April 2019
2019
-
[23]
Ong and M
F. Ong and M. Lustig. Sigpy: A python package for high performance iterative reconstruc- tion. ISMRM Workshop on Data Sampling & Image Reconstruction, 2019. Accessed from conference archive
2019
-
[24]
J. I. Hamilton, Y. Jiang, D. Ma, W. C. Lo, V. Gulani, M. Griswold, and N. Seiberlich. Investigating and reducing the effects of confounding factors for robust t1 and t2 mapping with cardiac mr fingerprinting.Magn Reson Imaging, 53:40–51, November 2018
2018
-
[25]
C. R. Wyatt, T. M. Barbara, and A. R. Guimaraes. T1ρmagnetic resonance fingerprinting. NMR Biomed, 33(5):e4284, May 2020. 24
2020
-
[26]
Sharafi, K
A. Sharafi, K. Medina, M. W. V. Zibetti, S. Rao, M. A. Cloos, R. Brown, and R. R. Regatte. Simultaneous t1, t2, and t1ρrelaxation mapping of the lower leg muscle with mr fingerprinting.Magn Reson Med, 86(1):372–381, July 2021
2021
-
[27]
Simultaneous t1, t2, and t1ρcardiac magnetic resonance fingerprinting for contrast agent-free myocardial tissue characterization.Magn Reson Med, 87(4):1992–2002, April 2022
C.Velasco, G.Cruz, B.Lavin, A.Hua, A.Fotaki, R.M.Botnar, andC.Prieto. Simultaneous t1, t2, and t1ρcardiac magnetic resonance fingerprinting for contrast agent-free myocardial tissue characterization.Magn Reson Med, 87(4):1992–2002, April 2022
1992
-
[28]
Kaplan, G
S. Kaplan, G. L. da Cruz, C. Madamanchi, V. L. Murthy, S. Swanson, J. Hamilton, and N. Seiberlich. Simultaneous t1, t2, and t1ρmapping of the myocardium using cardiac mr fingerprinting with a deep image prior reconstruction.Magn Reson Med, 94(4):1500–1513, October 2025
2025
-
[29]
Schuenke, C
P. Schuenke, C. Koehler, A. Korzowski, J. Windschuh, P. Bachert, M. E. Ladd, S. Mundiyanapurath, D. Paech, S. Bickelhaupt, D. Bonekamp, H. P. Schlemmer, A. Rad- bruch, and M. Zaiss. Adiabatically prepared spin-lock approach for t1ρ-based dynamic glucose enhanced mri at ultrahigh fields.Magn Reson Med, 78(1):215–225, July 2017
2017
-
[30]
2nd Witschey, W. R., A. Borthakur, M. A. Elliott, E. Mellon, S. Niyogi, D. J. Wallman, C. Wang, and R. Reddy. Artifacts in t1ρ-weighted imaging: compensation for b(1) and b(0) field imperfections.J Magn Reson, 186(1):75–85, May 2007
2007
-
[31]
M. Gram, M. Seethaler, D. Gensler, J. Oberberger, P. M. Jakob, and P. Nordbeck. Balanced spin-lock preparation for b1-insensitive and b0-insensitive quantification of the rotating frame relaxation time t1ρ.Magn Reson Med, 85(5):2771–2780, May 2021
2021
-
[32]
C. Wan, M. Gram, W. He, Z. Xu, Q. Chen, S. Littin, T. Lange, and M. Zaitsev. Quadruple- refocused spin-locking: A robust method for high-amplitude t1ρimaging.Magn Reson Med, 94(5):2023–2037, November 2025
2023
-
[33]
Mangia, T
S. Mangia, T. Liimatainen, M. Garwood, and S. Michaeli. Rotating frame relaxation during adiabatic pulses vs. conventional spin lock: simulations and experimental results at 4 t. Magn Reson Imaging, 27(8):1074–1087, October 2009
2009
-
[34]
M. Gram, M. Christa, F. T. Gutjahr, P. Albertova, T. Williams, P. M. Jakob, W. R. Bauer, and P. Nordbeck. Quantification of the rotating frame relaxation time t2ρ: Comparison of balanced spin-lock and continuous-wave malcolm-levitt preparations.NMR Biomed, 37(11):e5199, November 2024
2024
-
[35]
D. F. McGivney, E. Pierre, D. Ma, Y. Jiang, H. Saybasili, V. Gulani, and M. A. Griswold. Svd compression for magnetic resonance fingerprinting in the time domain.IEEE Trans Med Imaging, 33(12):2311–2322, December 2014
2014
-
[36]
J. A. Fessler and B. P. Sutton. Nonuniform fast fourier transforms using min-max interpo- lation.IEEE Trans Signal Process, 51(2):560–574, February 2003
2003
-
[37]
J. I. Hamilton, Y. Jiang, D. Ma, Y. Chen, W. C. Lo, M. Griswold, and N. Seiberlich. Simul- taneous multislice cardiac magnetic resonance fingerprinting using low rank reconstruction. NMR Biomed, 32(2):e4041, February 2019
2019
-
[38]
D. Kim, S. F. Cauley, K. S. Nayak, R. M. Leahy, and J. P. Haldar. Region-optimized virtual (rovir) coils: Localization and/or suppression of spatial regions using sensor-domain beamforming.Magn Reson Med, 86(1):197–212, July 2021
2021
-
[39]
Uecker, P
M. Uecker, P. Lai, M. J. Murphy, P. Virtue, M. Elad, J. M. Pauly, S. S. Vasanawala, and M. Lustig. Espirit–an eigenvalue approach to autocalibrating parallel mri: where sense meets grappa.Magn Reson Med, 71(3):990–1001, March 2014. 25
2014
-
[40]
Jiang, D
Y. Jiang, D. Ma, N. Seiberlich, V. Gulani, and M. A. Griswold. Mr fingerprinting using fast imaging with steady state precession (fisp) with spiral readout.Magn Reson Med, 74(6):1621–1631, December 2015
2015
-
[41]
K. F. Stupic, M. Ainslie, M. A. Boss, C. Charles, A. M. Dienstfrey, J. L. Evelhoch, P. Finn, Z. Gimbutas, J. L. Gunter, D. L. G. Hill, C. R. Jack, E. F. Jackson, T. Karaulanov, K. E. Keenan, G. Liu, M. N. Martin, P. V. Prasad, N. S. Rentz, C. Yuan, and S. E. Russek. A standard system phantom for magnetic resonance imaging.Magn Reson Med, 86(3):1194– 1211,...
2021
-
[42]
Simultaneous mapping of water shift and b1 (wasabi)-application to field-inhomogeneity correction of cest mri data.Magn Reson Med, 77(2):571–580, February 2017
P.Schuenke, J.Windschuh, V.Roeloffs, M.E.Ladd, P.Bachert, andM.Zaiss. Simultaneous mapping of water shift and b1 (wasabi)-application to field-inhomogeneity correction of cest mri data.Magn Reson Med, 77(2):571–580, February 2017
2017
-
[43]
Cardiacmrfoptimization at 3t using rosette trajectories and mt modeling in openmrf
S.Kaplan, E.Cummings, T.Griesler, M.Gram, andN.Seiberlich. Cardiacmrfoptimization at 3t using rosette trajectories and mt modeling in openmrf. InProceedings of the ISMRM 2026 Annual Meeting, Capetown, May 2026. Abstract #00881
2026
-
[44]
Griesler, E
T. Griesler, E. Cummings, S. Kaplan, M. Gram, J. Hamilton, M. Davenport, V. Gulani, H. Hussain, N. Seiberlich, and G. Cruz. Kidney t1/t2/t2*/t1ρ/pdff mapping with openmrf: Initial results and adiabatic vs continuous wave spin locking. InProceedings of the ISMRM 2026 Annual Meeting, Capetown, May 2026. Abstract #00661
2026
-
[45]
Hernando, P
D. Hernando, P. Kellman, J. P. Haldar, and Z. P. Liang. Robust water/fat separation in the presence of large field inhomogeneities using a graph cut algorithm.Magn Reson Med, 63(1):79–90, January 2010
2010
-
[46]
Y. Liu, J. Hamilton, B. Eck, M. Griswold, and N. Seiberlich. Myocardial t1 and t2 quan- tification and water-fat separation using cardiac mr fingerprinting with rosette trajectories at 3t and 1.5t.Magn Reson Med, 85(1):103–119, January 2021
2021
-
[47]
Scholten, D
H. Scholten, D. Lohr, T. Wech, and H. Köstler. Fast measurement of the gradient system transfer function at 7 t.Magn Reson Med, 89(4):1644–1659, April 2023
2023
-
[48]
Coletti, A
C. Coletti, A. Fotaki, J. Tourais, Y. Zhao, C. van de Steeg-Henzen, M. Akçakaya, Q. Tao, C. Prieto, and S. Weingärtner. Robust cardiac t1ρmapping at 3t using adiabatic spin-lock preparations.Magn Reson Med, 90(4):1363–1379, October 2023
2023
-
[49]
D. Si, M. G. Crabb, S. J. Littlewood, K. P. Kunze, J. Varghese, K. Binzel, M. Khan, O. P. Simonetti, C. Prieto, and R. M. Botnar. Initial experience of cardiac t1ρmapping at 0.55 t: Continuous wave versus adiabatic spin-lock preparation pulses.Magn Reson Med, 94(4):1644–1653, October 2025
2025
-
[50]
M. Weigel. Extended phase graphs: dephasing, rf pulses, and echoes - pure and simple.J Magn Reson Imaging, 41(2):266–295, February 2015
2015
-
[51]
Kellman and E
P. Kellman and E. R. McVeigh. Image reconstruction in snr units: a general method for snr measurement.Magn Reson Med, 54(6):1439–1447, December 2005
2005
-
[52]
Stebani, I
J. Stebani, I. Angelov, P. Dawood, T. Griesler, P. Albertova, T. Kampf, K. Rak, P. M. Jakob, M. Blaimer, and M. Gram. High-resolution quantitative imaging of the inner ear using 3d magnetic resonance fingerprinting. InProceedings of the ISMRM 2025 Annual Meeting, Honolulu, May 2025. Abstract #0926
2025
-
[53]
Jiang, D
Y. Jiang, D. Ma, K. E. Keenan, K. F. Stupic, V. Gulani, and M. A. Griswold. Repeatability of magnetic resonance fingerprinting t1 and t2 estimates assessed using the ismrm/nist mri system phantom.Magn Reson Med, 78(4):1452–1457, October 2017. 26
2017
-
[54]
W. C. Lo, L. K. Bittencourt, A. Panda, Y. Jiang, J. Tokuda, R. Seethamraju, C. Tempany- Afdhal, V. Obmann, K. Wright, M. Griswold, N. Seiberlich, and V. Gulani. Multicenter repeatability and reproducibility of mr fingerprinting in phantoms and in prostatic tissue. Magn Reson Med, 88(4):1818–1827, October 2022
2022
-
[55]
B. K. Statton, J. Smith, M. E. Finnegan, G. Koerzdoerfer, R. A. Quest, and M. Grech- Sollars. Temperature dependence, accuracy, and repeatability of t1 and t2 relaxation times for the ismrm/nist system phantom measured using mr fingerprinting.Magn Reson Med, 87(3):1446–1460, March 2022
2022
-
[56]
Notni, M
L. Notni, M. Gram, B. Fänger, D. Güllmar, M. Krämer, and J. R. Reichenbach. Quan- titative comparison of t1, t2, t1ρ, and adc for assessing age-related intervertebral disc degeneration.Z Med Phys, pages S0939–3889(25)00132–1, October 2025
2025
-
[57]
M. Gram, D. Gensler, P. Winter, M. Seethaler, P. A. Arias-Loza, J. Oberberger, P. M. Jakob, and P. Nordbeck. Fast myocardial t1ρmapping in mice using k-space weighted image contrast and a bloch simulation-optimized radial sampling pattern.MAGMA, 35(2):325– 340, April 2022
2022
-
[58]
M. Gram, D. Gensler, P. Albertova, F. T. Gutjahr, K. Lau, P. A. Arias-Loza, P. M. Jakob, and P. Nordbeck. Quantification correction for free-breathing myocardial t1ρmapping in mice using a recursively derived description of a t1ρ* relaxation pathway.J Cardiovasc Magn Reson, 24(1):30, May 2022
2022
-
[59]
M. Gram, P. Albertova, V. Schirmer, M. Blaimer, M. Gamer, M. J. Herrmann, P. Nordbeck, and P. M. Jakob. Towards robust in vivo quantification of oscillating biomagnetic fields using rotary excitation based mri.Sci Rep, 12(1):15375, September 2022
2022
-
[60]
Albertova, M
P. Albertova, M. Gram, M. Blaimer, W. R. Bauer, P. M. Jakob, and P. Nordbeck. Rotary excitation of non-sinusoidal pulsed magnetic fields: Towards non-invasive direct detection of cardiac conduction.Magn Reson Med, 92(5):1965–1979, November 2024
1965
-
[61]
Lustig, S
M. Lustig, S. J. Kim, and J. M. Pauly. A fast method for designing time-optimal gradient waveforms for arbitrary k-space trajectories.IEEE Trans Med Imaging, 27(6):866–873, June 2008
2008
-
[62]
Vaziri and M
S. Vaziri and M. Lustig. The fastest arbitrary k-space trajectories. InProceedings of the International Society for Magnetic Resonance in Medicine, volume 20, 2012
2012
-
[63]
X. Wang, Q. Chen, C. Liu, L. Wang, P. Wang, X. Zhou, Y. Pu, M. Gram, T. Griesler, Y. Ni, P. Cao, S. Littin, J. Cai, M. Zaitsev, and T. Li. Astrad: Acquisition-sequence and k-space trajectory co-design for accelerated mr fingerprinting. InProceedings of the ISMRM 2026 Annual Meeting, Capetown, May 2026. Abstract #01511
2026
-
[64]
J. I. Hamilton. A self-supervised deep learning reconstruction for shortening the breathhold andacquisitionwindowincardiacmagneticresonancefingerprinting.Front Cardiovasc Med, 9:928546, June 2022
2022
-
[65]
J. I. Hamilton, W. Truesdell, M. Galizia, N. Burris, P. Agarwal, and N. Seiberlich. A low-rank deep image prior reconstruction for free-breathing ungated spiral functional cmr at 0.55 t and 1.5 t.MAGMA, 36(3):451–464, July 2023. 27 Supplementary Material Supplementary Figure 1: Pulseq sequence diagram of the IR-FISP MRF sequence. The im- plemented method ...
2023
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.