Recognition: unknown
The Genetic and Environmental Architecture of the Human Functional Connectome
Pith reviewed 2026-05-07 16:53 UTC · model grok-4.3
The pith
By incorporating measurement error from repeated fMRI scans into classical twin models, genetic and environmental influences on the functional connectome separate into distinct, hierarchical brain networks.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
We extended ACE/ADE models to include a repeated-scan derived error term by analyzing monozygotic and dizygotic twins from the Young-Adult Human Connectome Project dataset. Genetic and environment variance components were estimated for all functional couplings across resting-state and task conditions, integrated across conditions using a minimum-error criterion, and analyzed using multilayer community detection across resolution scales. Functional couplings segregated into distinct categories characterized by shared environmental, additive, dominant, or epistatic influences, with a substantial fraction not meeting twin-model assumptions. Integrating across conditions revealed hierarchical社区y
What carries the argument
Extended ACE/ADE twin models that add an explicit error term estimated from repeated scans, followed by integration across conditions and multilayer community detection on the resulting variance maps.
If this is right
- Functional couplings can be grouped into categories driven by shared environment, additive genetics, dominance, or epistasis.
- A substantial portion of connections do not satisfy twin-model assumptions even after error correction.
- Genetic and environmental components display hierarchical community structure that persists across different resolution scales.
- Model fit and the resulting network patterns depend on scan duration and the choice of brain parcellation.
Where Pith is reading between the lines
- The same error-modeling step could be used to refine heritability estimates for other neuroimaging measures such as structural connectivity or task activation.
- Multiscale community patterns suggest that future genome-wide association studies might benefit from testing connections at several levels of network resolution rather than single edges.
- If the categories of influence prove stable, they could serve as phenotypes for linking specific genetic variants to distinct aspects of brain organization.
Load-bearing premise
The error term calculated from repeated scans isolates only measurement noise and leaves true biological variance untouched, while the usual twin-model assumptions still hold after this correction.
What would settle it
Re-running the models on scans of increasing length shows that the estimated genetic or environmental components shrink or reorganize in ways the error term cannot explain, or the same community structure fails to appear in a new independent twin sample.
Figures










read the original abstract
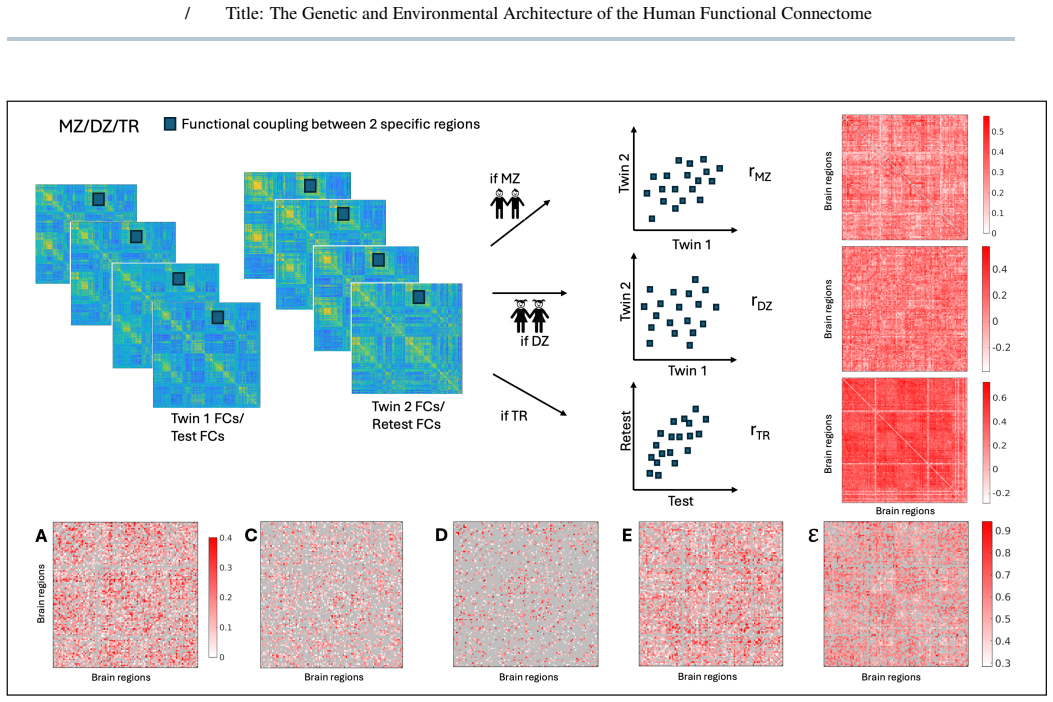
Functional connectivity varies across individuals due to genetic and environmental factors, yet classical twin models typically confound non-shared environment with measurement error and are largely limited to resting-state analyses. We hypothesized that: i) explicitly modeling measurement error from repeated fMRI sessions enables more accurate application of classical twin models (ACE/ADE) to functional connectivity; ii) model applicability depends on scan-length and parcellation granularity; iii) genetic and environmental effects on functional connectomes show differentiated functional modules across conditions. We extended ACE/ADE models to include a repeated-scan derived error term by analyzing monozygotic and dizygotic twins from the Young-Adult Human Connectome Project dataset. Genetic and environment variance components were estimated for all functional couplings across resting-state and task conditions, integrated across conditions using a minimum-error criterion, and analyzed using multilayer community detection across resolution scales. Functional couplings segregated into distinct categories characterized by shared environmental, additive, dominant, or epistatic influences, with a substantial fraction not meeting twin-model assumptions. Integrating across conditions revealed hierarchical community structure in genetic and environmental components observed across community resolution scales. Incorporating measurement error into twin models improves interpretability and applicability at the functional connectome level, revealing that genetic and environmental influences are structured into coherent, multiscale brain networks.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript extends classical ACE/ADE twin models to functional connectivity estimates from the Young-Adult HCP dataset by adding an error term derived from repeated fMRI sessions. It reports that couplings segregate into categories of shared environmental, additive genetic, dominant genetic, or epistatic influence (with a substantial fraction failing model assumptions), and that integrating variance components across resting-state and task conditions via a minimum-error criterion yields hierarchical, multiscale community structure in the genetic and environmental matrices.
Significance. If the repeated-scan error term validly isolates measurement noise, the work would supply more accurate variance-component maps of the functional connectome and demonstrate that genetic and environmental influences organize into coherent multiscale networks rather than diffuse effects. The use of public twin data and multilayer community detection across resolution scales are strengths that could improve interpretability over standard twin analyses that confound error with non-shared environment.
major comments (2)
- [Abstract / Methods] Abstract and Methods (error-term derivation): The central claim that incorporating the repeated-scan error term improves ACE/ADE applicability rests on the assumption that this term cleanly captures scanner/physiological noise without absorbing heritable biological state variance (arousal, attention, fatigue). No quantitative checks—such as correlation of the error variance with independent state measures, comparison against published test-retest ICCs, or sensitivity to scan length—are reported, leaving the adjusted estimates and downstream community detection vulnerable to bias.
- [Results] Results (model applicability and integration): The manuscript states that model applicability depends on scan length and parcellation granularity and that a substantial fraction of couplings fail twin-model assumptions, yet it supplies no tabulated breakdown of failure rates or variance-component stability across these factors. This omission weakens the ability to evaluate the robustness of the minimum-error integration criterion and the claimed hierarchical structure.
minor comments (2)
- [Methods] The phrase 'minimum-error criterion' for integrating across conditions is introduced without a concise definition or equation; adding a short methods paragraph or supplementary equation would clarify how the criterion is computed from the per-condition error variances.
- [Results / Figures] Figure legends and results text should explicitly state whether the multilayer community detection operates on point estimates of the variance components or incorporates their uncertainty (e.g., via bootstrapped or Bayesian community detection).
Simulated Author's Rebuttal
We thank the referee for their detailed and constructive comments on our manuscript. We have carefully considered each point and made revisions to enhance the clarity and robustness of our findings. Our point-by-point responses are provided below.
read point-by-point responses
-
Referee: [Abstract / Methods] Abstract and Methods (error-term derivation): The central claim that incorporating the repeated-scan error term improves ACE/ADE applicability rests on the assumption that this term cleanly captures scanner/physiological noise without absorbing heritable biological state variance (arousal, attention, fatigue). No quantitative checks—such as correlation of the error variance with independent state measures, comparison against published test-retest ICCs, or sensitivity to scan length—are reported, leaving the adjusted estimates and downstream community detection vulnerable to bias.
Authors: We acknowledge the importance of validating that the repeated-scan error term primarily captures measurement noise rather than heritable state variance. In the original manuscript, we derived the error term from the within-subject variance across repeated fMRI sessions, which by construction isolates session-specific fluctuations. To address this concern, we have added a new supplementary analysis in the revised manuscript that examines the sensitivity of error estimates to scan length and compares the magnitude of error variances to published test-retest reliability metrics for functional connectivity from the HCP and other datasets. Unfortunately, the HCP Young-Adult dataset does not include concurrent measures of arousal, attention, or fatigue for the repeated sessions, precluding direct correlation analyses with independent state variables. We have expanded the Discussion to explicitly note this limitation and its potential implications for interpreting the error term. revision: partial
-
Referee: [Results] Results (model applicability and integration): The manuscript states that model applicability depends on scan length and parcellation granularity and that a substantial fraction of couplings fail twin-model assumptions, yet it supplies no tabulated breakdown of failure rates or variance-component stability across these factors. This omission weakens the ability to evaluate the robustness of the minimum-error integration criterion and the claimed hierarchical structure.
Authors: We agree that providing a detailed breakdown of model failure rates would improve the transparency and evaluability of our results. In the revised manuscript, we have included a new supplementary table (Table S1) that reports the percentage of functional couplings failing twin-model assumptions (such as negative variance estimates or inadequate model fit) stratified by scan length categories and parcellation granularity. The table also includes metrics of variance-component stability across these factors. These additions allow readers to better assess the robustness of the minimum-error integration criterion used for combining resting-state and task conditions, as well as the resulting hierarchical community structures. revision: yes
Circularity Check
No circularity: empirical variance-component estimation on public twin data
full rationale
The paper performs standard ACE/ADE twin-model fitting on HCP data with an added repeated-scan error term, followed by multilayer community detection on the resulting matrices. No equation reduces the reported community structure or heritability estimates to quantities defined by the fitting procedure itself; the error term is computed directly from independent repeated sessions rather than being fitted to the target genetic/environmental components. All steps are data-driven estimation against external benchmarks (public twin dataset, established twin-model assumptions), with no self-citation load-bearing the central claims and no renaming of known results as novel derivations. This is a normal empirical analysis whose validity rests on data and model assumptions, not on internal definitional closure.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Repeated fMRI sessions yield an independent estimate of measurement error separable from genetic and environmental variance
- domain assumption Classical twin model assumptions (no gene-environment interaction, random mating, etc.) continue to hold after error adjustment
Reference graph
Works this paper leans on
-
[1]
Go˜ni, J
Abbas, K., Liu, M., Wang, M., Duong-Tran, D., Tipnis, U., Amico, E., . . . Go˜ni, J. (2023). Tangent functional connectomes uncover more unique phenotypic traits.iScience,26(9), 107048. Abdul Wahab, N. S., Yahya, N., Yusoff, A. N., Zakaria, R., Thanabalan, J., Othman, E., . . . Manan, H. A. (2022). Effects of different scan duration on brain effective con...
2023
-
[2]
M., & Rescorla, L
Achenbach, T. M., & Rescorla, L. A. (2003).Manual for the ASEBA adult forms and profiles. University of Vermont, Research Center for Children, Youth, and Families. Amico, E., Arenas, A., & Go ˜ni, J. (2019). Centralized and distributed cognitive task processing in the human connectome. Netw Neurosci,3(2), 455–474. Bianciardi, M., Fukunaga, M., van Geldere...
2003
-
[3]
A., Sch¨olvinck, M
Chang, C., Leopold, D. A., Sch¨olvinck, M. L., Mandelkow, H., Picchioni, D., Liu, X., . . . Duyn, J. H. (2016). Tracking brain arousal fluctuations with fmri.Proceedings of the National Academy of Sciences,113(16), 4518–4523. Chen, G., Moraczewski, D., & Taylor, P. A. (2025). Improving accuracy and precision of heritability estimation in twin studies thro...
2016
-
[4]
Go˜ni, J
Guerrero, D., Dzemidzic, M., Moghaddam, M., Liu, M., Avena-Koenigsberger, A., Harezlak, J., . . . Go˜ni, J. (2025). Resting state functional connectivity patterns associate with alcohol use disorder characteristics: Insights from the triple network model.Neuroimage Clin,45, 103715. Hagenbeek, F. A., Hirzinger, J. S., Breunig, S., Bruins, S., Kuznetsov, D....
2025
-
[5]
O., Snyder, A
Laumann, T. O., Snyder, A. Z., Mitra, A., Gordon, E. M., Gratton, C., Adeyemo, B., . . . others (2017). On the stability of bold fmri correlations.Cerebral cortex,27(10), 4719–4732. Liu, T. T. (2016). Noise contributions to the fmri signal: An overview.NeuroImage,143, 141–151. McIntosh, A. R., & Lobaugh, N. J. (2004). Partial least squares analysis of neu...
2017
-
[6]
J., Richardson, T., Macon, K., Porter, M
Mucha, P. J., Richardson, T., Macon, K., Porter, M. A., & Onnela, J.-P. (2010). Community structure in time-dependent, multiscale, and multiplex networks.Science,328(5980), 876–878. Murphy, K., Birn, R. M., & Bandettini, P. A. (2013). Resting-state fmri confounds and cleanup.Neuroimage,80, 349–359. Neale, M. C., & Cardon, L. R. (1992). Methodology for gen...
2010
-
[7]
D., Mitra, A., Laumann, T
Power, J. D., Mitra, A., Laumann, T. O., Snyder, A. Z., Schlaggar, B. L., & Petersen, S. E. (2014). Methods to detect, characterize, and remove motion artifact in resting state fMRI.Neuroimage,84, 320–341. Schaefer, A., Kong, R., Gordon, E. M., Laumann, T. O., Zuo, X.-N., Holmes, A. J., . . . Yeo, B. T. T. (2018). Local-global parcellation of the human ce...
2014
-
[8]
M., Beckmann, C
Smith, S. M., Beckmann, C. F., Andersson, J., et al. (2013). Resting-state fMRI in the human connectome project.Neuroim- age,80, 144–168. Smith, S. M., Miller, K. L., Salimi-Khorshidi, G., Webster, M., Beckmann, C. F., Nichols, T. E., . . . Woolrich, M. W. (2011). Network modelling methods for fmri.Neuroimage,54(2), 875–891. Tassi, E., Maggioni, E., Mauri...
2013
-
[9]
M., Koenis, M
Teeuw, J., Brouwer, R. M., Koenis, M. M. G., Swagerman, S. C., Boomsma, D. I., & Hulshoff Pol, H. E. (2019). Genetic influences on the development of cerebral cortical thickness during childhood and adolescence in a dutch longitudinal twin sample: The BrainScale study.Cereb Cortex,29(3), 978–993. Ulijaszek, S. J., & Kerr, D. A. (1999). Anthropometric meas...
2019
-
[10]
K., Yang, H., & Biswal, B
Yao, X., Brown, B. K., Yang, H., & Biswal, B. (2023). Structural and functional network analysis of twins using fMRI data. Cereb Cortex,33(22), 11060–11069. Yeo, B. T. T., Krienen, F. M., Sepulcre, J., Sabuncu, M. R., Lashkari, D., Hollinshead, M., . . . Buckner, R. L. (2011). The organization of the human cerebral cortex estimated by intrinsic functional...
2023
-
[11]
Zuo, X.-N., & Xing, X.-X. (2014). Test-retest reliabilities of resting-state fmri measurements in human brain functional connectomics: a systems neuroscience perspective.Neuroscience & biobehavioral reviews,45, 100–118. ACKNOWLEDGMENTS This work was supported by NIH CTSI CTR EPAR2169, NIH R21 AA029614, NIH R01 AA029607, Indiana Alcohol Research Center P60...
2014
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.