Recognition: unknown
Propelling catalytic structures using active phase separation
Pith reviewed 2026-05-07 05:32 UTC · model grok-4.3
The pith
Active phase separation enables indefinite self-propulsion of a spherical colloid via condensate repulsion.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
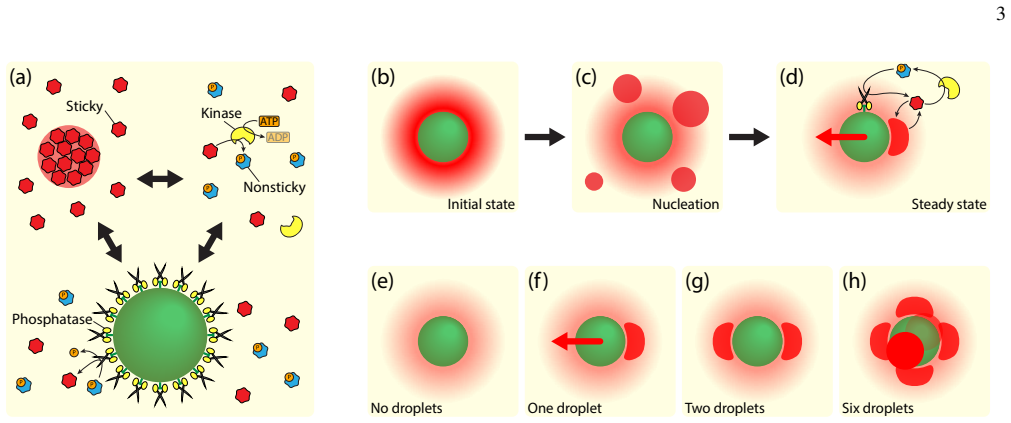
Active droplets can sustain indefinite self-propulsion of a spherical colloid in an otherwise homogeneous, isotropic, and autonomous environment. The minimal mechanism consists of phase-separating proteins, enzymes passivating them, and complementary enzymes anchored to the colloid surface that reactivate the proteins. This passivation-activation cycle gives rise to symmetry breaking via nucleation and stabilization of a condensate near the colloid surface, which exerts a repulsive force on the colloid. Numerical demonstrations show propulsion of micron-sized colloids at speeds up to a hundred microns per second, with strong resistance to Brownian fluctuations and external forces.
What carries the argument
The passivation-activation cycle of phase-separating proteins that nucleates a stabilizing condensate near the colloid to exert sustained repulsion.
If this is right
- The colloid moves indefinitely without motors or external gradients.
- Propulsion speeds can reach up to 100 μm/s for micron-scale particles.
- The motion resists Brownian fluctuations and external forces.
- Biomolecular condensates offer a motor-free route for biological transport.
- This works in fully homogeneous and isotropic conditions.
Where Pith is reading between the lines
- Synthetic colloids with surface enzymes could be designed for autonomous transport in microfluidic systems.
- Cells might exploit similar condensate-based mechanisms for organelle or vesicle motility.
- Extending the model to multiple colloids could reveal collective behaviors or clustering.
- Identifying natural protein pairs that perform the passivation and activation roles would enable direct tests.
Load-bearing premise
The passivation-activation cycle of phase-separating proteins and enzymes will nucleate and stabilize a condensate near the colloid surface that exerts a sustained repulsive force.
What would settle it
An experiment or simulation in which the surface-anchored enzymes are present but no sustained directional motion of the colloid occurs, despite the presence of phase-separating proteins and passivating enzymes, would falsify the proposed mechanism.
Figures





read the original abstract
Living systems routinely consume energy to achieve motility, often using intricate biomolecular machinery. In this work, we show that active droplets can sustain indefinite self-propulsion of a spherical colloid in an otherwise homogeneous, isotropic, and autonomous environment. Our proposed minimal mechanism consists of phase-separating proteins, enzymes passivating them, and complementary enzymes anchored to the colloid surface that reactivate the proteins. This passivation-activation cycle gives rise to a symmetry breaking - nucleation and stabilization of a condensate near the colloid surface, which in turn exerts a repulsive force on the colloid. We numerically demonstrate that this mechanism can propel micron-sized colloids at speeds of up to a hundred microns per second. This propulsion mode is strongly resistant to Brownian fluctuations and external forces, suggesting that propulsion mechanisms based on biomolecular condensates may offer a complementary, motor-free route to biological transport.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript proposes a minimal biochemical mechanism for indefinite self-propulsion of a spherical colloid in a homogeneous, isotropic environment. Phase-separating proteins are passivated by bulk enzymes and reactivated by surface-anchored enzymes on the colloid, producing nucleation and stabilization of an asymmetric condensate that exerts a net repulsive force. Numerical simulations of this passivation-activation cycle are reported to yield propulsion speeds up to 100 μm/s for micron-sized colloids, with strong resistance to Brownian fluctuations and external forces.
Significance. If the numerical results hold under broader conditions, the work identifies a motor-free, condensate-based route to motility that could complement existing mechanisms in biological transport and synthetic biology. The reported combination of high speed and robustness to noise is a notable strength of the proposed cycle. However, the absence of a parameter scan over reaction-diffusion timescales limits assessment of whether the symmetry breaking and sustained propulsion are generic or restricted to a narrow regime.
major comments (2)
- [Numerical model section] Numerical model section (reaction-diffusion equations and boundary conditions): the passivation-activation cycle is implemented with fixed enzymatic rates and a single free parameter set. No sweep is presented over the ratio of surface reactivation rate to bulk passivation rate relative to the colloid diffusion time. Because net propulsion requires persistent asymmetry in the condensate, this omission leaves open whether the reported speeds and robustness persist when timescales are varied, directly affecting the central claim of generic, indefinite self-propulsion.
- [Results on propulsion speeds and robustness] Results on propulsion speeds and robustness (figures showing trajectories and force balance): the headline values (up to 100 μm/s, resistance to Brownian motion and external forces) are shown for the chosen parameter set. Without accompanying data on the range of rates where the condensate remains localized and asymmetric, it is unclear whether these outcomes are robust or artifacts of the specific timescale separation assumed in the minimal mechanism.
minor comments (2)
- [Abstract and Methods] The abstract states that 'numerical simulations demonstrate' the speeds and robustness, yet the methods description should explicitly list the integration scheme, grid resolution, and how Brownian dynamics are incorporated to allow independent verification.
- [Model description] Notation for the passivation threshold and reactivation boundary condition is introduced without a dedicated table of symbols; adding one would improve readability.
Simulated Author's Rebuttal
We are grateful to the referee for their thorough review and for recognizing the potential significance of our proposed mechanism for condensate-based propulsion. We have carefully considered the comments regarding the lack of parameter exploration and have revised the manuscript to include additional simulations that address these concerns directly.
read point-by-point responses
-
Referee: [Numerical model section] Numerical model section (reaction-diffusion equations and boundary conditions): the passivation-activation cycle is implemented with fixed enzymatic rates and a single free parameter set. No sweep is presented over the ratio of surface reactivation rate to bulk passivation rate relative to the colloid diffusion time. Because net propulsion requires persistent asymmetry in the condensate, this omission leaves open whether the symmetry breaking and sustained propulsion are generic or restricted to a narrow regime.
Authors: We concur that a systematic exploration of the ratio between the surface reactivation rate and the bulk passivation rate, scaled by the colloid diffusion time, is necessary to substantiate the claim that the propulsion is generic. In the revised version of the manuscript, we have expanded the Numerical model section to include a parameter sweep over this dimensionless ratio. The new results, presented in an additional figure, demonstrate that symmetry breaking and sustained propulsion occur robustly for rate ratios spanning approximately one order of magnitude, corresponding to conditions where the reactivation timescale is shorter than the passivation and diffusion timescales. Propulsion speeds remain above 50 μm/s in this regime, and the mechanism fails gracefully outside it when asymmetry cannot be maintained. This supports the generality of the indefinite self-propulsion within the relevant biophysical parameter space. revision: yes
-
Referee: [Results on propulsion speeds and robustness] Results on propulsion speeds and robustness (figures showing trajectories and force balance): the headline values (up to 100 μm/s, resistance to Brownian motion and external forces) are shown for the chosen parameter set. Without accompanying data on the range of rates where the condensate remains localized and asymmetric, it is unclear whether these outcomes are robust or artifacts of the specific timescale separation assumed in the minimal mechanism.
Authors: We thank the referee for this observation. To clarify the robustness, we have added to the Results section a detailed analysis of how the propulsion speed, trajectory stability, and resistance to perturbations vary with the key rate ratio. We quantify the condensate localization and asymmetry using order parameters and show that the high speeds (up to 100 μm/s) and strong resistance to Brownian motion and external forces are achieved precisely in the parameter region where the condensate is localized and asymmetric. We also include data for cases where the rate ratio leads to delocalized or symmetric condensates, in which propulsion is absent. These additions confirm that the reported performance is not an artifact but a feature of the mechanism when the passivation-activation cycle maintains the necessary asymmetry. revision: yes
Circularity Check
No significant circularity; propulsion emerges from explicit numerical integration of the defined reaction cycle.
full rationale
The paper defines a minimal biochemical mechanism (phase-separating proteins, bulk passivation enzymes, surface-anchored reactivation enzymes) and reports propulsion speeds obtained by numerical solution of the resulting dynamics. No equations, parameters, or self-citations are shown that reduce the reported speeds or symmetry breaking to a fitted input or tautological redefinition. The central result is a forward simulation outcome under stated assumptions rather than an algebraic identity or load-bearing self-reference. The derivation chain is therefore self-contained.
Axiom & Free-Parameter Ledger
free parameters (1)
- enzymatic reaction rates and passivation thresholds
axioms (1)
- domain assumption Phase-separating proteins can be reversibly passivated by enzymes and reactivated by surface-anchored complementary enzymes in an isotropic solution.
invented entities (1)
-
Nucleated active condensate near the colloid surface
no independent evidence
Reference graph
Works this paper leans on
-
[1]
Without loss of gen- erality, we may measure length in units of the dense phase- dilute phase interface widthξand time in units of molecular diffusionξ 2/(µmolkBT)
Parameter values We first specify the parameter values. Without loss of gen- erality, we may measure length in units of the dense phase- dilute phase interface widthξand time in units of molecular diffusionξ 2/(µmolkBT). Nonetheless, we will assign partic- ular values to these quantities to make concrete magnitude estimates of the typical forces and propu...
-
[2]
Value (main text)
Simulation: self-propelled colloid We perform finite-volume simulations of Eqs. (8) and (9), where the fluxes are explicitly given by 7 We also performed simulations withU 0 = 0andU 0 =k BT, which permitted wetting. However, the phase diagrams were significantly more intricate to control due to exceedingly long running times needed to reach stable wetting...
-
[3]
phosphatase condensate
Simulation: self-propelled condensate To extend the generality of our propulsion scenario, we performed equivalent simulations to the above, but with a “phosphatase condensate” (p-condensate) replacing the col- loid. We use again the quasi-two-dimensional (cylindrical) geometry, where the p-condensate has an initial radius20ξ and is centered at the origin...
-
[4]
We ignore protein-colloid repul- sion and discuss an ideal solution for simplicity (no phase sep- aration)
Instantaneous dephosphorylation Prior to considering a phosphatase with a finite rate, we compute the concentration profile for instantaneous phospho- rylation as in the main text. We ignore protein-colloid repul- sion and discuss an ideal solution for simplicity (no phase sep- aration). Equations (8) and (9) at steady state simplify to 1 r2 d dr [r2Js,r(...
-
[5]
catalytic volume
Nonzero phosphatase volume Suppose the phosphatase occupies a particular “catalytic volume”, namely, a shell of widthwp around the colloid. This modifies Eqs. (B1) and (B2) to 1 r2 d dr [r2Js,r(r)] =−kρ s(r) +k pΘ(R+w p −r)ρ ns(r), (B9) 1 r2 d dr [r2Jns,r(r)] = +kρ s(r)−k pΘ(R+w p −r)ρ ns(r), (B10) wherek p is the effective rate of the phosphatase, depend...
-
[6]
Thus, the phosphatase does not operate any- where in the bulk, and the steady concentration profiles fol- low Eqs
Linear adsorption theory Here, we ignore the volume occupied by phosphatases, and instead consider a model where nonsticky molecules should first adsorb to the colloid surface, undergo phosphorylation, and then desorb. Thus, the phosphatase does not operate any- where in the bulk, and the steady concentration profiles fol- low Eqs. (B1) and (B2). However,...
-
[7]
Estimating energetic efficiency In this section, we detail the estimates behind the energy consumption stated for our system in Sec. VI C. Consider theR= 0.2µmcolloid. Its Stokes mobility isµ col = (6πηR) −1 ∼300µm/(pN×s), and when mobile it expe- riences forces of orderF∼0.3pN(Fig. 4). Therefore, the energy investment needed to oppose the friction experi...
-
[8]
Miyata, R
M. Miyata, R. C. Robinson, T. Q. P. Uyeda, Y . Fukumori, S.-i. Fukushima, S. Haruta, M. Homma, K. Inaba, M. Ito, C. Kaito, K. Kato, T. Kenri, Y . Kinosita, S. Kojima, T. Mi- namino, H. Mori, S. Nakamura, D. Nakane, K. Nakayama, M. Nishiyama, S. Shibata, K. Shimabukuro, M. Tamakoshi, A. Taoka, Y . Tashiro, I. Tulum, H. Wada, and K.-i. Wak- abayashi, Tree o...
2020
-
[9]
Wadhwa and H
N. Wadhwa and H. C. Berg, Bacterial motility: machinery and mechanisms, Nat. Rev. Microbiol.20, 161 (2022)
2022
-
[10]
Nakamura and T
S. Nakamura and T. Minamino, Flagella-driven motility of bac- teria, Biomolecules9, 279 (2019)
2019
-
[11]
H. L. Sweeney and E. L. Holzbaur, Motor proteins, Cold Spring Harb. Perspect. Biol.10, 10.1101/cshperspect.a021931 (2018)
-
[12]
H. Noji, R. Yasuda, M. Yoshida, and K. Kinosita, Direct obser- vation of the rotation of F1-ATPase, Nature386, 299 (1997)
1997
-
[13]
Bechinger, R
C. Bechinger, R. Di Leonardo, H. L ¨owen, C. Reichhardt, G. V olpe, and G. V olpe, Active particles in complex and crowded environments, Rev. Mod. Phys.88, 045006 (2016)
2016
-
[14]
Scott, M
A. Scott, M. Xu, I. Murphy, D. S. J. Jang, Z. M. Rana, A. Estrada, W. W. Ahmed, W. B. Rogers, and J. L. Ross, En- hanced diffusion of single, lipid-tethered enzymes, Nano Lett. 26, 2458 (2026)
2026
-
[15]
Ghosh, A
S. Ghosh, A. Somasundar, and A. Sen, Enzymes as active mat- ter, Annu. Rev. Cond. Matter Phys.12, 177 (2021)
2021
-
[16]
Michelin, Self-propulsion of chemically active droplets, Annu Rev
S. Michelin, Self-propulsion of chemically active droplets, Annu Rev. Fluid Mech.55, 77 (2023)
2023
-
[17]
Izzet, P
A. Izzet, P. G. Moerman, P. Gross, J. Groenewold, A. D. Hollingsworth, J. Bibette, and J. Brujic, Tunable persistent ran- dom walk in swimming droplets, Phys. Rev. X10, 021035 (2020)
2020
-
[18]
J. R. Howse, R. A. L. Jones, A. J. Ryan, T. Gough, R. Vafabakhsh, and R. Golestanian, Self-motile colloidal par- ticles: From directed propulsion to random walk, Phys. Rev. Lett.99, 048102 (2007)
2007
-
[19]
Thutupalli, R
S. Thutupalli, R. Seemann, and S. Herminghaus, Swarming be- havior of simple model squirmers, New J. Phys.13, 073021 (2011)
2011
-
[20]
S. J. Ebbens and J. R. Howse, In pursuit of propulsion at the nanoscale, Soft Matter6, 726 (2010)
2010
-
[21]
Michelin and E
S. Michelin and E. Lauga, Phoretic self-propulsion at finite P´eclet numbers, J. Fluid Mech.747, 572–604 (2014)
2014
-
[22]
Shin and C
Y . Shin and C. P. Brangwynne, Liquid phase condensation in cell physiology and disease, Science357, eaaf4382 (2017)
2017
-
[23]
S. F. Banani, H. O. Lee, A. A. Hyman, and M. K. Rosen, Biomolecular condensates: Organizers of cellular biochemistry, Nat. Rev. Mol. Cell Biol.18, 285 (2017)
2017
-
[24]
X. Su, J. A. Ditlev, E. Hui, W. Xing, S. Banjade, J. Okrut, D. S. King, J. Taunton, M. K. Rosen, and R. D. Vale, Phase sepa- ration of signaling molecules promotes T cell receptor signal transduction, Science352, 595 (2016)
2016
-
[25]
Molliex, J
A. Molliex, J. Temirov, J. Lee, M. Coughlin, A. P. Kanagaraj, H. J. Kim, T. Mittag, and J. P. Taylor, Phase separation by low complexity domains promotes stress granule assembly and drives pathological fibrillization, Cell163, 123 (2015)
2015
-
[26]
Feric, N
M. Feric, N. Vaidya, T. S. Harmon, D. M. Mitrea, L. Zhu, T. M. Richardson, R. W. Kriwacki, R. V . Pappu, and C. P. Brang- wynne, Coexisting liquid phases underlie nucleolar subcom- partments, Cell165, 1686 (2016)
2016
-
[27]
D. S. W. Lee, C.-H. Choi, D. W. Sanders, L. Beckers, J. A. Rib- ack, C. P. Brangwynne, and N. S. Wingreen, Size distributions of intracellular condensates reflect competition between coales- cence and nucleation, Nat. Phys.19, 586 (2023)
2023
-
[28]
Mart ´ınez-Calvo, J
A. Mart ´ınez-Calvo, J. Zhou, Y . Zhang, and N. S. Wingreen, Sticky enzymes: Increased metabolic efficiency via substrate- dependent enzyme clustering, PRX Life3, 033011 (2025)
2025
-
[29]
Buchner, F
A. Buchner, F. Tostevin, F. Hinzpeter, and U. Gerland, Opti- mization of collective enzyme activity via spatial localization, J. Chem. Phys.139, 135101 (2013)
2013
-
[30]
Castellana, M
M. Castellana, M. Z. Wilson, Y . Xu, P. Joshi, I. M. Cristea, J. D. Rabinowitz, Z. Gitai, and N. S. Wingreen, Enzyme cluster- ing accelerates processing of intermediates through metabolic channeling, Nat. Biotechnol.32, 1011 (2014)
2014
-
[31]
Zwicker, O
D. Zwicker, O. W. Paulin, and C. ter Burg, Physics of droplet regulation in biological cells, Rep. Prog. Phys.88, 116601 (2025)
2025
-
[32]
Zwicker, A
D. Zwicker, A. A. Hyman, and F. J ¨ulicher, Suppression of Ost- wald ripening in active emulsions, Phys. Rev. E92, 012317 (2015)
2015
-
[33]
Zwicker, M
D. Zwicker, M. Decker, S. Jaensch, A. A. Hyman, and F. J¨ulicher, Centrosomes are autocatalytic droplets of pericen- triolar material organized by centrioles, Proc. Natl. Acad. Sci. U.S.A.111, E2636 (2014)
2014
-
[34]
Sorkin and N
B. Sorkin and N. S. Wingreen, Accelerated ostwald ripening by chemical activity, Journal of the American Chemical Society 147, 44673 (2025)
2025
-
[35]
S. He, L. M. Lemma, A. Martinez-Calvo, G. He, J. H. Hen- nacy, L. Wang, S. L. Ergun, A. K. Rai, C. Wang, L. Bun- day, A. Kayser-Browne, Q. Wang, C. P. Brangwynne, N. S. Wingreen, and M. C. Jonikas, Kinase KEY1 controls pyrenoid condensate size throughout the cell cycle by disrupting phase separation interactions, Nat. Cell Biol.28, 725 (2026)
2026
-
[36]
A. R. Strom, Y . Kim, H. Zhao, Y .-C. Chang, N. D. Orlovsky, A. Koˇsmrlj, C. Storm, and C. P. Brangwynne, Condensate in- 18 terfacial forces reposition DNA loci and probe chromatin vis- coelasticity, Cell , 5282 (2024)
2024
-
[37]
Semeigazin, K
A. Semeigazin, K. Minami, M. A. Shimazoe, S. Khochbin, D. Panne, S. Ide, and K. Maeshima, Partition coefficients reveal changes in properties of low-contrast biomolecular condensates (2025), bioRxiv:2025.07.05.663270
2025
-
[38]
S ¨oding, D
J. S ¨oding, D. Zwicker, S. Sohrabi-Jahromi, M. Boehning, and J. Kirschbaum, Mechanisms for active regulation of biomolec- ular condensates, Trends Cell Biol.30, 4 (2020)
2020
-
[39]
E. H. Brumbaugh-Reed, Y . Gao, K. Aoki, and J. E. Toettcher, Rapid and reversible dissolution of biomolecular condensates using light-controlled recruitment of a solubility tag, Nat. Com- mun.15, 6717 (2024)
2024
-
[40]
Doi,Soft Matter Physics(Oxford University Press, 2013)
M. Doi,Soft Matter Physics(Oxford University Press, 2013)
2013
-
[41]
J. F. Joanny, L. Leibler, and P. G. De Gennes, Effects of polymer solutions on colloid stability, J. Pol. Sci.: Pol. Phys. Ed.17, 1073 (1979)
1979
-
[42]
G. J. Fleer, M. A. Cohen Stuart, J. M. H. M. Scheutjens, T. Cos- grove, and B. Vincent,Polymers at Interfaces(Springer, 1998)
1998
-
[43]
S. He, L. M. Lemma, A. Martinez-Calvo, G. He, J. H. Hen- nacy, L. Wang, S. L. Ergun, A. K. Rai, C. Wang, L. Bun- day, A. Kayser-Browne, Q. Wang, C. P. Brangwynne, N. S. Wingreen, and M. C. Jonikas, Kinase KEY1 controls pyrenoid condensate size throughout the cell cycle by disrupting phase separation interactions, Nat. Cell Biol. (2026)
2026
-
[44]
A. B. R. McIntyre, A. B. Tschan, K. Meyer, S. Walser, A. K. Rai, K. Fujita, and L. Pelkmans, Phosphorylation of a nuclear condensate regulates cohesion and mRNA retention, Nat. Com- mun.16, 390 (2025)
2025
-
[45]
Bray, Theory of phase-ordering kinetics, Adv
A. Bray, Theory of phase-ordering kinetics, Adv. Phys.43, 357 (1994)
1994
-
[46]
P. J. Flory, Thermodynamics of high polymer solutions, J. Chem. Phys.10, 51 (1942)
1942
-
[47]
M. L. Huggins, Solutions of long chain compounds, J. Chem. Phys.9, 440 (1941)
1941
-
[48]
J. W. Cahn and J. E. Hilliard, Free energy of a nonuniform sys- tem. I. Interfacial free energy, J. Chem. Phys.28, 258 (1958)
1958
-
[49]
S. Mao, D. Kuldinow, M. P. Haataja, and A. Ko ˇsmrlj, Phase behavior and morphology of multicomponent liquid mixtures, Soft Matter15, 1297 (2019)
2019
-
[50]
S. R. De Groot and P. Mazur,Non-equilibrium thermodynamics (Dover Publications, 1984)
1984
-
[51]
Schuss,Theory and Applications of Stochastic Processes (Springer, 2010)
Z. Schuss,Theory and Applications of Stochastic Processes (Springer, 2010)
2010
-
[52]
Einstein, ¨Uber die von der molekularkinetischen theorie der w¨arme geforderte bewegung von in ruhenden fl¨ussigkeiten sus- pendierten teilchen, Ann
A. Einstein, ¨Uber die von der molekularkinetischen theorie der w¨arme geforderte bewegung von in ruhenden fl¨ussigkeiten sus- pendierten teilchen, Ann. Phys.322, 549 (1905)
1905
-
[53]
Lifshitz and V
I. Lifshitz and V . Slyozov, The kinetics of precipitation from supersaturated solid solutions, J. Phys. Chem. Solids19, 35 (1961)
1961
-
[54]
E. D. Siggia, Late stages of spinodal decomposition in binary mixtures, Phys. Rev. A20, 595 (1979)
1979
-
[55]
M. E. Cates, Complex fluids: The physics of emulsions, inSoft Interfaces: Lecture Notes of the Les Houches Summer, V ol. 98, edited by L. Bocquet, D. Qu ´er´e, T. A. Witten, and L. Cuglian- dolo (Oxford University Press, 2017) p. 317
2017
-
[56]
H. B. Eral, G. Manukyan, and J. M. Oh, Wetting of a drop on a sphere, Langmuir27, 5340 (2011)
2011
-
[57]
Smith and T
P. Smith and T. Van De Ven, A note on the thermodynamics of partial engulfment, Colloids Surf.2, 387 (1981)
1981
-
[58]
K. C. Neuman and S. M. Block, Optical trapping, Rev. Sci. In- strum.75, 2787 (2004)
2004
-
[59]
Baiesi and C
M. Baiesi and C. Maes, An update on the nonequilibrium linear response, New J. Phys.15, 013004 (2013)
2013
-
[60]
Y . Dai, L. You, and A. Chilkoti, Engineering synthetic biomolecular condensates, Nat. Rev. Bioeng.1, 466 (2023)
2023
-
[61]
Qiang, C
Y . Qiang, C. Luo, and D. Zwicker, Self-propulsion via nontran- sitive phase coexistence in chemically active mixtures, Phys. Rev. Lett.135, 268301 (2025)
2025
-
[62]
Demarchi, A
L. Demarchi, A. Goychuk, I. Maryshev, and E. Frey, Enzyme- enriched condensates show self-propulsion, positioning, and coexistence, Phys. Rev. Lett.130, 128401 (2023)
2023
-
[63]
Raßhofer, S
F. Raßhofer, S. Bauer, A. Ziepke, I. Maryshev, and E. Frey, Cap- illary wave formation in conserved active emulsions, Phys. Rev. Res.7, 043240 (2025)
2025
-
[64]
E. H. Hauge and A. Martin-L ¨of, Fluctuating hydrodynamics and Brownian motion, J. Stat. Phys.7, 259 (1973)
1973
-
[65]
L. D. Landau and E. M. Lifshitz,Fluid Mechanics: V olume 6, 2nd ed. (Butterworth-Heinemann, 1987)
1987
-
[66]
S. Shim, B. Gouveia, B. Ramm, V . A. Valdez, S. Petry, and H. A. Stone, Motorless transport of microtubules along tubulin, ranGTP, and salt gradients, Nat. Commun.15, 9434 (2024)
2024
-
[67]
B. Ramm, A. Goychuk, A. Khmelinskaia, P. Blumhardt, H. Eto, K. A. Ganzinger, E. Frey, and P. Schwille, A diffusiophoretic mechanism for ATP-driven transport without motor proteins, Nat. Phys.17, 850 (2021)
2021
-
[68]
R. P. Sear, Diffusiophoresis in cells: A general nonequilibrium, nonmotor mechanism for the metabolism-dependent transport of particles in cells, Phys. Rev. Lett.122, 128101 (2019)
2019
-
[69]
Shim, Diffusiophoresis, diffusioosmosis, and microfluidics: Surface-flow-driven phenomena in the presence of flow, Chem
S. Shim, Diffusiophoresis, diffusioosmosis, and microfluidics: Surface-flow-driven phenomena in the presence of flow, Chem. Rev.122, 6986 (2022)
2022
-
[70]
Marbach, H
S. Marbach, H. Yoshida, and L. Bocquet, Local and global force balance for diffusiophoretic transport, J. Fluid Mech.892, A6 (2020)
2020
-
[71]
N. Shi, A. a. Fattah, and T. M. Squires, Generalized, concep- tually unified theory of linear phoretic drift and osmotic slip, Phys. Rev. Fluids10, 043701 (2025)
2025
-
[72]
D. C. Prieve, J. L. Anderson, J. P. Ebel, and M. E. Lowell, Mo- tion of a particle generated by chemical gradients. Part 2. Elec- trolytes, J. Fluid Mech.148, 247–269 (1984)
1984
-
[73]
J. L. Anderson and D. C. Prieve, Diffusiophoresis: Migration of colloidal particles in gradients of solute concentration, Sep. Purif. Methods13, 67 (1984)
1984
-
[74]
Michelin, E
S. Michelin, E. Lauga, and D. Bartolo, Spontaneous au- tophoretic motion of isotropic particles, Phys. Fluids25, 061701 (2013)
2013
-
[75]
B. V . Hokmabad, R. Dey, M. Jalaal, D. Mohanty, M. Almukam- betova, K. A. Baldwin, D. Lohse, and C. C. Maass, Emergence of bimodal motility in active droplets, Phys. Rev. X11, 011043 (2021)
2021
-
[76]
Peliti and S
L. Peliti and S. Pigolotti,Stochastic Thermodynamics(Prince- ton University Press, 2021)
2021
-
[77]
Ariga, M
T. Ariga, M. Tomishige, and D. Mizuno, Nonequilibrium ener- getics of molecular motor kinesin, Phys. Rev. Lett.121, 218101 (2018)
2018
-
[78]
Markovich, E
T. Markovich, E. Fodor, E. Tjhung, and M. E. Cates, Thermo- dynamics of active field theories: Energetic cost of coupling to reservoirs, Phys. Rev. X11, 021057 (2021)
2021
-
[79]
Varma, D
K. Varma, D. Matthias, C. B. Shapiro, S. Bailey-Darland, T. Matsuzawa, C. Lorenz, T. Bate, S. J. Thornton, S. Duraivel, R. W. Style, J. P. Sethna, and E. R. Dufresne, Partition coeffi- cients reveal changes in properties of low-contrast biomolecular condensates (2026), bioRxiv:2026.02.20.707107
2026
-
[80]
J. N. Israelachvili,Intermolecular and Surface F orces, 3rd ed. (Academic Press, 2011)
2011
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.