Recognition: unknown
Lumens as active balloons: a biological physics review
Pith reviewed 2026-05-09 17:08 UTC · model grok-4.3
The pith
Lumens function as active balloons inflated, sculpted, and maintained by coupled active processes.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Lumens are cavities enclosed by polarized cells that can be understood as active balloons: pressurized cavities that are inflated, sculpted, and maintained through tightly coupled active processes within a biological physics framework that incorporates hydraulic flows, instabilities, and mechanochemical feedbacks.
What carries the argument
The active balloon model, in which lumens are pressurized cavities inflated, sculpted, and maintained by coupled active processes including osmotically driven flows and mechanochemical feedbacks.
Load-bearing premise
The phenomena across biological systems share sufficient common physical principles to allow unification under the active balloon framework.
What would settle it
Observation of a functional biological lumen that develops and persists without pressurized cavity dynamics, osmotically driven flows, or mechanochemical pressure feedbacks would falsify the unifying claim.
Figures
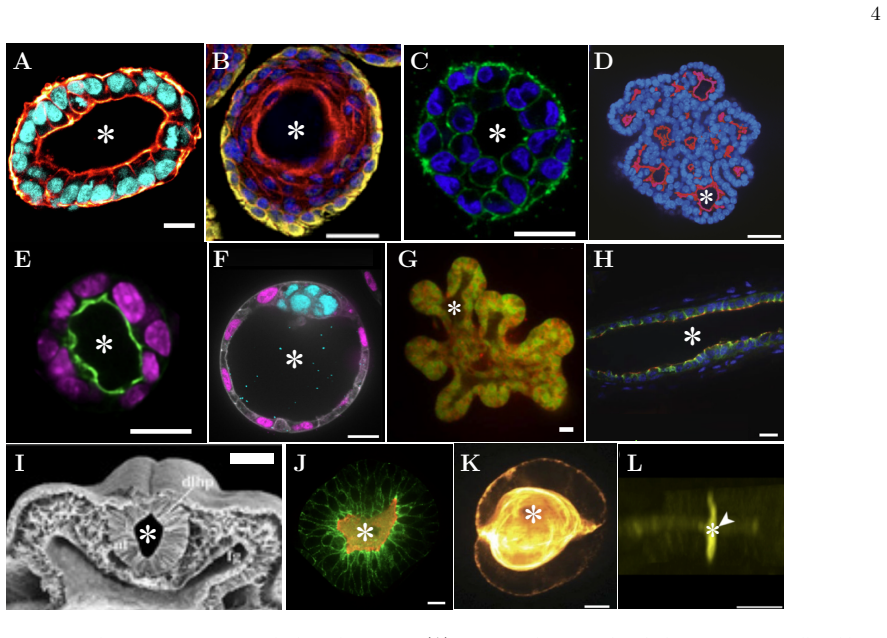













read the original abstract
Lumens are cavities enclosed by polarized cells that are essential for organ function, from nutrient transport in the gut to gas exchange in the lungs. Defects in lumen formation are associated with severe diseases, including polycystic kidney disease and respiratory malformations. The emergence, growth, and maintenance of lumens involve a rich set of phenomena that can be framed within out-of-equilibrium physics and biological active matter, including osmotically driven hydraulic flows, coarsening-like dynamics, morphological instabilities, and mechanochemical feedbacks linking luminal pressure to tissue response. Yet experimental and theoretical efforts to study these phenomena have largely developed within specific biological systems, complicating the identification of shared physical principles across them. In this review, we bring these efforts together and present lumenogenesis within a biological physics framework in which lumens are viewed as active balloons: pressurized cavities that are inflated, sculpted, and maintained through tightly coupled active processes.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript is a review article that synthesizes existing literature on lumen formation (lumenogenesis) across diverse biological systems. It proposes a unifying biological physics framework in which lumens are conceptualized as 'active balloons': pressurized cavities that are inflated, sculpted, and maintained through tightly coupled active processes, including osmotically driven hydraulic flows, coarsening-like dynamics, morphological instabilities, and mechanochemical feedbacks linking luminal pressure to tissue response.
Significance. If the proposed framing holds, the review could help identify shared physical principles underlying lumenogenesis in contexts ranging from organ development to disease states such as polycystic kidney disease. By collecting and reframing system-specific studies within out-of-equilibrium physics and active matter, it provides a descriptive lens that may guide future modeling and experiments without introducing new derivations, data, or parameter fits.
minor comments (2)
- The abstract and introduction introduce the 'active balloons' metaphor without a dedicated early section that formally defines its minimal physical ingredients (e.g., the minimal set of active processes required for the analogy to hold). Adding such a definition would improve clarity for readers outside the immediate subfield.
- Several literature examples are cited to illustrate hydraulic flows and instabilities; a short table summarizing the key physical parameters (e.g., pressure ranges, timescales) extracted from those studies would help readers assess the degree of quantitative unification achieved by the framework.
Simulated Author's Rebuttal
We thank the referee for their positive and supportive review of our manuscript. We are pleased that the 'active balloons' framing is viewed as a useful unifying lens for synthesizing lumenogenesis across systems, and we appreciate the recommendation to accept.
Circularity Check
No significant circularity: review synthesis without derivations
full rationale
This is a review paper that collects and reframes existing lumenogenesis studies from independent biological systems under a descriptive 'active balloons' lens. No new equations, derivations, parameter fits, or predictions are introduced. The central claim reduces to whether the cited external examples are consistent with the proposed viewpoint, with no internal reduction to self-defined inputs or load-bearing self-citations. The argument is self-contained against external benchmarks and does not exhibit any of the enumerated circularity patterns.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Lumen formation involves hydraulic flows, instabilities and mechanochemical feedbacks.
invented entities (1)
-
active balloons
no independent evidence
Reference graph
Works this paper leans on
-
[1]
While significant experimental and theoretical progress has been made, several open questions remain that we believe are particularly well-suited for physics-based approaches
Outlook In this review, we have characterized lumens as active balloons whose emergence and maintenance depend on a nonlinear and reciprocal interplay between hydraulic forces, cell mechanics, biochemical signaling, ECM properties, and cell dynamics. While significant experimental and theoretical progress has been made, several open questions remain that ...
-
[2]
Acknowledgments This work was supported by NSF MCB 2426002 and NSF PHY 2310496 to W.-J.R., and by a Prebys Foundation Research Heroes grant to S.I.F. S.E.- A. thanks Magdalena Fadic Repetto for the careful reading of the text and for her help in designing Fig. 2
-
[3]
Die isolirten blastomeren des echinidenkeimes.Development Genes and Evolu- tion, 10(2):361–410, 1900
Hans Driesch. Die isolirten blastomeren des echinidenkeimes.Development Genes and Evolu- tion, 10(2):361–410, 1900
1900
-
[4]
On growth and form, 1917
J Arthur Thomson. On growth and form, 1917
1917
-
[5]
The chemical basis of morpho- genesis.Bulletin of mathematical biology, 52:153–197, 1990
Alan Mathison Turing. The chemical basis of morpho- genesis.Bulletin of mathematical biology, 52:153–197, 1990
1990
-
[6]
Positional information and the spatial pattern of cellular differentiation.Journal of theoretical biology, 25(1):1–47, 1969
Lewis Wolpert. Positional information and the spatial pattern of cellular differentiation.Journal of theoretical biology, 25(1):1–47, 1969
1969
-
[7]
A gene complex controlling segmenta- tion in drosophila.Nature, 276(5688):565–570, 1978
Edward B Lewis. A gene complex controlling segmenta- tion in drosophila.Nature, 276(5688):565–570, 1978
1978
-
[8]
On symmetry- breaking instabilities in dissipative systems.The Journal of Chemical Physics, 46(9):3542–3550, 1967
Ilya Prigogine and Gr´ egoire Nicolis. On symmetry- breaking instabilities in dissipative systems.The Journal of Chemical Physics, 46(9):3542–3550, 1967
1967
-
[9]
Symmetry breaking instabilities in dissipative systems
Ilya Prigogine and Ren´ e Lefever. Symmetry breaking instabilities in dissipative systems. ii.The Journal of Chemical Physics, 48(4):1695–1700, 1968
1968
-
[10]
Fluctuations in nonequilibrium systems.Proceedings of the National Academy of Sciences, 68(9):2102–2107, 1971
Gr´ egoire Nicolis and Ilya Prigogine. Fluctuations in nonequilibrium systems.Proceedings of the National Academy of Sciences, 68(9):2102–2107, 1971
1971
-
[11]
Left– right patterning: breaking symmetry to asymmetric morphogenesis.Trends in Genetics, 33(9):616–628, 2017
Daniel T Grimes and Rebecca D Burdine. Left– right patterning: breaking symmetry to asymmetric morphogenesis.Trends in Genetics, 33(9):616–628, 2017
2017
-
[12]
Symmetry breaking in the mammalian embryo.Annual review of cell and developmental biology, 34(1):405–426, 2018
Hui Ting Zhang and Takashi Hiiragi. Symmetry breaking in the mammalian embryo.Annual review of cell and developmental biology, 34(1):405–426, 2018
2018
-
[13]
Mechanochemical symmetry breaking during morpho- 37 genesis of lateral-line sensory organs.Nature physics, 16(9):949–957, 2020
A Erzberger, A Jacobo, A Dasgupta, and AJ Hudspeth. Mechanochemical symmetry breaking during morpho- 37 genesis of lateral-line sensory organs.Nature physics, 16(9):949–957, 2020
2020
-
[14]
Inflationary theory of branching morphogenesis in the mouse salivary gland.Nature communications, 14(1):3422, 2023
Ignacio Bordeu, Lemonia Chatzeli, and Benjamin D Si- mons. Inflationary theory of branching morphogenesis in the mouse salivary gland.Nature communications, 14(1):3422, 2023
2023
-
[15]
Dynamical systems of fate and form in development
Alex M Plum and Mattia Serra. Dynamical systems of fate and form in development. InSeminars in Cell & Developmental Biology, volume 172, page 103620. Elsevier, 2025
2025
-
[16]
Patterning in motion: Cell interfaces guide mesenchymal collective migration and morphogenesis.Journal of Cell Biology, 224(11):e202505198, 2025
Maik C Bischoff and Roberto Mayor. Patterning in motion: Cell interfaces guide mesenchymal collective migration and morphogenesis.Journal of Cell Biology, 224(11):e202505198, 2025
2025
-
[17]
From morphogen to morphogenesis and back.Nature, 541(7637):311–320, 2017
Darren Gilmour, Martina Rembold, and Maria Leptin. From morphogen to morphogenesis and back.Nature, 541(7637):311–320, 2017
2017
-
[18]
Computational approaches to developmental patterning.Science, 336(6078):187–191, 2012
Luis G Morelli, Koichiro Uriu, Sa´ ul Ares, and Andrew C Oates. Computational approaches to developmental patterning.Science, 336(6078):187–191, 2012
2012
-
[19]
Hydrodynamics of soft active matter.Reviews of modern physics, 85(3):1143– 1189, 2013
M Cristina Marchetti, Jean-Fran¸ cois Joanny, Sriram Ramaswamy, Tanniemola B Liverpool, Jacques Prost, Madan Rao, and R Aditi Simha. Hydrodynamics of soft active matter.Reviews of modern physics, 85(3):1143– 1189, 2013
2013
-
[20]
Active gel physics.Nature physics, 11(2):111–117, 2015
Jacques Prost, Frank J¨ ulicher, and Jean-Fran¸ cois Joanny. Active gel physics.Nature physics, 11(2):111–117, 2015
2015
-
[21]
Courier Dover Publications, 2017
Ilya Prigogine.Non-equilibrium statistical mechanics. Courier Dover Publications, 2017
2017
-
[22]
Turing’s next steps: the mechanochemical basis of morphogenesis.Nature Reviews Molecular Cell Biology, 12(6):392–398, 2011
Jonathon Howard, Stephan W Grill, and Justin S Bois. Turing’s next steps: the mechanochemical basis of morphogenesis.Nature Reviews Molecular Cell Biology, 12(6):392–398, 2011
2011
-
[23]
Adam Navis and Celeste M. Nelson. Pulling together: Tissue-generated forces that drive lumen morphogene- sis.Seminars in Cell & Developmental Biology, 55:139– 147, 2016
2016
-
[24]
Tight junctions control lumen morphology via hydrostatic pressure and junctional tension.Developmental Cell, 59(21):2866– 2881, 2024
Markus Mukenhirn, Chen-Ho Wang, Tristan Guyomar, Matthew J Bovyn, Michael F Staddon, Rozemarijn E van der Veen, Riccardo Maraspini, Linjie Lu, Cecilie Martin-Lemaitre, Masaki Sano, et al. Tight junctions control lumen morphology via hydrostatic pressure and junctional tension.Developmental Cell, 59(21):2866– 2881, 2024
2024
-
[25]
Dynamic changes in epithelial cell packing during tissue morphogenesis
Sandra B Lemke and Celeste M Nelson. Dynamic changes in epithelial cell packing during tissue morphogenesis. Current Biology, 31(18):R1098–R1110, 2021
2021
-
[26]
Mechanochemical dynamics of collective cells and hierarchical topological defects in multicellular lumens.Science Advances, 10(18):eadn0172, 2024
Pengyu Yu, Yue Li, Wei Fang, Xi-Qiao Feng, and Bo Li. Mechanochemical dynamics of collective cells and hierarchical topological defects in multicellular lumens.Science Advances, 10(18):eadn0172, 2024
2024
-
[27]
Physics of lumen growth
Sabyasachi Dasgupta, Kapish Gupta, Yue Zhang, Virgile Viasnoff, and Jacques Prost. Physics of lumen growth. Proceedings of the National Academy of Sciences, 115(21):E4751–E4757, 2018
2018
-
[28]
Actin cortex mechanics and cellular morphogenesis
Guillaume Salbreux, Guillaume Charras, and Ewa Paluch. Actin cortex mechanics and cellular morphogenesis. Trends in cell biology, 22(10):536–545, 2012
2012
-
[29]
Cadherin cell adhesion receptors as a morphogenetic regulator.Science, 251(5000):1451– 1455, 1991
Masatoshi Takeichi. Cadherin cell adhesion receptors as a morphogenetic regulator.Science, 251(5000):1451– 1455, 1991
1991
-
[30]
Tube morpho- genesis: making and shaping biological tubes.Cell, 112(1):19–28, 2003
Barry Lubarsky and Mark A Krasnow. Tube morpho- genesis: making and shaping biological tubes.Cell, 112(1):19–28, 2003
2003
-
[31]
Regulation of cell polarity during epithelial morphogenesis.Cur- rent opinion in cell biology, 20(2):227–234, 2008
Fernando Martin-Belmonte and Keith Mostov. Regulation of cell polarity during epithelial morphogenesis.Cur- rent opinion in cell biology, 20(2):227–234, 2008
2008
-
[32]
Luminal signalling links cell communica- tion to tissue architecture during organogenesis.Na- ture, 515(7525):120–124, 2014
Sevi Durdu, Murat Iskar, Celine Revenu, Nicole Schieber, Andreas Kunze, Peer Bork, Yannick Schwab, and Dar- ren Gilmour. Luminal signalling links cell communica- tion to tissue architecture during organogenesis.Na- ture, 515(7525):120–124, 2014
2014
-
[33]
Mouse embryo geometry drives formation of robust signaling gradients through receptor localization.Nature communications, 10(1):4516, 2019
Zhechun Zhang, Steven Zwick, Ethan Loew, Joshua S Grimley, and Sharad Ramanathan. Mouse embryo geometry drives formation of robust signaling gradients through receptor localization.Nature communications, 10(1):4516, 2019
2019
-
[34]
Deciphering epiblast lumenogenesis reveals proamniotic cavity control of embryo growth and patterning
Yung Su Kim, Rui Fan, Ludmila Kremer, Nannette Kuempel-Rink, Karina Mildner, Dagmar Zeuschner, Liesbeth Hekking, Martin Stehling, and Ivan Bedzhov. Deciphering epiblast lumenogenesis reveals proamniotic cavity control of embryo growth and patterning. Science Advances, 7(11):eabe1640, 2021
2021
-
[35]
Lumen expansion is initially driven by api- cal actin polymerization followed by osmotic pressure in a human epiblast model.Cell stem cell, 31(5):640–656, 2024
Dhiraj Indana, Andrei Zakharov, Youngbin Lim, Alexan- der R Dunn, Nidhi Bhutani, Vivek B Shenoy, and Ovijit Chaudhuri. Lumen expansion is initially driven by api- cal actin polymerization followed by osmotic pressure in a human epiblast model.Cell stem cell, 31(5):640–656, 2024
2024
-
[36]
Long-term expansion and differentiation of adult murine epidermal stem cells in 3d organoid cultures.Proceedings of the National Academy of Sciences, 116(29):14630–14638, 2019
Kim E Boonekamp, Kai Kretzschmar, Dominique J Wiener, Priyanca Asra, Sepideh Derakhshan, Jens Puschhof, Carmen L´ opez-Iglesias, Peter J Peters, Onur Basak, and Hans Clevers. Long-term expansion and differentiation of adult murine epidermal stem cells in 3d organoid cultures.Proceedings of the National Academy of Sciences, 116(29):14630–14638, 2019
2019
-
[37]
Global versus local matrix remodeling drives rotational versus invasive collective migration of epithelial cells.Developmental Cell, 60(6):871–884, 2025
Sural K Ranamukhaarachchi, Alyssa Walker, Man-Ho Tang, William D Leineweber, Sophia Lam, Wouter- Jan Rappel, and Stephanie I Fraley. Global versus local matrix remodeling drives rotational versus invasive collective migration of epithelial cells.Developmental Cell, 60(6):871–884, 2025
2025
-
[38]
Predicting organoid morphology through a phase field model: insights into cell division and lumenal pressure.PLoS computational biology, 21(8):e1012090, 2025
Sakurako Tanida, Kana Fuji, Linjie Lu, Tristan Guyomar, Byung Ho Lee, Alf Honigmann, Anne Grapin-Botton, Daniel Riveline, Tetsuya Hiraiwa, Makiko Nonomura, et al. Predicting organoid morphology through a phase field model: insights into cell division and lumenal pressure.PLoS computational biology, 21(8):e1012090, 2025
2025
-
[39]
Generic comparison of lumen nucleation and fusion in epithelial organoids with and without hydrostatic pressure.Nature Communications, 16(1):6307, 2025
Linjie Lu, Kana Fuji, Tristan Guyomar, Mich` ele Lieb, Marie Andr´ e, Sakurako Tanida, Makiko Nonomura, Tetsuya Hiraiwa, Yara Alcheikh, Siham Yennek, et al. Generic comparison of lumen nucleation and fusion in epithelial organoids with and without hydrostatic pressure.Nature Communications, 16(1):6307, 2025
2025
-
[40]
Multiscale analysis of single and double maternal-zygotic myh9 and myh10 mutants during mouse preimplantation development
Markus Frederik Schliffka, Anna Francesca Tortorelli, ¨Ozge ¨Ozg¨ u¸ c, Ludmilla de Plater, Oliver Polzer, Diane Pelzer, and Jean-L´ eon Maˆ ıtre. Multiscale analysis of single and double maternal-zygotic myh9 and myh10 mutants during mouse preimplantation development. Elife, 10:e68536, 2021
2021
-
[41]
Collective epithe- lial migration and cell rearrangements drive mam- mary branching morphogenesis.Developmental cell, 14(4):570–581, 2008
Andrew J Ewald, Audrey Brenot, Myhanh Duong, Bianca S Chan, and Zena Werb. Collective epithe- lial migration and cell rearrangements drive mam- mary branching morphogenesis.Developmental cell, 14(4):570–581, 2008
2008
-
[42]
A series of normal stages in the development of the chick embryo
Viktor Hamburger and Howard L Hamilton. A series of normal stages in the development of the chick embryo. Journal of morphology, 88(1):49–92, 1951
1951
-
[43]
Towards a cellular and molecular understanding of neurulation
Jean-Fran¸ cois Colas and Gary C Schoenwolf. Towards a cellular and molecular understanding of neurulation. Developmental dynamics: an official publication of the American Association of Anatomists, 221(2):117–145, 2001
2001
-
[44]
Integrin is required for basement membrane crossing and branching of an invading intracellular tube
Lauren N Meyer, Michael Hertel, and Jeremy Nance. Integrin is required for basement membrane crossing and branching of an invading intracellular tube. Development, 153(1):dev204893, 2026
2026
-
[45]
Tissue hydraulics: Physics of lu- men formation and interaction.Cells & Development, 168:203724, 2021
Alejandro Torres-S´ anchez, Max Kerr Winter, and Guil- 38 laume Salbreux. Tissue hydraulics: Physics of lu- men formation and interaction.Cells & Development, 168:203724, 2021
2021
-
[46]
Steps in the morphogenesis of a polarized epithelium: I
Allan Z Wang, George K Ojakian, and W James Nelson. Steps in the morphogenesis of a polarized epithelium: I. uncoupling the roles of cell-cell and cell-substratum contact in establishing plasma membrane polarity in multicellular epithelial (mdck) cysts.Journal of cell science, 95(1):137–151, 1990
1990
-
[47]
Polycystic kidney disease.Annual review of medicine, 60(1):321–337, 2009
Peter C Harris and Vicente E Torres. Polycystic kidney disease.Annual review of medicine, 60(1):321–337, 2009
2009
-
[48]
Cilia and polycystic kidney disease
Ming Ma. Cilia and polycystic kidney disease. In Seminars in Cell & Developmental Biology, volume 110, pages 139–148. Elsevier, 2021
2021
-
[49]
Contrast-enhanced computed tomography assessment of aortic stenosis
Timothy RG Cartlidge, Rong Bing, Jacek Kwiecinski, Ezequiel Guzzetti, Tania A Pawade, Mhairi K Doris, Philip D Adamson, Daniele Massera, Maria Lembo, Frederique ECM Peeters, et al. Contrast-enhanced computed tomography assessment of aortic stenosis. Heart, 107(23):1905–1911, 2021
1905
-
[50]
Capillary changes precede disordered alveolarization in a mouse model of bronchopulmonary dysplasia.American Journal of Respiratory Cell and Molecular Biology, 65(1):81–91, 2021
Svenja V Appuhn, Sara Siebert, Despoina Myti, Christoph Wrede, David E Surate Solaligue, David P´ erez-Bravo, Christina Brandenberger, Julia Schipke, Rory E Morty, Roman Grothausmann, et al. Capillary changes precede disordered alveolarization in a mouse model of bronchopulmonary dysplasia.American Journal of Respiratory Cell and Molecular Biology, 65(1):...
2021
-
[51]
Xuelai Liu, Peiyu Hao, Vincent Chi Hang Lui, Xianghui Xie, Yingchao Li, Yanbiao Song, Long Li, and Zhe-Wu Jin. Gut lumen formation defect can cause intestinal atresia: evidence from histological studies of human embryos and intestinal atresia septum.Journal of developmental origins of health and disease, 13(1):61– 67, 2022
2022
-
[52]
Size control of the inner ear via hydraulic feedback.Elife, 8:e39596, 2019
Kishore R Mosaliganti, Ian A Swinburne, Chon U Chan, Nikolaus D Obholzer, Amelia A Green, Shreyas Tanksale, L Mahadevan, and Sean G Megason. Size control of the inner ear via hydraulic feedback.Elife, 8:e39596, 2019
2019
-
[53]
Molecular regulation of lumen morphogenesis.Current Biology, 21(3):R126–R136, 2011
Anirban Datta, David M Bryant, and Keith E Mostov. Molecular regulation of lumen morphogenesis.Current Biology, 21(3):R126–R136, 2011
2011
-
[54]
Lumen formation is an intrinsic property of isolated human pluripotent stem cells.Stem cell reports, 5(6):954–962, 2015
Kenichiro Taniguchi, Yue Shao, Ryan F Townshend, Yu-Hwai Tsai, Cynthia J DeLong, Shawn A Lopez, Srimonta Gayen, Andrew M Freddo, Deming J Chue, Dennis J Thomas, et al. Lumen formation is an intrinsic property of isolated human pluripotent stem cells.Stem cell reports, 5(6):954–962, 2015
2015
-
[55]
From cells to organs: building polarized tissue.Nature reviews Molecular cell biology, 9(11):887–901, 2008
David M Bryant and Keith E Mostov. From cells to organs: building polarized tissue.Nature reviews Molecular cell biology, 9(11):887–901, 2008
2008
-
[56]
Molecular mechanisms of de novo lumen formation.Nature reviews Molecular cell biology, 15(10):665–676, 2014
Sara Sigurbj¨ ornsd´ ottir, Renjith Mathew, and Maria Leptin. Molecular mechanisms of de novo lumen formation.Nature reviews Molecular cell biology, 15(10):665–676, 2014
2014
-
[57]
Claudin-1 and-2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin.The Journal of cell biology, 141(7):1539–1550, 1998
Mikio Furuse, Kohji Fujita, Takashi Hiiragi, Kazushi Fujimoto, and Shoichiro Tsukita. Claudin-1 and-2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin.The Journal of cell biology, 141(7):1539–1550, 1998
1998
-
[58]
Multifunctional strands in tight junctions.Nature reviews Molecular cell biology, 2(4):285–293, 2001
Shoichiro Tsukita, Mikio Furuse, and Masahiko Itoh. Multifunctional strands in tight junctions.Nature reviews Molecular cell biology, 2(4):285–293, 2001
2001
-
[59]
Physiology and function of the tight junction.Cold Spring Harbor perspectives in biology, 1(2):a002584, 2009
James M Anderson and Christina M Van Itallie. Physiology and function of the tight junction.Cold Spring Harbor perspectives in biology, 1(2):a002584, 2009
2009
-
[60]
Tight junction pore and leak pathways: a dynamic duo.Annual review of physiology, 73(1):283–309, 2011
Le Shen, Christopher R Weber, David R Raleigh, Dan Yu, and Jerrold R Turner. Tight junction pore and leak pathways: a dynamic duo.Annual review of physiology, 73(1):283–309, 2011
2011
-
[61]
Tight junction, selective permeability, and related diseases
Susanne M Krug, J¨ org D Schulzke, and Michael Fromm. Tight junction, selective permeability, and related diseases. InSeminars in cell & developmental biology, volume 36, pages 166–176. Elsevier, 2014
2014
-
[62]
Phase separation of zonula occludens proteins drives formation of tight junctions.Cell, 179(4):923–936, 2019
Oliver Beutel, Riccardo Maraspini, Karina Pombo-Garcia, C´ ecilie Martin-Lemaitre, and Alf Honigmann. Phase separation of zonula occludens proteins drives formation of tight junctions.Cell, 179(4):923–936, 2019
2019
-
[63]
Mobility measurement by analysis of fluorescence photobleaching recovery kinetics.Biophysical journal, 16(9):1055–1069, 1976
Daniel Axelrod, DE Koppel, J Schlessinger, Elliot Elson, and Watt W Webb. Mobility measurement by analysis of fluorescence photobleaching recovery kinetics.Biophysical journal, 16(9):1055–1069, 1976
1976
-
[64]
Organization of vesicular trafficking in epithelia.Nature reviews Molecular cell biology, 6(3):233–247, 2005
Enrique Rodriguez-Boulan, Geri Kreitzer, and Anne M¨ usch. Organization of vesicular trafficking in epithelia.Nature reviews Molecular cell biology, 6(3):233–247, 2005
2005
-
[65]
Two distinct integrin-mediated mechanisms contribute to apical lumen formation in epithelial cells
Satu Marja Myllym¨ aki, Terhi Piritta Ter¨ av¨ ainen, and Aki Manninen. Two distinct integrin-mediated mechanisms contribute to apical lumen formation in epithelial cells. PLoS One, 6(5):e19453, 2011
2011
-
[66]
Development and dynamics of cell polarity at a glance.Journal of cell science, 130(7):1201–1207, 2017
Joseph P Campanale, Thomas Y Sun, and Denise J Montell. Development and dynamics of cell polarity at a glance.Journal of cell science, 130(7):1201–1207, 2017
2017
-
[67]
A molecular switch for the orientation of epithelial cell polarization.Developmental cell, 31(2):171–187, 2014
David M Bryant, Julie Roignot, Anirban Datta, Arend W Overeem, Minji Kim, Wei Yu, Xiao Peng, Dennis J Eastburn, Andrew J Ewald, Zena Werb, et al. A molecular switch for the orientation of epithelial cell polarization.Developmental cell, 31(2):171–187, 2014
2014
-
[68]
A molecular network for de novo generation of the apical surface and lumen.Nature cell biology, 12(11):1035–1045, 2010
David M Bryant, Anirban Datta, Alejo E Rodr´ ıguez- Fraticelli, Johan Per¨ anen, Fernando Mart´ ın-Belmonte, and Keith E Mostov. A molecular network for de novo generation of the apical surface and lumen.Nature cell biology, 12(11):1035–1045, 2010
2010
-
[69]
Developing pressures: fluid forces driving morphogenesis.Current opinion in genetics & development, 32:24–30, 2015
Adam Navis and Michel Bagnat. Developing pressures: fluid forces driving morphogenesis.Current opinion in genetics & development, 32:24–30, 2015
2015
-
[70]
Cell-polarity dynamics controls the mechanism of lumen formation in epithelial morphogenesis.Current Biology, 18(7):507–513, 2008
Fernando Mart´ ın-Belmonte, Wei Yu, Alejo E Rodr´ ıguez- Fraticelli, Andrew Ewald, Zena Werb, Miguel A Alonso, and Keith Mostov. Cell-polarity dynamics controls the mechanism of lumen formation in epithelial morphogenesis.Current Biology, 18(7):507–513, 2008
2008
-
[71]
Gp135/podocalyxin and nherf- 2 participate in the formation of a preapical domain during polarization of mdck cells.The Journal of cell biology, 168(2):303–313, 2005
Doris Meder, Anna Shevchenko, Kai Simons, and Joachim F¨ ullekrug. Gp135/podocalyxin and nherf- 2 participate in the formation of a preapical domain during polarization of mdck cells.The Journal of cell biology, 168(2):303–313, 2005
2005
-
[72]
In vivo coupling of cell elongation and lumen formation in a single cell.Current Biology, 20(4):359–366, 2010
Louis Gervais and Jordi Casanova. In vivo coupling of cell elongation and lumen formation in a single cell.Current Biology, 20(4):359–366, 2010
2010
-
[73]
sma-1 encodes aβh-spectrin homolog required for caenorhabditis elegans morphogenesis.Development, 125(11):2087–2098, 1998
Caroline McKeown, Vida Praitis, and Judith Austin. sma-1 encodes aβh-spectrin homolog required for caenorhabditis elegans morphogenesis.Development, 125(11):2087–2098, 1998
2087
-
[74]
Cystic canal mutants in caenorhabditis elegans are defective in the apical membrane domain of the renal (excretory) cell
Matthew Buechner, David H Hall, Harshida Bhatt, and Edward M Hedgecock. Cystic canal mutants in caenorhabditis elegans are defective in the apical membrane domain of the renal (excretory) cell. Developmental biology, 214(1):227–241, 1999
1999
-
[75]
Mechanisms that regulate adaptor binding toβ-integrin cytoplasmic tails
Kyle R Legate and Reinhard F¨ assler. Mechanisms that regulate adaptor binding toβ-integrin cytoplasmic tails. Journal of cell science, 122(2):187–198, 2009
2009
-
[76]
Levi, Amin S
Boaz P. Levi, Amin S. Ghabrial, and Mark A. Krasnow. Drosophila talin and integrin genes are required for maintenance of tracheal terminal branches and luminal organization.Development, 133(12):2383–2393, 2006
2006
-
[77]
Kif3a guides microtubular dynamics, migration and lumen formation of mdck cells.PloS one, 8(5):e62165, 2013
Christopher Boehlke, Fruzsina Kotsis, Bjoern Buchholz, 39 Christian Powelske, Kai-Uwe Eckardt, Gerd Walz, Roland Nitschke, and E Wolfgang Kuehn. Kif3a guides microtubular dynamics, migration and lumen formation of mdck cells.PloS one, 8(5):e62165, 2013
2013
-
[78]
Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells.Nature genetics, 33(2):129–137, 2003
Surya M Nauli, Francis J Alenghat, Ying Luo, Eric Williams, Peter Vassilev, Xiaogang Li, Andrew EH Elia, Weining Lu, Edward M Brown, Stephen J Quinn, et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells.Nature genetics, 33(2):129–137, 2003
2003
-
[79]
Zonula occludens-1 and-2 regulate apical cell structure and the zonula adherens cytoskeleton in polarized epithelia.Molecular biology of the cell, 23(4):577–590, 2012
Alan S Fanning, Christina M Van Itallie, and James M Anderson. Zonula occludens-1 and-2 regulate apical cell structure and the zonula adherens cytoskeleton in polarized epithelia.Molecular biology of the cell, 23(4):577–590, 2012
2012
-
[80]
Control of cell–cell forces and collective cell dynamics by the intercellular adhesome.Nature cell biology, 17(4):409–420, 2015
Elsa Bazelli` eres, Vito Conte, Alberto Elosegui-Artola, Xavier Serra-Picamal, Mar´ ıa Bintanel-Morcillo, Pere Roca-Cusachs, Jos´ e J Mu˜ noz, Marta Sales-Pardo, Roger Guimer` a, and Xavier Trepat. Control of cell–cell forces and collective cell dynamics by the intercellular adhesome.Nature cell biology, 17(4):409–420, 2015
2015
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.