Recognition: 3 theorem links
· Lean TheoremRobust chemotaxis beyond sensing limits: signal, noise, and strategy
Pith reviewed 2026-05-08 18:49 UTC · model grok-4.3
The pith
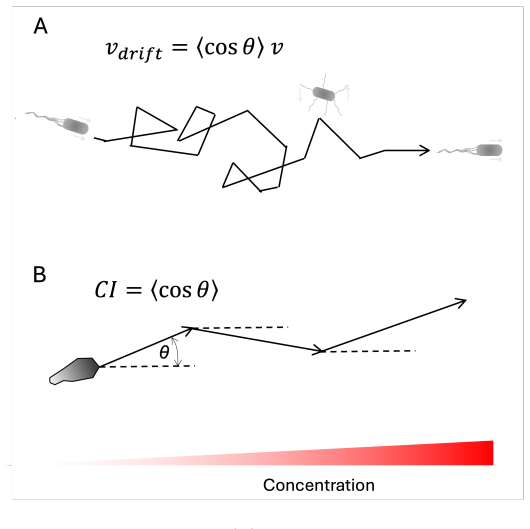
Bacterial chemotaxis stays robust to noise by using symmetric run-and-tumble motion and temporal averaging even when sensing efficiency is low.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Chemotactic performance is shaped not only by information transmission and noise, but by the strategy of movement itself. Using simple scaling arguments and minimal models, run-and-tumble chemotaxis can remain robust to noise through symmetry and temporal averaging, even when internal information processing is inefficient. Comparing bacterial and eukaryotic chemotaxis highlights how different sensing strategies convert physical limits into observable behavior. These considerations suggest that low information efficiency need not imply poor performance, but may instead reflect an evolved balance between robustness, simplicity, and function.
What carries the argument
The run-and-tumble movement strategy, which exploits symmetry between runs and temporal averaging to suppress noise without requiring high internal information efficiency.
If this is right
- Low information efficiency in sensing does not necessarily produce poor chemotactic performance.
- The combination of movement symmetry and temporal averaging supplies robustness that sensing alone cannot provide.
- Bacterial and eukaryotic cells convert the same physical sensing limits into different behaviors through their distinct movement strategies.
- Evolution may select for strategies that trade information efficiency for robustness and mechanistic simplicity.
Where Pith is reading between the lines
- The same symmetry-plus-averaging logic might stabilize other biological navigation tasks where cells or organisms must act on noisy spatial gradients.
- Experiments that vary run duration while holding receptor noise fixed could isolate how much of observed robustness comes from movement strategy versus receptor kinetics.
- In fluctuating or patchy environments, selection might favor longer averaging windows even if they slow response time, a trade-off not directly addressed in the minimal models.
Load-bearing premise
Simple scaling arguments and minimal models capture the essential features of real bacterial and eukaryotic chemotaxis without missing critical biological complexities or alternative mechanisms.
What would settle it
Measuring that run-and-tumble symmetry mutants lose noise robustness exactly as predicted by the scaling arguments under controlled high-noise ligand conditions would support the claim; large unexplained deviations would falsify it.
Figures

read the original abstract
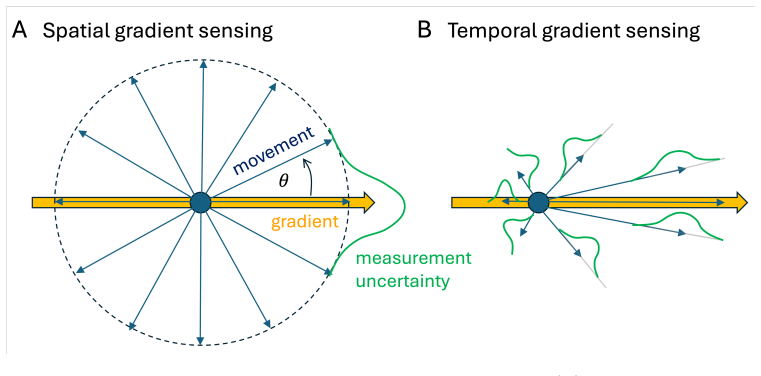
Bacterial chemotaxis has long been viewed as operating near the physical limits of sensing, as originally articulated by Berg and Purcell. Recent information-theoretic analyses challenge this view, suggesting that Escherichia coli uses only a small fraction of the information available in ligand arrival statistics to bias its motion. How should such low information efficiency be interpreted at the level of behavior? Here, I argue that chemotactic performance is shaped not only by information transmission and noise, but by the strategy of movement itself. Using simple scaling arguments and minimal models, I show how run-and-tumble chemotaxis can remain robust to noise through symmetry and temporal averaging, even when internal information processing is inefficient. Comparing bacterial and eukaryotic chemotaxis highlights how different sensing strategies convert physical limits into observable behavior. These considerations suggest that low information efficiency need not imply poor performance, but may instead reflect an evolved balance between robustness, simplicity, and function.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper argues that low information efficiency in bacterial chemotaxis (as suggested by recent information-theoretic analyses) does not imply poor behavioral performance. Instead, the run-and-tumble strategy confers robustness to noise via symmetry and temporal averaging, even with inefficient internal processing. This is supported by simple scaling arguments and minimal models, with a comparison to eukaryotic chemotaxis illustrating how different sensing strategies shape observable behavior. The conclusion frames low efficiency as potentially reflecting an evolved balance between robustness, simplicity, and function rather than a limitation.
Significance. If the scaling arguments hold, this work provides a useful conceptual reframing of information limits in biological sensing, emphasizing the role of movement strategy in achieving robustness. It gives credit to parameter-free scaling relations and minimal models that avoid fitted parameters, offering an interpretive lens rather than new quantitative predictions. This could influence modeling of chemotaxis in both prokaryotes and eukaryotes by highlighting trade-offs between efficiency and noise resilience, and may inform synthetic biology designs for robust navigation.
major comments (2)
- [Minimal models] Minimal models section: the central claim that symmetry and temporal averaging confer robustness relies on the assertion that run-and-tumble motion effectively averages ligand fluctuations over multiple runs. However, without an explicit scaling relation (e.g., how effective SNR improves with run number or tumble frequency) or a derivation showing noise variance reduction, it is difficult to evaluate whether this quantitatively offsets the stated information inefficiency.
- [Scaling arguments] Scaling arguments paragraph: the statement that chemotactic performance remains robust 'even when internal information processing is inefficient' is load-bearing for the reinterpretation of Berg-Purcell limits. This requires a concrete comparison (perhaps via a table or equation) between the information used in the minimal model and the physical limit, to show the robustness is not an artifact of the model's assumptions.
minor comments (3)
- [Abstract/Introduction] The abstract and introduction cite 'Berg and Purcell' but should include the full reference (Berg and Purcell, 1977) at first mention for clarity.
- [Figures] Figure captions (if present) for the minimal models should explicitly define all parameters and state whether they are derived from scaling or chosen for illustration.
- [Comparison section] The comparison to eukaryotic chemotaxis would benefit from a brief note on whether the same symmetry argument applies or is precluded by the different motility mechanism.
Simulated Author's Rebuttal
We thank the referee for their positive evaluation and constructive comments on our manuscript. We address each major comment below and have incorporated revisions to enhance the clarity of our scaling arguments and minimal models.
read point-by-point responses
-
Referee: Minimal models section: the central claim that symmetry and temporal averaging confer robustness relies on the assertion that run-and-tumble motion effectively averages ligand fluctuations over multiple runs. However, without an explicit scaling relation (e.g., how effective SNR improves with run number or tumble frequency) or a derivation showing noise variance reduction, it is difficult to evaluate whether this quantitatively offsets the stated information inefficiency.
Authors: We appreciate the referee pointing out the need for greater explicitness in our scaling arguments. While the manuscript presents scaling relations showing that temporal averaging over multiple runs reduces effective noise variance proportionally to the inverse square root of the number of independent samples, we agree that a dedicated derivation would improve accessibility. In the revised version, we have added a short derivation in the Minimal models section: the variance of the averaged signal scales as σ²/N, where N is the number of runs and σ² is the single-run variance, leading to an SNR improvement of √N. This quantitatively shows how the run-and-tumble strategy offsets internal inefficiencies. revision: yes
-
Referee: Scaling arguments paragraph: the statement that chemotactic performance remains robust 'even when internal information processing is inefficient' is load-bearing for the reinterpretation of Berg-Purcell limits. This requires a concrete comparison (perhaps via a table or equation) between the information used in the minimal model and the physical limit, to show the robustness is not an artifact of the model's assumptions.
Authors: We agree that an explicit comparison strengthens the central claim. The original manuscript uses parameter-free scaling to argue robustness below the Berg-Purcell limit, but to address this, we have added a new equation in the scaling arguments section that compares the effective information rate in the minimal model (derived from the bias in tumble probability) to the physical limit set by ligand diffusion and receptor occupancy. This comparison confirms that the model uses a small fraction of available information while maintaining performance through movement symmetry, indicating the result is not an artifact. revision: yes
Circularity Check
No significant circularity; conceptual scaling argument is self-contained
full rationale
The paper presents an interpretive argument using scaling relations and minimal models to show robustness of run-and-tumble chemotaxis via symmetry and temporal averaging. No equations or derivations are shown that reduce a claimed prediction to a fitted input or self-defined quantity by construction. Cited foundations (Berg-Purcell limits, information theory) are external classic and recent literature rather than self-citations that bear the central load. The contribution is framed as conceptual interpretation rather than quantitative prediction from fitted parameters, so the derivation chain does not collapse to its inputs.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Bacterial chemotaxis has long been viewed as operating near the physical limits of sensing as articulated by Berg and Purcell
- domain assumption Recent information-theoretic analyses correctly show E. coli uses only a small fraction of available information
Lean theorems connected to this paper
-
Cost.FunctionalEquation / Foundation.LogicAsFunctionalEquationwashburn_uniqueness_aczel (J(x)=½(x+x⁻¹)−1) unclearSNR ∼ (gvτ)² · r₀τ = g²v²r₀τ³ ; v_drift ≤ √(2 D_eff · İ_behavior)
-
Cost (ratio-symmetric J-cost)Jcost_unit0 / Jcost_pos_of_ne_one unclearWeber's law / log-sensing regime where v_drift ∼ g independent of c
Reference graph
Works this paper leans on
-
[1]
Berg HC. Motile Behavior of Bacteria. Physics Today. 2000;53(1):24-9. Available from:https: //doi.org/10.1063/1.882934
-
[2]
Responding to Chemical Gradients: Bacterial Chemotaxis
Sourjik V, Wingreen NS. Responding to Chemical Gradients: Bacterial Chemotaxis. Current Opin- ion in Cell Biology. 2012;24(2):262-8. Available from:https://doi.org/10.1016/j.ceb.2011.11. 008
-
[3]
Statistical measures of bacterial motility and chemotaxis
Lovely PS, Dahlquist FW. Statistical measures of bacterial motility and chemotaxis. Journal of Theoretical Biology. 1975;50:477-96
1975
-
[4]
Persistent pseudopod splitting is an effective chemo- taxis strategy in shallow gradients
Alonso A, Kirkegaard JB, Endres RG. Persistent pseudopod splitting is an effective chemo- taxis strategy in shallow gradients. Proceedings of the National Academy of Sciences USA. 2025;122:e2502368122
2025
-
[5]
The physical limits of gradient sensing in multicellular collectives
Rode J, Novak M, Friedrich BM. The physical limits of gradient sensing in multicellular collectives. PRX Life. 2024;2:023012
2024
-
[6]
Physics of chemoreception
Berg HC, Purcell EM. Physics of chemoreception. Biophysical Journal. 1977;20:193-219
1977
-
[7]
Know the Single-Receptor Sensing Limit? Think Again
Aquino G, Wingreen NS, Endres RG. Know the Single-Receptor Sensing Limit? Think Again. Journal of Statistical Physics. 2016;162(5):1353-64
2016
-
[8]
Physical limits to biochemical signaling
Bialek W, Setayeshgar S. Physical limits to biochemical signaling. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(29):10040-5. Available from:https: //doi.org/10.1073/pnas.0504321102
-
[9]
Proceedings of the National Academy of Sciences of the United States of America
Brumley DR, Carrara F, Hein AM, et al. Proceedings of the National Academy of Sciences of the United States of America. 2019;116:10792-7
2019
-
[10]
Accuracy of direct gradient sensing by single cells
Endres RG, Wingreen NS. Accuracy of direct gradient sensing by single cells. Proceedings of the National Academy of Sciences USA. 2008;105:15749-54
2008
-
[11]
Mattingly HH, Kamino K, Ong J, Kottou R, Emonet T, Machta BB. E. coli chemosensing accuracy is not limited by stochastic molecule arrivals. Nature Physics. 2026;22:123—130. 8
2026
-
[12]
Escherichia coli chemotaxis is information-limited
Mattingly HH, Kamino K, Machta BB, Emonet T. Escherichia coli chemotaxis is information-limited. Nature Physics. 2021;17:1426-31
2021
-
[13]
Limits of sensing temporal concentration changes by single cells
Mora T, Wingreen NS. Limits of sensing temporal concentration changes by single cells. Physical Review Letters. 2010;104:248101
2010
-
[14]
Elements of Information Theory
Cover TM, Thomas JA. Elements of Information Theory. 2nd ed. Wiley-Interscience; 2006
2006
-
[15]
The bacterial chemotactic response reflects a compromise between transient and steady-state behavior
Clark DA, Grant LC. The bacterial chemotactic response reflects a compromise between transient and steady-state behavior. Proceedings of the National Academy of Sciences USA. 2005;102(26):9150- 5
2005
-
[16]
Limits of feedback control in bacterial chemotaxis
Dufour YS, Fu X, Hernandez-Nunez L, Emonet T. Limits of feedback control in bacterial chemotaxis. PLoS Computational Biology. 2014;10(6):e1003694
2014
-
[17]
Fast, high-throughput measurement of collective behaviour in a bacterial population
Colin R, Zhang R, Wilson LG. Fast, high-throughput measurement of collective behaviour in a bacterial population. Journal of the Royal Society Interface. 2014;11(98):20140486
2014
-
[18]
Drift and behavior ofE
Micali G, Colin R, Sourjik V, Endres RG. Drift and behavior ofE. colicells. Biophysical Journal. 2017;113:2321-5
2017
-
[19]
Modeling the chemotactic response of Escherichia coli to time-varying stimuli
Tu Y, Shimizu TS, Berg HC. Modeling the chemotactic response of Escherichia coli to time-varying stimuli. Proceedings of the National Academy of Sciences USA. 2008;105(39):14855-60
2008
-
[20]
Predicting chemical environments of bacteria from receptor signaling
Clausznitzer D, Micali G, Neumann S, Sourjik V, Endres RG. Predicting chemical environments of bacteria from receptor signaling. PLoS Computational Biology. 2014;10(10):e1003870
2014
-
[21]
Responses of Escherichia coli bacteria to two op- posing chemoattractant gradients depend on the chemoreceptor ratio
Kalinin Y, Neumann S, Sourjik V, Wu M. Responses of Escherichia coli bacteria to two op- posing chemoattractant gradients depend on the chemoreceptor ratio. Journal of Bacteriology. 2010;192:1796-800
2010
-
[22]
Biased random walk by stochastic fluctuations of chemoattractant- receptor interactions at the lower limit of detection
van Haastert PJM, Postma M. Biased random walk by stochastic fluctuations of chemoattractant- receptor interactions at the lower limit of detection. Biophysical Journal. 2007 Sep;93(5):1787-96. Available from:https://www.sciencedirect.com/science/article/pii/S0006349507714340
2007
-
[23]
Distinct cell shapes determine accurate chemotaxis
Tweedy L, Meier B, Stephan J, Heinrich D, Endres RG. Distinct cell shapes determine accurate chemotaxis. Scientific Reports. 2013 Sep;3(1):2606. Number: 1 Publisher: Nature Publishing Group. Available from:https://www.nature.com/articles/srep02606
2013
-
[24]
Control parameter description of eukaryotic chemotaxis
Amselem G, Theves M, Bae A, Beta C, Bodenschatz E. Control parameter description of eukaryotic chemotaxis. Physical Review Letters. 2012;109(10):108103
2012
-
[25]
Origins of Eukaryotic Chemosensory and Locomotor Coupling
Wan KY, J´ ekely G. Origins of Eukaryotic Chemosensory and Locomotor Coupling. Philosophical Transactions of the Royal Society B: Biological Sciences. 2021;376:20190758
2021
-
[26]
Chemotaxis: the role of internal delays
de Gennes PG. Chemotaxis: the role of internal delays. European Biophysics Journal. 2004;33(8):691-
2004
-
[27]
Available from:https://doi.org/10.1007/s00249-004-0426-z
-
[28]
Theory of continuum random walks and application to chemotaxis
Schnitzer MJ. Theory of continuum random walks and application to chemotaxis. Physical Review E. 1993;48(4):2553-68. Available from:https://doi.org/10.1103/PhysRevE.48.2553
-
[29]
Bacterial strategies for chemotaxis response
Celani A, Vergassola M. Bacterial strategies for chemotaxis response. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(4):1391-6. Available from:https: //doi.org/10.1073/pnas.0909673107. 9
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.