Recognition: 3 theorem links
· Lean TheoremFrom Individual-Based Models to Macroscopic Dynamics of Antimicrobial Resistance
Pith reviewed 2026-05-08 18:23 UTC · model grok-4.3
The pith
Boltzmann equations for infection compartments reduce to coupled Fokker-Planck equations in the grazing limit, revealing stable equilibria driven by interaction rates and antimicrobial misuse.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
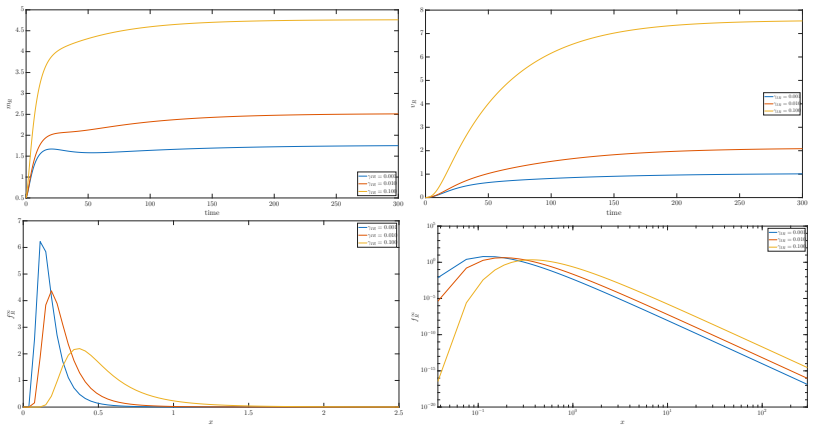
The authors introduce a system of Boltzmann-type equations for the statistical distributions of susceptible, infectious, recovered and resistant individuals, driven by binary susceptible-infectious interactions and supplemented by linear redistribution operators for recovery, resistance development and reinfection. They prove that, in the grazing collision limit, this system is well approximated by coupled Fokker-Planck equations. The limiting description permits explicit characterization of the long-time asymptotics, demonstrating convergence to a stable equilibrium whose resistant subpopulation increases with the intensity of inappropriate antimicrobial use.
What carries the argument
The grazing collision limit applied to Boltzmann-type equations for binary susceptible-infectious interactions, which yields a tractable Fokker-Planck system whose equilibria quantify resistance spread.
If this is right
- Population densities converge to a unique stable equilibrium whose resistant share is an increasing function of inappropriate antimicrobial use.
- Interaction-driven drift terms dominate the long-time dynamics and set the location of the equilibrium.
- The Fokker-Planck reduction supplies explicit formulas for the equilibrium profiles that can be compared with observed resistance fractions.
- Linear redistribution operators for recovery and reinfection modulate but do not override the equilibrium determined by the interaction terms.
Where Pith is reading between the lines
- The same grazing-limit technique could be applied to other compartmental epidemic models that include additional state transitions such as vaccination or treatment failure.
- Equilibrium resistant fractions obtained from the Fokker-Planck system could serve as inputs for cost-benefit calculations of antimicrobial stewardship policies.
- Spatial or network extensions of the kinetic model would test whether local interaction structure alters the global resistance equilibrium predicted here.
Load-bearing premise
Binary encounters between susceptible and infectious individuals are faithfully represented by Boltzmann collision operators, and the grazing limit produces an accurate Fokker-Planck approximation to the full system that includes resistance transitions.
What would settle it
Numerical solutions of the original Boltzmann system and the derived Fokker-Planck system for successively smaller interaction parameters, compared at long times to check whether the resistant fraction converges to the same value within a controlled tolerance.
Figures




read the original abstract
We introduce and discuss a kinetic framework describing the time evolution of the statistical distributions of a population divided into the compartments of susceptible, infectious, recovered, and resistant in the presence of a microbial infection driven by susceptible infectious interactions. Our main objective is to quantify the impact of excessive and inappropriate antimicrobial use, which accelerates the spread of resistance by enabling a fraction of infectious individuals to transition into the resistant compartment. The model consists of a system of Boltzmann type equations capturing binary interactions between susceptible and infectious individuals, complemented by linear redistribution operators that represent recovery, the development of resistance, and reinfection processes. In the grazing collision limit, we show that this Boltzmann system is well approximated by a system of coupled Fokker Planck equations. This limiting description allows for a more tractable analysis of the dynamics, including the characterization of the long-time behavior of the population densities. Our analysis highlights how interaction terms drive the system toward a stable equilibrium and quantifies the effects of inappropriate antimicrobial use on the distribution of resistant individuals. Overall, the results offer a multiscale perspective that bridges kinetic theory with classical epidemic modeling.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript introduces a kinetic framework for antimicrobial resistance using Boltzmann-type equations to model binary susceptible-infectious interactions in an SIRR compartmental system, augmented by linear redistribution operators for recovery, resistance acquisition, and reinfection. It claims that the grazing collision limit yields a well-approximated system of coupled Fokker-Planck equations, which then permits tractable analysis of the dynamics and characterization of long-time equilibria, highlighting the role of interaction terms and inappropriate antimicrobial use in driving resistance distributions.
Significance. If the derivation is complete and all operators are consistently scaled, the work supplies a multiscale bridge from individual-based interactions to macroscopic epidemic dynamics, extending standard kinetic-theory reductions (Boltzmann to Fokker-Planck) to resistance modeling. This offers a parameter-light route to long-time behavior that could complement classical SIR models and quantify misuse effects on resistant subpopulations.
major comments (1)
- [Grazing limit derivation] Grazing-limit derivation (model section and limit procedure): the linear redistribution operators for resistance development, recovery, and reinfection are stated as O(1) complements to the Boltzmann collision terms. The manuscript must explicitly show their scaling with the grazing parameter ε; without rescaling, these terms are either dropped or enter the Fokker-Planck drift/diffusion coefficients incorrectly, which is load-bearing for the claimed long-time equilibrium characterization and the assertion that the FP system faithfully approximates the full SIRR dynamics.
minor comments (2)
- [Abstract] Abstract: the SIRR compartments are referenced but not defined on first use; a brief parenthetical expansion would improve accessibility.
- [Notation] Notation: ensure the population density functions are denoted consistently between the Boltzmann system and the limiting Fokker-Planck equations to avoid reader confusion in the transition.
Simulated Author's Rebuttal
We thank the referee for the detailed and constructive report. The single major comment concerns the explicit scaling of the linear redistribution operators in the grazing-limit derivation. We address this point directly below and will revise the manuscript accordingly to strengthen the rigor of the limiting procedure.
read point-by-point responses
-
Referee: Grazing-limit derivation (model section and limit procedure): the linear redistribution operators for resistance development, recovery, and reinfection are stated as O(1) complements to the Boltzmann collision terms. The manuscript must explicitly show their scaling with the grazing parameter ε; without rescaling, these terms are either dropped or enter the Fokker-Planck drift/diffusion coefficients incorrectly, which is load-bearing for the claimed long-time equilibrium characterization and the assertion that the FP system faithfully approximates the full SIRR dynamics.
Authors: We agree that the scaling must be made fully explicit for the derivation to be complete. In the revised manuscript we will introduce the grazing parameter ε explicitly in the Boltzmann collision operator (standard 1/ε scaling) and state the precise scaling chosen for each linear redistribution operator (recovery, resistance acquisition, and reinfection). We will then carry out the limit procedure term by term, showing how the rescaled linear operators contribute to the drift and diffusion coefficients of the resulting Fokker-Planck system. This will also clarify the regime in which the FP equations faithfully approximate the original SIRR dynamics and support the long-time equilibrium analysis. The changes will be confined to the model section and the grazing-limit subsection; no alterations to the main results or conclusions are required. revision: yes
Circularity Check
No significant circularity; standard grazing-limit reduction from Boltzmann to Fokker-Planck is self-contained
full rationale
The paper's central step is the grazing-collision limit applied to a Boltzmann system augmented by linear redistribution operators for recovery, resistance, and reinfection. This follows the classical kinetic-theory procedure (weak-form integration against test functions, Taylor expansion of the collision kernel to second order, and passage to the limit ε→0) without any parameter fitting to the target macroscopic equilibrium or redefinition of inputs as outputs. The linear operators remain O(1) and are carried unchanged into the Fokker-Planck system; no self-citation is invoked as the sole justification for the limit or for the long-time analysis. The derivation is therefore independent of the result it produces and does not reduce to a tautology or to a fitted input renamed as prediction.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Binary interactions between susceptible and infectious individuals are described by Boltzmann-type equations
- standard math The grazing collision limit yields a valid Fokker-Planck approximation for the full system including recovery, resistance, and reinfection operators
Lean theorems connected to this paper
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclearIn the grazing collision limit, we show that this Boltzmann system is well approximated by a system of coupled Fokker Planck equations.
-
IndisputableMonolith/Foundation/AlphaCoordinateFixation.leanJ_uniquely_calibrated_via_higher_derivative unclearf^q_K(x,t) = ω_K(t)^{ν_K(t)} / Γ(ν_K(t)) · (1/x)^{ν_K+1} exp(-ω_K/x), with ν_K = 1 + 2λ_K/σ_K and ω_K = 2μ_K/σ_K
-
IndisputableMonolith/Foundation/BranchSelection.leanbranch_selection unclearThe model consists of a system of Boltzmann-type equations capturing binary interactions between susceptible and infectious individuals, complemented by linear redistribution operators that represent recovery, the development of resistance, and reinfection processes.
Reference graph
Works this paper leans on
-
[1]
G. Albi, G. Bertaglia, W. Boscheri, G. Dimarco, L. Pareschi, G. Toscani, and M. Zanella. Kinetic modelling of epidemic dynamics: social contacts, control with uncertain data, and multiscale spatial dynamics. In Predicting Pandemics in a Globally Connected World, Volume 1: Toward a Multiscale, Multidisciplinary Framework through Modeling and Sim- ulation, ...
2022
-
[2]
Alexandre and C
R. Alexandre and C. Villani. On the Landau approximation in plasma physics. In Annales de l’Institut Henri Poincaré C, Analyse non linéaire , volume 21, pages 61–95. Elsevier, 2004
2004
-
[3]
Auricchio, G
G. Auricchio, G. Brigati, P. Giudici, and G. Toscani. From kinetic theory to AI: a redis- covery of high-dimensional divergences and their properties. Math. Mod. Meth. Appl. Scie. , 36(06):1185–1233, 2026
2026
-
[4]
Bellomo and M.A.J
N. Bellomo and M.A.J. Chaplain. Predicting Pandemics in a Globally Connected World, Volume 1. Birkhäuser Cham, 2022
2022
-
[5]
Ecological theory suggests that an- timicrobial cycling will not reduce antimicrobial resistance in hospitals
Carl T Bergstrom, Monique Lo, and Marc Lipsitch. Ecological theory suggests that an- timicrobial cycling will not reduce antimicrobial resistance in hospitals. Proceedings of the National Academy of Sciences , 101(36):13285–13290, 2004
2004
-
[6]
Bertaglia, A
G. Bertaglia, A. Bondesan, D. Burini, R. Eftimie, L. Pareschi, and G. Toscani. New trends on the systems approach to modeling SARS-CoV-2 pandemics in a globally connected planet. Math. Mod. Meth Appl. Scie. , 34(11):1995–2054, 2024
1995
-
[7]
Bertaglia, L
G. Bertaglia, L. Pareschi, and G. Toscani. Modelling contagious viral dynamics: a kinetic approach based on mutual utility. Math. Biosci. Eng. , 21(3):4241–4268, 2024
2024
-
[8]
A. C. Birkegård, T. Halasa, N. Toft, A. Folkesson, and K. Græsbøll. Send more data: a systematic review of mathematical models of antimicrobial resistance. Antimicrob. Resist. Infect. Control, 7(1):117, 2018
2018
-
[9]
Bisi and S
M. Bisi and S. Lorenzani. Mathematical models for the large spread of a contact-based infection: a statistical mechanics approach. J. Nonlinear Sci. , 34(5):Paper No. 84, 44, 2024
2024
-
[10]
M. Bisi, G. Spiga, and G. Toscani. Kinetic models of conservative economies with wealth redistribution. Commun. Math. Sci. , 7(4):901–916, 2009
2009
-
[11]
Bolley, J
F. Bolley, J. A. Cañizo, and J. A. Carrillo. Mean-field limit for the stochastic Vicsek model. Appl. Math. Lett. , 25(3):339–343, 2012. 28
2012
-
[12]
Bondesan, M
A. Bondesan, M. Menale, G. Toscani, and M. Zanella. Lotka–Volterra-type kinetic equa- tions for interacting species. Nonlinearity, 38(7):075026, 2025
2025
-
[13]
Evaluating treatment proto- cols to prevent antibiotic resistance
Sebastian Bonhoeffer, Marc Lipsitch, and Bruce R Levin. Evaluating treatment proto- cols to prevent antibiotic resistance. Proceedings of the National Academy of Sciences , 94(22):12106–12111, 1997
1997
-
[14]
Burger, L
M. Burger, L. M. Kreusser, and C. Totzeck. Mean-field optimal control for biological pattern formation. ESAIM Control Optim. Calc. Var. , 27:Paper No. 40, 24, 2021
2021
-
[15]
J. A. Carrillo, M. Fornasier, J. Rosado, and G. Toscani. Asymptotic flocking dynamics for the kinetic Cucker-Smale model. SIAM J. Math. Anal. , 42(1):218–236, 2010
2010
-
[16]
Caudill and J
L. Caudill and J. R Wares. The role of mathematical modeling in designing and evaluating antimicrobial stewardship programs. Curr. Treat. Options Infect. Dis. , 8(2):124–138, 2016
2016
-
[17]
Ciallella, M
A. Ciallella, M. Pulvirenti, and S. Simonella. Inhomogeneities in Boltzmann-SIR models. Math. Mech. Compl. Syst. , 9(3):273–292, 2021
2021
-
[18]
Degond, A
P. Degond, A. Frouvelle, and J.-G. Liu. Macroscopic limits and phase transition in a system of self-propelled particles. J. Nonlinear Sci. , 23(3):427–456, 2013
2013
-
[19]
Degond and S
P. Degond and S. Motsch. Continuum limit of self-driven particles with orientation inter- action. Math. Models Methods Appl. Sci. , 18:1193–1215, 2008
2008
-
[20]
Della Marca, N
R. Della Marca, N. Loy, and A. Tosin. An SIR model with viral load-dependent transmis- sion. J. Math. Biol. , 86(4):61, 2023
2023
-
[21]
Di Francesco and F
M. Di Francesco and F. G. Zefreh. Kermack-McKendrick type models for epidemics with nonlocal aggregation terms. Discrete Contin. Dyn. Syst. , 45(11):4202–4237, 2025
2025
-
[22]
Dimarco, B
G. Dimarco, B. Perthame, G. Toscani, and M. Zanella. Kinetic models for epidemic dy- namics with social heterogeneity. J. Math. Biol. , 83(1):4, 2021
2021
-
[23]
Düring, P
B. Düring, P. Markowich, J.-F. Pietschmann, and M.-T. Wolfram. Boltzmann and Fokker- Planck equations modelling opinion formation in the presence of strong leaders. Proc. R. Soc. Lond. Ser. A Math. Phys. Eng. Sci. , 465(2112):3687–3708, 2009
2009
-
[24]
Düring and M.-T
B. Düring and M.-T. Wolfram. Opinion dynamics: inhomogeneous Boltzmann-type equa- tions modelling opinion leadership and political segregation. Proc. A., 471(2182):20150345, 21, 2015
2015
-
[25]
Furioli, A
G. Furioli, A. Pulvirenti, E. Terraneo, and G. Toscani. Fokker-Planck equations in the modeling of socio-economic phenomena. Math. Mod. Meth. Appl. Scie. , 27(01):115–158, 2017
2017
-
[26]
Grunnill, I
M. Grunnill, I. Hall, and T. Finnie. Check your assumptions: Further scrutiny of basic model frameworks of antimicrobial resistance. J. Theoret. Biol. , 554:111277, 2022
2022
-
[27]
Ha and J.-G
S.-Y. Ha and J.-G. Liu. A simple proof of the Cucker-Smale flocking dynamics and mean- field limit. Commun. Math. Sci. , 7(2):297–325, 2009
2009
-
[28]
S.-Y. Ha, H. Park, and S. Yang. Relaxation dynamics of SIR-flocks with random epidemic states. Commun. Pure Appl. Anal. , 21(11):3887–3918, 2022
2022
-
[29]
Handel, E
A. Handel, E. Margolis, and B. R. Levin. Exploring the role of the immune response in preventing antibiotic resistance. J. Theoret. Biol. , 256(4):655–662, 2009. 29
2009
-
[30]
Hethcote
H.W. Hethcote. The mathematics of infectious diseases. SIAM Rev., 42(4):599–653, 2000
2000
-
[31]
S.H. Ho, D. He, and R. Eftimie. Mathematical models of transmission dynamics and vaccine strategies in Hong Kong during the 2017–2018 winter influenza season. J. Theoret. Biol. , 476:74–94, 2019
2017
-
[32]
Kermack and A.G
W.O. Kermack and A.G. McKendrick. A contribution to the mathematical theory of epidemics. Proc. R. Soc. London A , 115(772):700–721, 1927
1927
-
[33]
Existence and uniqueness of solutions to Fokker–Planck type equations with irregular coefficients
C LeBris and P-L Lions. Existence and uniqueness of solutions to Fokker–Planck type equations with irregular coefficients. Commun. Partial Differ. Equ. , 33(7):1272–1317, 2008
2008
-
[34]
Evolution of antibiotic resistance is linked to any genetic mechanism affecting bacterial duration of carriage
Sonja Lehtinen, François Blanquart, Nicholas J Croucher, Paul Turner, Marc Lipsitch, and Christophe Fraser. Evolution of antibiotic resistance is linked to any genetic mechanism affecting bacterial duration of carriage. Proceedings of the National Academy of Sciences , 114(5):1075–1080, 2017
2017
-
[35]
Lopez-Lozano, D
J.-M. Lopez-Lozano, D. L Monnet, A. Yagüe, A. Burgos, N. Gonzalo, P. Campillos, and M. Saez. Modelling and forecasting antimicrobial resistance and its dynamic relationship to antimicrobial use: a time series analysis. Int. J. Antimicrob. Agents , 14(1):21–31, 2000
2000
-
[36]
Lorenzi, E
T. Lorenzi, E. Paparelli, and A. Tosin. Modelling coevolutionary dynamics in heterogeneous SI epidemiological systems across scales. Commun. Math. Sci. , 22(8):2131–2165, 2024
2024
-
[37]
Martalò, G
G. Martalò, G. Toscani, and M. Zanella. Individual-based foundation of SIR-type epidemic models: mean-field limit and large-time behaviour. Proc. R. Soc. A , 482(2331), 2026
2026
-
[38]
Motsch and E
S. Motsch and E. Tadmor. Heterophilious dynamics enhances consensus. SIAM Rev. , 56(4):577–621, 2014
2014
-
[39]
Mulberry, A
N. Mulberry, A. Rutherford, and C. Colijn. Systematic comparison of coexistence in models of drug-sensitive and drug-resistant pathogen strains. Theor. Popul. Biol. , 133:150–158, 2020
2020
-
[40]
J. O’Neill. Tackling drug-resistant infections globally: final report and recommendations . Government of the United Kingdom, 2016
2016
-
[41]
Opatowski, D
L. Opatowski, D. Guillemot, P.-Y. Boëlle, and L. Temime. Contribution of mathematical modeling to the fight against bacterial antibiotic resistance. Curr. Opin. Infect. Dis. , 24(3):279–287, 2011
2011
-
[42]
Pareschi and G
L. Pareschi and G. Toscani. Interacting Multiagent Systems: Kinetic Equations and Monte Carlo Methods . OUP Oxford, 2013
2013
-
[43]
Pareschi, G
L. Pareschi, G. Toscani, and C. Villani. Spectral methods for the non cut-off boltzmann equation and numerical grazing collision limit. Numer. Math. , 93(3):527–548, 2003
2003
-
[44]
Pareschi and M
L. Pareschi and M. Zanella. Structure preserving schemes for nonlinear Fokker–Planck equations and applications. J. Sci. Comput. , 74(3):1575–1600, 2018
2018
-
[45]
Preziosi, G
L. Preziosi, G. Toscani, and M. Zanella. Control of tumor growth distributions through kinetic methods. J. Theoret. Biol. , 514:110579, 2021
2021
-
[46]
Pulvirenti and S
M. Pulvirenti and S. Simonella. A kinetic model for epidemic spread. Math. Mech. Compl. Syst., 8(3):249–260, 2020. 30
2020
-
[47]
Spicknall, B
I.H. Spicknall, B. Foxman, C.F. Marrs, and J.N.S. Eisenberg. A modeling framework for the evolution and spread of antibiotic resistance: literature review and model categorization. Am. J. Epidemiol. , 178(4):508–520, 2013
2013
-
[48]
Torregrossa and G
M. Torregrossa and G. Toscani. Wealth distribution in presence of debts. A Fokker–Planck description. Commun. Math. Sci. , 16(2):537–560, 2018
2018
-
[49]
Toscani and M
G. Toscani and M. Zanella. On a kinetic description of Lotka–Volterra dynamics. Riv. Mat. Univ. Parma , 15(1):61–77, 2024
2024
-
[50]
Toscani and M
G. Toscani and M. Zanella. Large-time behaviour for coupled systems of Lotka-Volterra- type Fokker-Planck equations. Commun. Pure Appl. Anal. , 2026
2026
-
[51]
R. A. Weinstein, M.J.M. Bonten, D.J. Austin, and M. Lipsitch. Understanding the spread of antibiotic resistant pathogens in hospitals: mathematical models as tools for control. Clin. Infect. Dis. , 33(10):1739–1746, 2001
2001
-
[52]
M. Zanella. Kinetic models for epidemic dynamics in the presence of opinion polarization. Bull. Math. Biol. , 85(5):36, 2023. 31
2023
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.