Recognition: 2 theorem links
· Lean TheoremConcentration-Dependent Membrane Destabilization in DPPC Bilayers: Distinct Insertion Mechanisms and Stress Redistribution by Chloroform and Alkanols
Pith reviewed 2026-05-12 00:57 UTC · model grok-4.3
The pith
Membrane destabilization arises from solute-specific insertion depths, interfacial crowding, and lipid packing changes in DPPC bilayers.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
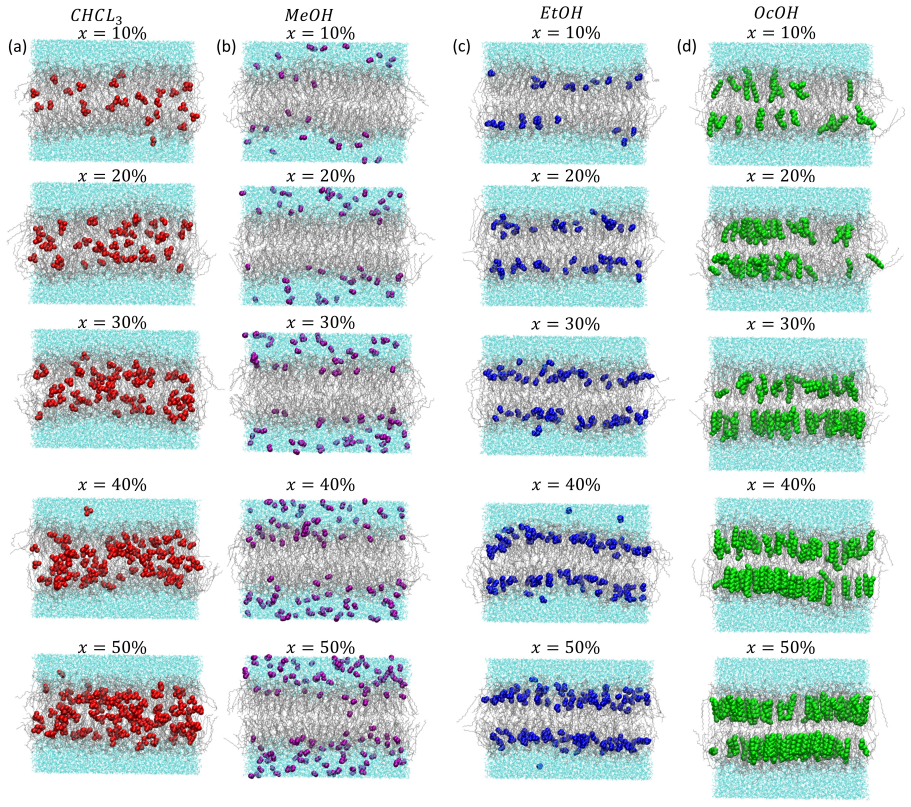
Although complete membrane melting is not observed within 1000 ns, all systems exhibit clear precursors of destabilization, including enhanced thickness fluctuations, reduced lipid order, and mechanical softening. Chloroform induces pronounced thinning and large fluctuations consistent with deep transient insertion, methanol perturbs primarily the headgroup region, ethanol shows intermediate behavior, and octanol preserves thickness but increases fluctuations and interdigitation. Increasing concentration decreases the area compressibility modulus and deuterium order parameter while smoothing lateral pressure profiles, and free-energy analysis shows increased partitioning and reduced barriers
What carries the argument
The interplay of insertion depth, interfacial crowding, and lipid packing disruption, tracked through thickness fluctuations, order parameters, and lateral pressure profiles.
Load-bearing premise
The observed early changes in fluctuations, order, and softening within 1000 ns indicate the path to destabilization even without seeing full melting, and the chosen force fields match real membrane behavior.
What would settle it
Experimental measurements or much longer simulations showing stable membranes without the predicted increases in fluctuations or drops in order at high concentrations would contradict the claim.
Figures







read the original abstract
How do solute concentration and molecular chemistry govern the transition from membrane saturation to destabilization? We address this using microsecond-scale molecular dynamics simulations of dipalmitoylphosphatidylcholine (DPPC) bilayers with chloroform (CHCl$_3$) and a homologous series of alkanols (methanol, ethanol, octanol) over $0-50\%$ concentrations. Although complete membrane melting is not observed within $1000\, ns$, all systems exhibit clear precursors of destabilization, including enhanced thickness fluctuations, reduced lipid order, and mechanical softening. Chloroform induces pronounced thinning and large fluctuations, consistent with deep, transient insertion. Methanol perturbs primarily the headgroup region, while ethanol shows intermediate behavior with partial insertion. Octanol preserves bilayer thickness at high concentrations due to lipid-like insertion but significantly increases fluctuations and interdigitation. Across all systems, increasing concentration decreases the area compressibility modulus and deuterium order parameter, accompanied by smoothing of lateral pressure profiles, indicating stress redistribution. Free energy analysis reveals increased membrane partitioning and reduced translocation barriers with concentration, strongest for octanol and weakest for methanol. These results demonstrate that membrane destabilization is governed by the interplay of insertion depth, interfacial crowding, and lipid packing disruption.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper reports microsecond-scale MD simulations of DPPC bilayers with chloroform and alkanols (methanol, ethanol, octanol) at 0-50% concentrations. While no complete membrane melting occurs within 1000 ns, the authors identify precursors of destabilization (enhanced thickness fluctuations, reduced deuterium order parameters, lowered area compressibility modulus, smoothed lateral pressure profiles) and conclude that destabilization is governed by the interplay of insertion depth, interfacial crowding, and lipid packing disruption, with solute-specific mechanisms supported by free-energy partitioning analysis.
Significance. If the precursor observables can be shown to reliably forecast the transition, the comparative study across insertion depths provides useful mechanistic insight into concentration-dependent membrane perturbation by small molecules. The work supplies concrete data on mechanical softening and stress redistribution that could inform models of anesthetic action or membrane leakage. The absence of an observed transition, however, keeps the governing-factor claim at the level of correlation rather than direct demonstration.
major comments (1)
- [Abstract and concluding discussion] Abstract and concluding discussion: the central claim that 'membrane destabilization is governed by the interplay of insertion depth, interfacial crowding, and lipid packing disruption' rests entirely on interpreting enhanced fluctuations, reduced order, and mechanical softening as faithful precursors. Because the manuscript explicitly states that complete melting is not observed within 1000 ns for any system, the causal mapping from these observables to an actual destabilization transition remains correlative; the same trends could reflect reversible, stable perturbations. A load-bearing revision would require either (a) extended simulations that capture the transition or (b) an explicit test (e.g., order-parameter threshold or fluctuation spectrum) showing that the measured precursors cross into instability.
minor comments (2)
- [Methods] Methods section: ensure that system sizes, lipid-to-solute ratios, force-field parameters (including any modifications to standard DPPC or solute models), equilibration protocols, and statistical error estimation for order parameters and compressibility moduli are reported with sufficient detail for reproducibility.
- [Figures and Results] Figure captions and text: clarify whether the reported thickness fluctuations and lateral-pressure profiles are averaged over the entire trajectory or over equilibrated windows, and indicate the number of independent replicas used for each concentration.
Simulated Author's Rebuttal
We thank the referee for the careful reading and constructive comments on our manuscript. We address the major concern regarding the correlative nature of our claims on membrane destabilization below.
read point-by-point responses
-
Referee: the central claim that 'membrane destabilization is governed by the interplay of insertion depth, interfacial crowding, and lipid packing disruption' rests entirely on interpreting enhanced fluctuations, reduced order, and mechanical softening as faithful precursors. Because the manuscript explicitly states that complete melting is not observed within 1000 ns for any system, the causal mapping from these observables to an actual destabilization transition remains correlative; the same trends could reflect reversible, stable perturbations. A load-bearing revision would require either (a) extended simulations that capture the transition or (b) an explicit test (e.g., order-parameter threshold or fluctuation spectrum) showing that the measured precursors cross into instability.
Authors: We agree that the absence of an observed melting transition within 1000 ns means our mapping from the measured precursors (fluctuations, order parameters, compressibility) to destabilization is correlative rather than directly causal, and that these trends could represent stable perturbations. We will revise the abstract and concluding discussion to replace 'governed by' with 'consistent with an interplay of' to reflect this more precisely. We will also add a new analysis section comparing our fluctuation spectra and order-parameter reductions against literature-reported thresholds for instability in DPPC bilayers. We cannot perform extended simulations to capture the full transition, as 1000 ns already represents the practical limit for the system sizes and concentration series examined. revision: partial
- We are unable to extend the simulations to capture the complete membrane melting transition due to prohibitive computational costs for microsecond-scale runs across the full range of solutes and concentrations.
Circularity Check
No circularity: claims rest on independent simulation observables
full rationale
The manuscript is a molecular-dynamics study that reports numerical observations (thickness fluctuations, deuterium order parameters, area compressibility modulus, lateral pressure profiles, and free-energy profiles) obtained from 1 µs trajectories of DPPC bilayers at varying solute concentrations. No equations, first-principles derivations, or predictions are presented whose outputs are definitionally identical to their inputs. The central statement that destabilization is governed by insertion depth, interfacial crowding, and packing disruption is an interpretive summary of the observed trends, not a self-referential fit or a result forced by prior self-citations. External force fields and standard simulation protocols supply the independent content; no load-bearing step reduces to a renaming, an ansatz smuggled via citation, or a fitted parameter relabeled as a prediction.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Standard assumptions of classical molecular dynamics simulations including periodic boundary conditions, empirical force fields, and finite-size effects being negligible
Lean theorems connected to this paper
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclearAlthough complete membrane melting is not observed within 1000 ns, all systems exhibit clear precursors of destabilization, including enhanced thickness fluctuations, reduced lipid order, and mechanical softening.
-
IndisputableMonolith/Foundation/RealityFromDistinction.leanreality_from_one_distinction unclearIncreasing solute concentration decreases the area compressibility modulus and deuterium order parameter, accompanied by smoothing of lateral pressure profiles.
Reference graph
Works this paper leans on
-
[1]
The membrane actions of anesthetics and tranquilizers
Seeman, P. The membrane actions of anesthetics and tranquilizers. Pharmacol Rev. 1972, 24, 583--655
work page 1972
-
[2]
Turkyilmaz, S.; Chen, W. H.; Mitomo, H.; Regen, S. L. J. Loosening and Reorganization of Fluid Phospholipid Bilayers by Chloroform. J. Am. Chem. Soc. 2009, 131, 5068--5069
work page 2009
-
[3]
Ueda, I.; Suzuki, A. Does pressure antagonize anesthesia? Opposite effects on specific and nonspecific inhibitors of firefly luciferase. Biophys. J. 1998, 75, 1052--1057
work page 1998
-
[4]
Molecular mechanisms of anesthesia
Ueda, I.; Suzuki, A. Molecular mechanisms of anesthesia. Keio J.Med. 2001, 50, 20--25
work page 2001
-
[5]
Partitioning of ethanol in multi-component membranes: Effects on membrane structure
Polley, A.; Vemparala, S. Partitioning of ethanol in multi-component membranes: Effects on membrane structure. Chem Phys Lipids. 2013, 166, 1--11
work page 2013
-
[6]
Polley, A. Partition of common anesthetic molecules in the liquid disordered phase domain of a composite multicomponent membrane. Phys. Rev. E 2018, 98, 012409
work page 2018
-
[7]
Polley, A. Role of chain length in the penetration and clustering dynamics of 1-alkanols in lipid bilayer membranes. Phys. Rev. E 2025, 112, 064405
work page 2025
-
[8]
Rowe, E. S. Lipid chain length and temperature dependence of ethanol-phosphatidylcholine interactions. Biochemistry 1983, 22, 3299--3305
work page 1983
-
[9]
Chin, J. H.; Goldstein, D. B. Drug tolerance in biomembranes: a spin label study of the effects of ethanol. Science 1977, 196, 684--685
work page 1977
-
[10]
Cantor, R. S. The lateral pressure profile in membranes: a physical mechanism of general anesthesia. Biochem. 1997, 36, 2339--2344
work page 1997
-
[11]
Cantor, R. S. Breaking the Meyer--Overton rule: predicted effects of varying stiffness and interfacial activity on the intrinsic potency of anesthetics. Biophysical Journal 2001, 80, 2284--2297
work page 2001
-
[12]
Evans, E.; Needham, D. Physical properties of surfactant bilayer membranes: thermal transitions, elasticity, rigidity, cohesion and colloidal interactions. Journal of Physical Chemistry 1987, 91, 4219--4228
work page 1987
-
[13]
Franks, N. P.; Lieb, W. R. Molecular and cellular mechanisms of general anaesthesia. Nature 1997, 367, 607--614
work page 1997
-
[14]
Franks, N. P.; Lieb, W. R. Selectivity of general anesthetics: A new dimension. Nat. Med. 1997, 3, 377--378
work page 1997
-
[15]
Rowe, E. S. Alcohol Effects on Membrane Lipids. Biochimica et Biophysica Acta (BBA) - Biomembranes 1998, 1376, 219--242
work page 1998
-
[16]
Holte, L. L.; Gawrisch, K. Determining Alcohol Distribution in Lipid Bilayers with ^ 2 H NMR. Biochemistry 1998, 37, 2141--2149
work page 1998
-
[17]
Phase Behavior of Lipid Monolayers with Alcohols
Kranenburg, M.; Smit, B. Phase Behavior of Lipid Monolayers with Alcohols. Journal of Physical Chemistry B 2004, 108, 6479--6489
work page 2004
-
[18]
Roles of bilayer material properties in function and distribution of membrane proteins
McIntosh, T.; Simon, S. Roles of bilayer material properties in function and distribution of membrane proteins. Annu Rev Biophys Biomol Struct 2006, 35, 177--98
work page 2006
-
[19]
C.; McIntosh, T.; Needham, D.; Evans, E
Rawicz, W.; Olbrich, K. C.; McIntosh, T.; Needham, D.; Evans, E. Effect of Chain Length and Unsaturation on Elasticity of Lipid Bilayers. Biophysical Journal 2000, 79, 328--339
work page 2000
-
[20]
Suryabrahmam, B.; Chodnicki, P.; Sappati, S.; Jurkowski, M.; Agrawal, A.; Czub, J.; Raghunathan, V. A. Influence of Alcohols on the Bending Rigidity and the Thickness of Phospholipid Membranes: The Role of Chain Length Mismatch. Journal of Physical Chemistry B 2025, 129, 3988--3997
work page 2025
-
[21]
GROMACS 3.0: a package for molecular simulation and trajectory analysis
Lindahl, E.; Hess, B.; van der Spoel, D. GROMACS 3.0: a package for molecular simulation and trajectory analysis. Journal of Molecular Modeling 2001, 7, 306--11
work page 2001
-
[22]
Scherfeld, D.; Kahya, N.; Schwille, P. Lipid Dynamics and Domain Formation in Model Membranes Composed of Ternary Mixtures of Unsaturated and Saturated Phosphatidylcholines and Cholesterol. Biophys J. 2003, 85, 3758--68
work page 2003
-
[23]
S.; Yin, F.; Mori, M.; Zamorano, A.; Kindt, J
de Joannis, J.; Coppock, P. S.; Yin, F.; Mori, M.; Zamorano, A.; Kindt, J. T. Atomistic simulation of cholesterol effects on miscibility of saturated and unsaturated phospholipids: implications for liquid-ordered/liquid-disordered phase coexistence. J Am Chem Soc. 2011, 133, 3625--34
work page 2011
-
[24]
Atomistic simulations of a multicomponent asymmetric lipid bilayer
Polley, A.; Vemparala, S.; Rao, M. Atomistic simulations of a multicomponent asymmetric lipid bilayer. J Phys Chem B. 2012, 116(45), 13403--10
work page 2012
-
[25]
Tieleman, D. P.; Berendsen, H. J. A molecular dynamics study of the pores formed by Escherichia coli OmpF porin in a fully hydrated palmitoyloleoylphosphatidylcholine bilayer. Biophys J. 1998, 74(6), 2786--15
work page 1998
-
[26]
Niemel\" a , P. S.; Ollila, S.; Hyvönen, M. T.; Karttunen, M.; Vattulainen, I. Assessing the Nature of Lipid Raft Membranes. PLoS Comput Biol. 2007, 3(2), e34
work page 2007
-
[27]
Griepernau, B.; B\'' o ckmann, R. A. The influence of 1-alkanols and external pressure on the lateral pressure profiles of lipid bilayers. Biophys J. 2008, 95, 5766--78
work page 2008
-
[28]
W.; Holopainen, J.; Karttunen, M
Patra, M.; Salonen, E.; Terama, E.; Vattulainen, I.; Faller, R.; Lee, B. W.; Holopainen, J.; Karttunen, M. Under the Influence of Alcohol: The Effect of Ethanol and Methanol on Lipid Bilayers. Biophys. J. 2006, 90, 1121--1135
work page 2006
-
[29]
Influence of Chloroform in Liquid-Ordered and Liquid-Disordered Phases in Lipid Membranes
Reigada, R. Influence of Chloroform in Liquid-Ordered and Liquid-Disordered Phases in Lipid Membranes. J. Phys. Chem. B. 2011, 115, 2527--2535
work page 2011
-
[30]
Reigada, R. Atomistic Study of Lipid Membranes Containing Chloroform: Looking for a Lipid-Mediated Mechanism of Anesthesia. PLoS Comput Biol. 2013, 8, e52631
work page 2013
-
[31]
Polley, A.; Mayor, S.; Rao, M. Bilayer registry in a multicomponent asymmetric membrane: dependence on lipid composition and chain length. J. chem. physics. 2014, 141, 064903
work page 2014
-
[32]
Raghupathy, R.; Anilkumar, A. A.; Polley, A.; Singh, P. P.; Yadav, M.; Johnson, C.; Suryawanshi, S.; Saikam, V.; Sawant, S. D.; Panda, A.; Guo, Z.; Vishwakarma, R. A.; Rao, M.; Mayor, S. Transbilayer lipid interactions mediate nanoclustering of lipid-anchored proteins. Cell 2015, 161 (3), 581--594
work page 2015
-
[33]
A.; Polley, A.; Shabeeb, P.; Raghunathan, V
Kamal, M. A.; Polley, A.; Shabeeb, P.; Raghunathan, V. Closed-loop fluid--fluid immiscibility in binary lipid--sterol membranes. Proceedings of the National Academy of Sciences 2023, 120, e2216002120
work page 2023
-
[34]
PACKMOL: a package for building initial configurations for molecular dynamics simulations
Martínez, L.; Andrade, R.; Birgin, E.; Martínez, J. PACKMOL: a package for building initial configurations for molecular dynamics simulations. J Comput Chem. 2009, 30(13), 2157--64
work page 2009
-
[35]
Patra, M.; Karttunen, M. Lipid Bilayers Driven to a Wrong Lane in Molecular Dynamics Simulations by Subtle Changes in Long-Range Electrostatic Interactions. J. Phys. Chem. B 2004, 108 (14), 4485--9
work page 2004
-
[36]
Hess, B.; Bekker, H.; Berendsen, H. J. C.; Fraaije, J. G. E. M. LINCS: A linear constraint solver for molecular simulations. J Comput Chem. 1997, 18, 1463--9
work page 1997
-
[37]
Miyamoto, S.; Kollman, P. A. Settle: An analytical version of the SHAKE and RATTLE algorithm for rigid water models. J. Comput. Chem. 1992, 13, 952--10
work page 1992
-
[38]
Ollila, O. H. S.; Risselada, H. J.; Louhivuori, M.; Lindahl, E.; Vattulainen, I.; Marrink, S. J. 3D Pressure Field in Lipid Membranes and Membrane-Protein Complexes. Phys. Rev. Lett. 2009, 102, 078101
work page 2009
-
[39]
Chain-Length-Dependent Partitioning of 1-Alkanols in Raft-Like Lipid Membranes
Polley, A. Chain-Length-Dependent Partitioning of 1-Alkanols in Raft-Like Lipid Membranes. arXiv preprint arXiv:2601.18095 2026.,
-
[40]
Phase Segregation of Passive Advective Particles in an Active Medium
Das, A.; Polley, A.; Rao, M. Phase Segregation of Passive Advective Particles in an Active Medium. Phys. Rev. Lett. 2016, 116, 068306
work page 2016
-
[41]
Elastic properties of lipid bilayers: theory and possible experiments
Helfrich, W. Elastic properties of lipid bilayers: theory and possible experiments. Zeitschrift für Naturforschung C 1973, 28, 693--703
work page 1973
-
[42]
Franks, N. P. General anaesthesia: from molecular targets to neuronal pathways of sleep and arousal. Nature Reviews Neuroscience 2008, 9, 370--386
work page 2008
-
[43]
Establishment of the Meyer-Overton correlation in an artificial membrane without protein
Matsumoto, A.; Uesono, Y. Establishment of the Meyer-Overton correlation in an artificial membrane without protein. Biochimica et Biophysica Acta (BBA) - General Subjects 2024, 1868, 130717 mcitethebibliography document
work page 2024
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.