Recognition: 2 theorem links
· Lean TheoremUsing NonTargeted HPV Infections in Studies with Risk Compensation
Pith reviewed 2026-05-13 05:30 UTC · model grok-4.3
The pith
Using nontargeted HPV infections removes both confounding bias and behavioral mediation to isolate the vaccine's direct immunological effect on targeted strains.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
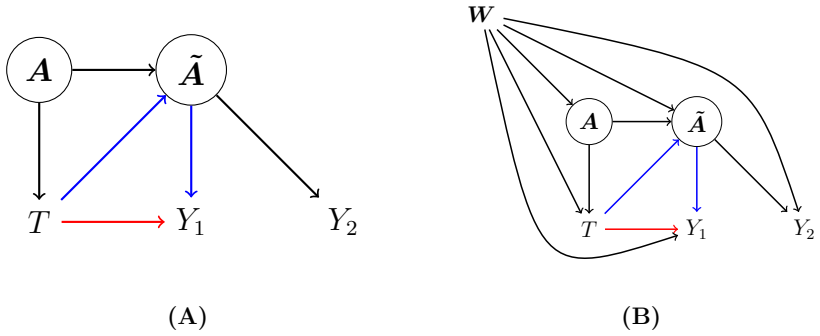
When unmeasured sexual behavior acts as both a confounder and a mediator between vaccination and targeted HPV infections, the quantity estimated using nontargeted HPV infections represents the direct causal effect of the vaccine on targeted strains through immune mechanisms alone, excluding any behavioral component.
What carries the argument
Nontargeted HPV infections used as a reference outcome to isolate the direct effect in a causal model with unmeasured confounding and mediation by sexual behavior.
If this is right
- The method produces an estimate with a clear causal meaning as the direct immunological effect.
- In the presence of risk compensation the estimate suggests higher protection than women actually experience in total.
- Unblinded randomized trials would allow nontargeted infections to isolate the indirect behavioral effect separately from the direct effect.
- Public health evaluations must distinguish the direct immunological quantity from the total effect that includes behavior changes.
Where Pith is reading between the lines
- The same reference-outcome approach could apply to other multi-strain vaccines where behavior change might confound efficacy estimates.
- Routine collection of nontargeted infection data in observational vaccine studies would support better causal claims about direct protection.
- Estimating the full total effect including behavioral responses likely requires different study designs such as blinded trials.
Load-bearing premise
The vaccine has no direct effect on nontargeted HPV strains and the causal structure contains no additional unmeasured paths that would prevent the nontargeted infections from correctly isolating the direct effect.
What would settle it
A controlled study showing that the vaccine directly affects nontargeted strains, or a dataset with measured behavior where the adjusted estimate fails to match the known immunological protection, would disprove the direct-effect interpretation.
Figures




read the original abstract
Studies of HPV vaccine efficacy usually record infections with vaccine targeted and nontargeted strains. Contrary to blinded randomized controlled trials, confounding bias can be a threat and risk compensation may occur in observational studies. Etievant et al. (Biometrics, 2023) proposed to use cervical infections with nontargeted HPV strains to reduce or remove confounding bias of estimates of vaccine efficacy on targeted strains. However, they assumed that vaccinated women could not change their behavior after vaccination. We consider a more plausible setting where unmeasured sexual behavior acts as both a confounder and a mediator, and investigate if the quantity estimated in practice with their method has a clear causal meaning. We demonstrate that using nontargeted HPV infections can remove both confounding bias and the portion of the vaccine effect on the targeted HPV strains that is mediated through the change of behavior. In that case, the estimated quantity has a clear causal interpretation as it represents the direct immunological effect of the vaccine. However, it could be considered misleading from a public health perspective, as in the presence of risk compensation it would suggest higher protection than what women effectively experience. An unblinded randomized controlled trial would allow estimation of the total causal effect of the vaccine, and infections with nontargeted HPV strains could then be used to isolate the indirect behavioral effect of the vaccine.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript extends prior work on using nontargeted HPV infections to adjust for confounding in observational studies of HPV vaccine efficacy. It considers a setting where unmeasured sexual behavior acts as both a confounder and a mediator due to risk compensation after vaccination. The central claim is that the estimator based on nontargeted infections removes both the confounding bias and the mediated behavioral component, yielding a quantity with a causal interpretation as the direct immunological effect of the vaccine on targeted strains (distinct from the total effect that includes behavioral mediation).
Significance. If the identification assumptions hold, the result offers a practical method for obtaining a causally interpretable direct-effect estimate from observational HPV data that would otherwise be biased by unmeasured behavior. This is relevant for vaccine effectiveness studies where RCTs are not feasible, and the paper appropriately flags that the direct effect may overstate the protection experienced by women if risk compensation occurs. The work is grounded in explicit causal modeling rather than post-hoc parameter fitting.
major comments (1)
- [Identification section / causal diagram] The identification result relies on the assumption of no direct vaccine effect on nontargeted strains and the absence of unmeasured paths from behavior to nontargeted infections that bypass the mediator structure. While the abstract and main argument state this, the manuscript would benefit from an explicit sensitivity analysis or bounding exercise showing how violations affect the estimator (e.g., via a simulation or analytic bound in the identification section).
minor comments (2)
- [Abstract] The abstract states the result clearly but could briefly note the key identifying assumptions (no direct effect on nontargeted strains, behavior as sole unmeasured common cause) to help readers assess applicability.
- [Methods / notation] Notation for the direct effect parameter and the adjusted estimator should be introduced with a short table or explicit mapping to the causal diagram for easier reference.
Simulated Author's Rebuttal
We thank the referee for the constructive review and the recommendation for minor revision. We address the single major comment below and will incorporate the suggested addition in the revised manuscript.
read point-by-point responses
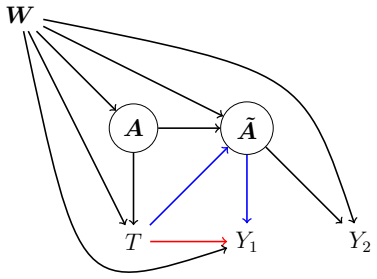
-
Referee: The identification result relies on the assumption of no direct vaccine effect on nontargeted strains and the absence of unmeasured paths from behavior to nontargeted infections that bypass the mediator structure. While the abstract and main argument state this, the manuscript would benefit from an explicit sensitivity analysis or bounding exercise showing how violations affect the estimator (e.g., via a simulation or analytic bound in the identification section).
Authors: We agree that the identification result depends on these assumptions (no direct vaccine effect on nontargeted strains and no unmeasured paths from behavior to nontargeted infections that bypass the mediator). While the manuscript already states the assumptions explicitly, we concur that a sensitivity analysis would strengthen the presentation. In the revised version we will add a brief simulation study to the identification section that quantifies the bias in the estimator under controlled violations of each assumption, thereby providing readers with a practical assessment of robustness. revision: yes
Circularity Check
No significant circularity detected
full rationale
The paper extends a prior causal identification strategy for HPV vaccine efficacy using nontargeted infections to a new setting that includes risk compensation as both confounder and mediator. The central result—that the estimator isolates the direct immunological effect—is obtained by applying standard potential-outcomes or DAG-based identification formulas under explicitly stated assumptions (no direct vaccine effect on nontargeted strains, unmeasured behavior as sole common cause/mediator). These formulas are derived from the model rather than fitted to the same data or defined in terms of the target quantity itself. The citation to Etievant et al. (2023) supplies the baseline method but does not carry the new claim about removal of the mediated behavioral component; that step is shown by direct manipulation of the identification expression. No self-definitional loops, fitted-input predictions, or ansatz smuggling appear in the derivation chain.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Standard causal assumptions including consistency, positivity, and no unmeasured confounding for the paths involving vaccination, behavior, and targeted/nontargeted infections.
Lean theorems connected to this paper
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclearAT E(Y1) = exp(β1) × EA[E{g1(Ã)|A,T=1}]/EA[E{g1(Ã)|A,T=0}], NDE(Y1,T=0)=exp(β1)
Reference graph
Works this paper leans on
-
[1]
Porras C, Sampson JN, Herrero R, Gail MH, Cort´ es B, Hildesheim A, et al. Ratio- nale and design of a double-blind randomized non-inferiority clinical trial to evaluate one or two doses of vaccine against human papillomavirus including an epidemio- logic survey to estimate vaccine efficacy: the Costa Rica ESCUDDO trial. Vaccine. 2022;40(1):76-88
work page 2022
-
[2]
Basu P, Malvi SG, Joshi S, Bhatla N, Muwonge R, Lucas E, et al. Vaccine efficacy against persistent human papillomavirus (HPV) 16/18 infection at 10 years after one, two, and three doses of quadrivalent HPV vaccine in girls in India: a multicentre, prospective, cohort study. The Lancet Oncology. 2021;22(11):1518-29
work page 2021
-
[3]
The impact of residual and unmeasured con- founding in epidemiologic studies: a simulation study
Fewell Z, Davey Smith G, Sterne JA. The impact of residual and unmeasured con- founding in epidemiologic studies: a simulation study. American journal of epidemi- ology. 2007;166(6):646-55
work page 2007
-
[4]
DiClemente RJ. Validity of self-reported sexual behavior among adolescents: where do we go from here? AIDS and Behavior. 2016;20:215-7. 16
work page 2016
-
[5]
M´ endez F, Mu˜ noz N, Posso H, Molano M, Moreno V, Van Den Brule AJ, et al. Cer- vical coinfection with human papillomavirus (HPV) types and possible implications for the prevention of cervical cancer by HPV vaccines. The Journal of infectious diseases. 2005;192(7):1158-65
work page 2005
-
[6]
Chaturvedi AK, Katki HA, Hildesheim A, Rodr´ ıguez AC, Quint W, Schiffman M, et al. Human papillomavirus infection with multiple types: pattern of coinfection and risk of cervical disease. The Journal of infectious diseases. 2011;203(7):910-20
work page 2011
-
[7]
Etievant L, Sampson JN, Gail MH. Increasing efficiency and reducing bias when assessing HPV vaccination efficacy by using nontargeted HPV strains. Biometrics. 2023;79(2):1534-45
work page 2023
-
[8]
Negative controls: a tool for detecting con- founding and bias in observational studies
Lipsitch M, Tchetgen ET, Cohen T. Negative controls: a tool for detecting con- founding and bias in observational studies. Epidemiology. 2010;21(3):383-8
work page 2010
-
[9]
Risk homeostasis theory: an overview
Wilde GJ. Risk homeostasis theory: an overview. Injury prevention. 1998;4(2):89-91
work page 1998
-
[10]
Risky business: safety regulations, risk compensation, and individual behavior
Hedlund J. Risky business: safety regulations, risk compensation, and individual behavior. Injury prevention. 2000;6(2):82-9
work page 2000
-
[11]
Brewer NT, Cuite CL, Herrington JE, Weinstein ND. Risk compensation and vacci- nation: can getting vaccinated cause people to engage in risky behaviors? Annals of Behavioral Medicine. 2007;34(1):95-9
work page 2007
-
[12]
Mullins Kowalczyk TL, Zimet GD, Rosenthal SL, Morrow C, Ding L, Shew M, et al. Adolescent perceptions of risk and need for safer sexual behaviors after first human papillomavirus vaccination. Archives of pediatrics & adolescent medicine. 2012;166(1):82-8. 17
work page 2012
-
[13]
Underhill K. Study designs for identifying risk compensation behavior among users of biomedical HIV prevention technologies: balancing methodological rigor and research ethics. Social science & medicine. 2013;94:115-23
work page 2013
-
[14]
Effect of human papillomavirus vaccination on sexual behaviour among young females
Donken R, Ogilvie GS, Bettinger JA, Sadarangani M, Goldman RD. Effect of human papillomavirus vaccination on sexual behaviour among young females. Canadian Family Physician. 2018;64(7):509-13
work page 2018
-
[15]
Amboree TL, Wermuth PP, Montealegre JR, Fujimoto K, Mgbere O, Darkoh C. Sexual Behaviors and Human Papillomavirus Vaccination in a Heterosexually Active Adult Population at Increased Risk for HIV Infection. Archives of sexual behavior. 2023;52(2):793-801
work page 2023
-
[16]
Identifiability and Exchangeability for Direct and Indirect Effects
Robins JM, Greenland S. Identifiability and Exchangeability for Direct and Indirect Effects. Epidemiology. 1992;3(2):143-55
work page 1992
-
[17]
Pearl J. Direct and Indirect Effects. In: Proceedings of the Seventeenth Conference on Uncertainty in Artificial Intelligence. UAI’01. San Francisco, CA, USA: Morgan Kaufmann Publishers Inc.; 2001. p. 411—420
work page 2001
-
[18]
Alternative analysis of the data from a HPV vaccine study in India
Sasieni P. Alternative analysis of the data from a HPV vaccine study in India. The Lancet Oncology. 2022;23(1):e9
work page 2022
-
[19]
Xu C, Zhang F, Doi SA, Furuya-Kanamori L, Lin L, Chu H, et al. Influence of lack of blinding on the estimation of medication-related harms: a retrospective cohort study of randomized controlled trials. BMC medicine. 2024;22(1):83
work page 2024
-
[20]
Agot KE, Kiarie JN, Nguyen HQ, Odhiambo JO, Onyango TM, Weiss NS. Male circumcision in Siaya and Bondo Districts, Kenya: prospective cohort study to assess 18 behavioral disinhibition following circumcision. JAIDS Journal of Acquired Immune Deficiency Syndromes. 2007;44(1):66-70
work page 2007
-
[21]
Male circumcision for HIV prevention in sub-Saharan Africa: who, what and when? Aids
White RG, Glynn JR, Orroth KK, Freeman EE, Bakker R, Weiss HA, et al. Male circumcision for HIV prevention in sub-Saharan Africa: who, what and when? Aids. 2008;22(14):1841-50
work page 2008
-
[22]
Dema E, Shing JZ, Checchi M, Beddows S, Liu D, Sierra MS, et al. Developing, Validating, and Testing Non-vaccine–Preventable Human Papillomavirus to Control for Differences in Sexual Behavior When Evaluating HPV Vaccination. Cancer Epi- demiology, Biomarkers & Prevention. 2025;34(1):1093-102
work page 2025
-
[23]
Benefit of COVID-19 vaccination accounting for potential risk com- pensation
Ioannidis JP. Benefit of COVID-19 vaccination accounting for potential risk com- pensation. npj Vaccines. 2021;6(1):99
work page 2021
-
[24]
Does vaccination elicit risk compensation? Insights from the COVID-19 pandemic in France
McColl K, Martin-Lapoirie D, Veltri GA, Arwidson P, Raude J. Does vaccination elicit risk compensation? Insights from the COVID-19 pandemic in France. Health Psychology and Behavioral Medicine. 2024;12(1):2287663
work page 2024
-
[25]
COVID-19 vaccine and risk-taking
Smart SJ, Polachek SW. COVID-19 vaccine and risk-taking. Journal of Risk and Uncertainty. 2024;68(1):25-49
work page 2024
-
[26]
Comparative analysis of inflight transmission of SARS- CoV-2, influenza, and SARS-CoV-1
Luo Y, Li Y, Xiao S, Lei H. Comparative analysis of inflight transmission of SARS- CoV-2, influenza, and SARS-CoV-1. Epidemiology & Infection. 2023;151:e111
work page 2023
-
[27]
Generalized estimating equations
Ziegler A. Generalized estimating equations. vol. 204. Springer Science & Business Media; 2011
work page 2011
-
[28]
On summary estimators of relative risk
Tarone RE. On summary estimators of relative risk. Journal of chronic diseases. 1981;34(9-10):463-8. 19
work page 1981
-
[29]
A selective review of negative control methods in epidemiology
Shi X, Miao W, Tchetgen ET. A selective review of negative control methods in epidemiology. Current epidemiology reports. 2020;7:190-202. 20 Relative bias Sample standard Mean sandwich Joint-MH Joint-Reg MH Reg deviation standard error P(Y 1 = 1) (a low, amedium, ahigh) corr(Y 1, Y2) Joint-MH Joint-Reg Joint-MH Joint-Reg 0.021 0.022 1.155 1.151 0.059 0.05...
work page 2020
-
[30]
As a reminder, this can be done by solving forθ= (α ∗ 1, β∗ 1, α∗ 2, β ∗
On the other hand, the approach proposed by [7] (the joint approach with no covariates, Joint- NC) uses data on{T, Y 1, Y2}and consists in jointly estimating E(Y1|T=1) E(Y1|T=0) and E(Y2|T=1) E(Y2|T=0) . As a reminder, this can be done by solving forθ= (α ∗ 1, β∗ 1, α∗ 2, β ∗
-
[31]
the joint estimating equationPn i=1 U(Y 1,i, Y2,i, Ti;θ) = 0, with U(Y 1,i, Y2,i, Ti;θ) = Y1,i−p∗ 1,i 1−p∗ 1,i Ti(Y1,i−p∗ 1,i) 1−p∗ 1,i Y2,i −p ∗ 2,i Ti(Y2,i −p ∗ 2,i) , 26 and wherep ∗ 1,i = exp(α ∗ 1 +β ∗ 1Ti), andp ∗ 2,i = exp(α ∗ 2 +β ∗ 2Ti). This estimating equation is obtained from the log-likelihoods of the models in Equ...
-
[32]
For example, assuming that the observed confounder is univariate and that s∗ 1 :w7→µ ∗ 1w+ν ∗ 1 w2 ands ∗ 2 :w7→µ ∗ 2w+ν ∗ 2 w2, we would solve forθ= (α ∗ 1, β∗ 1, µ∗ 1, ν∗ 1 , α∗ 2, β∗ 2, µ∗ 2, ν∗
-
[33]
the joint estimating equation Pn i=1 U(Y 1,i, Y2,i, Wi, Ti;θ) = 0, with U(Y 1,i, Y2,i, Wi, Ti;θ) = Y1,i−p∗ 1,i 1−p∗ 1,i Ti(Y1,i−p∗ 1,i) 1−p∗ 1,i Wi(Y1,i−p∗ 1,i) 1−p∗ 1,i W 2 i (Y1,i−p∗ 1,i) 1−p∗ 1,i Y2,i −p ∗ 2,i Ti(Y2,i −p ∗ 2,i) Wi(Y2,i −p ∗ 2,i) W 2 i (Y2,i −p ∗ 2,i) , and wherep ∗ 1,i = exp(α...
-
[34]
Again, following [7], this can be done by solving forθ= (β ∗ 1, β∗
On the other hand, the approach proposed by [7] consists in jointly estimatingβ ∗ 1 andβ ∗ 2 with the Mantel-Haenszel (MH) method, and then considering estimateβ ∗ 1 −β ∗ 2 (the joint Mantel-Haenszel approach, Joint-MH). Again, following [7], this can be done by solving forθ= (β ∗ 1, β∗
-
[35]
the joint estimating equationPK k=1 ωk U X1,k, Z1,k, X2,k, Z 2,k;θ = 0 with U(X 1,k, Z1,k, X2,k, Z2,k;θ) = X1,k n1,k −exp(β ∗
-
[36]
Z1,k n0,k X2,k n1,k −exp(β ∗
-
[37]
Z2,k n0,k , andω k = n1,kn0,k nk , and whereX 1,k =Pn i=1 TiY1,iI(W i =w k),Z 1,k =Pn i=1(1−Ti)Y1,iI(W i = wk),X 2,k = Pn i=1 TiY2,iI(W i =w k),Z 2,k = Pn i=1(1−T i)Y2,iI(W i =w k),n 1,k = Pn i=1 TiI(W i =w k),n 0,k =Pn i=1(1−T i)I(W i =w k),n k =Pn i=1 I(W i =w k), withI() the Indicator function. This would lead to ˆβ∗ 1 = log PK k=1 n0,kX1,k nk PK...
-
[38]
Z1,k n0,k 0 0−exp(β ∗
-
[39]
Z2,k n0,k . As a result, if Assumption A 1 and Assumption A 2,W hold under the setting of Figure 1(B)in the Main Document, ˆβ∗ 1 − ˆβ∗ 2 is an unbiased estimate of the log-natural direct effect. Again, the variance estimate can be obtained from adding the variances of ˆβ∗ 1 and ˆβ∗ 2 and subtracting two times their covariance. Finally, ifWonly has a...
work page 2030
-
[40]
named the composite primary outcome. If a model of the form in Equation (1) or in Equation (5) holds separately forY (16) 1 andY (18) 1 but with a common functiong 1, then this is also approximately true forY 1 whenY (16) 1 andY (18) 1 are rare and the vaccine offers identical protection against infection by HPV 16 and infection by HPV 18. Indeed, if E(Y ...
-
[41]
did not provide a rationale for their method. In particular, because the many articles discussing the use of negative control outcomes or proxies of the unmeasured confounders rely on more intricate methods (e.g., [7], and [29]), the approach proposed by [22] prob- ably does not allow for appropriate estimation the causal effect of the HPV vaccine on the ...
-
[42]
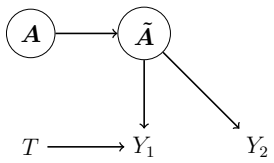
under a setting such as the one in [7] or in the current work. As a preliminary step, we consider the setting without risk compensation of [7] (i.e., when ˜A=A) and when W=∅; see Supplementary Figure 7. More precisely, we assume E(Y1 |A, T) =g 1(A) exp(α1 +β 1T). As a reminder, under such a setting the average total effect ofTonY 1 equals exp(β1). But wit...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.