Recognition: 2 theorem links
· Lean TheoremPoisoning mechanism of ammonia on proton transport and ionomer structure in cathode catalyst layer of PEM fuel cells
Pith reviewed 2026-05-13 04:19 UTC · model grok-4.3
The pith
Ammonia poisons PEM fuel cell catalyst layers when ammonium ions displace hydronium at sulfonic acid sites and amino and imino ions form clusters that absorb additional hydronium ions, reducing proton transport.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
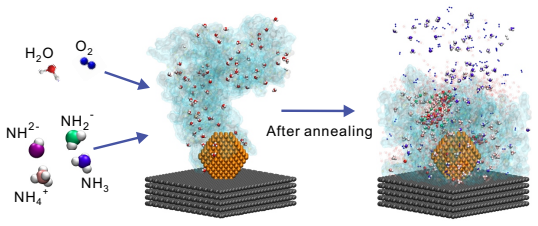
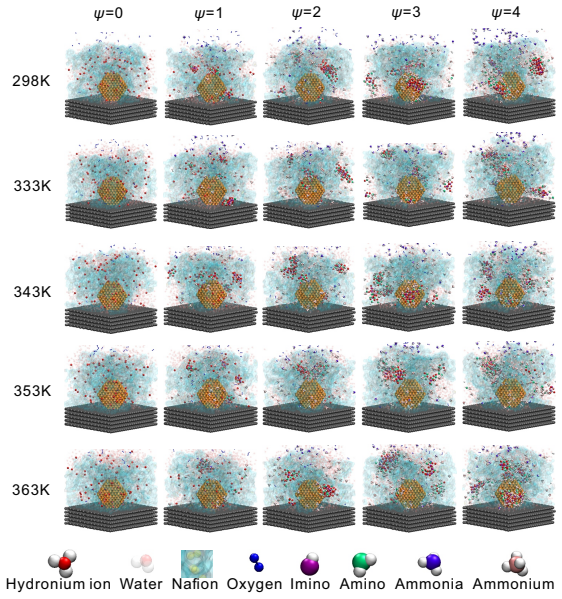
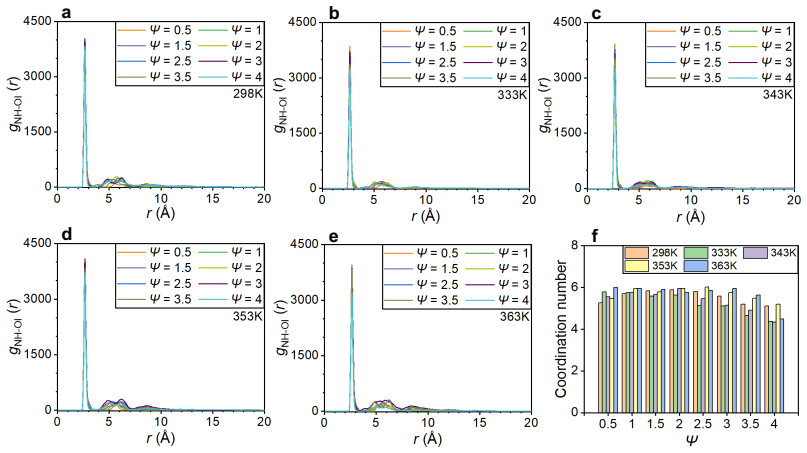
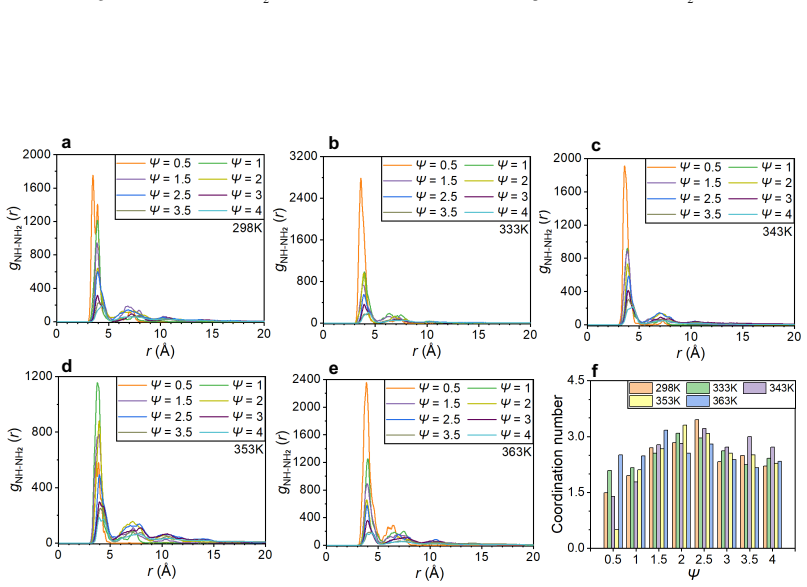
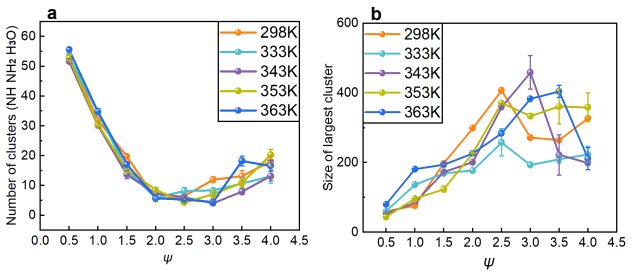
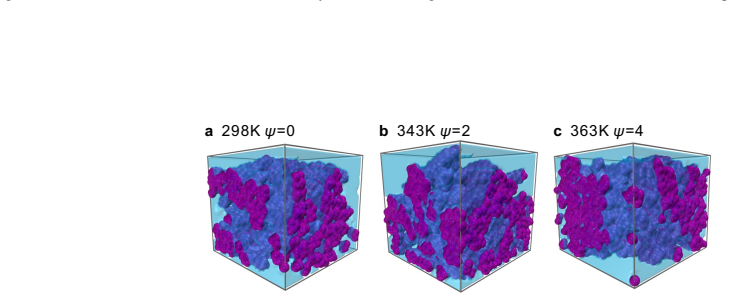
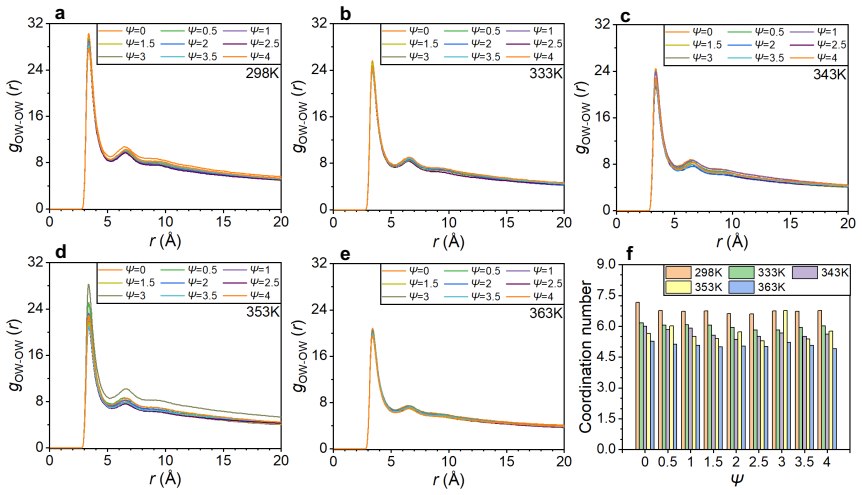
Ammonium can replace the hydronium ions at the charged sites of the sulfonic acid group of the ionomer side chain through van der Waals force and electrostatic interaction. Other ammonia derivatives, amino and imino ions, capture hydronium ions to form ion clusters that have a strong capability to absorb hydronium ions, with structures that change according to ammonia content and temperature due to stable hydrogen bonds within the clusters. These processes reduce the efficiency of proton transport and decrease the catalyst layer's performance. An increase in temperature leads to the dissociation of large ion clusters, alleviating the blockage in the ionomer layer and restoring proton t
What carries the argument
Ion clusters formed by amino and imino ions with hydronium ions, which absorb protons and block transport in the ionomer, along with ammonium adsorption at sulfonic acid sites.
If this is right
- Proton transport efficiency is reduced by the absorption of hydronium ions into the ion clusters.
- The electrochemical performance of the catalyst layer decreases due to impaired proton availability.
- Increasing temperature dissociates the ion clusters and restores proton transport efficiency.
- The degree of poisoning depends on ammonia content and temperature through changes in cluster structure.
Where Pith is reading between the lines
- Designers could target ionomer modifications that reduce the binding strength of ammonium to sulfonic groups to improve poison resistance.
- Short-term operation at higher temperatures might mitigate ammonia effects, though long-term material stability would require separate study.
- Validation through in-situ spectroscopy in fuel cells could confirm cluster formation and guide practical anti-poisoning measures.
Load-bearing premise
The all-atom molecular dynamics simulations accurately capture the interactions and structures that occur during ammonia poisoning in real PEM fuel cell cathode catalyst layers.
What would settle it
If experimental proton conductivity measurements in ammonia-exposed ionomer samples show no temperature-dependent recovery matching the simulated cluster dissociation, or if no evidence of ammonium displacement or cluster formation appears in spectroscopic data, the proposed mechanism would be falsified.
Figures











read the original abstract
Ammonia has strong poisoning effects on cathode catalyst layers of proton exchange membrane (PEM) fuel cells, but the poisoning mechanism is still unclear. In this study, all-atom molecular dynamics simulations are employed to investigate the poisoning mechanisms of ammonia. The results show that ammonium can replace the hydronium ions at the charged sites of sulfonic acid group of the ionomer side chain, and the adsorption of ammonium to sulfonic acid group can be attributed to van der Waals force and electrostatic interaction. Furthermore, other ammonia derivatives, amino and imino ions, can capture hydronium ions to form ion clusters. These ion clusters have strong capability to absorb hydronium ions, and their structures change with ammonia content and temperature. The main mechanism of formation of these clusters is due to the formation of relatively stable hydrogen bonds between ions within the clusters. These mechanisms significantly reduce the efficiency of proton transport, thereby decreasing the catalyst layer's performance in electrochemical reactions. We also discover that the increase in temperature leads to the dissociation of large ion clusters, the blockage in the ionomer layer can be alleviated, and the proton transport efficiency can be restored. The understanding of the poisoning mechanisms obtained in this study is helpful for subsequent research aimed at resolving ammonia poisoning and enhancing the anti-poisoning performance of catalyst layers.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript employs all-atom molecular dynamics simulations to probe the poisoning mechanism of ammonia in the cathode catalyst layer of PEM fuel cells. It reports that ammonium ions displace hydronium at sulfonic acid sites through combined van der Waals and electrostatic interactions, while amino and imino ions form hydrogen-bonded clusters that sequester hydronium, thereby lowering proton transport efficiency. The study further claims that elevated temperature dissociates these clusters, relieving ionomer blockage and restoring transport.
Significance. If the simulated structures and dynamics prove robust, the work supplies atomistic detail on a practically relevant degradation mode in PEM fuel cells and identifies temperature as a potential operational lever for mitigation. The emphasis on competitive ion adsorption and cluster formation aligns with known ionomer chemistry, but the absence of any experimental anchoring or force-field validation keeps the significance provisional rather than definitive.
major comments (3)
- [Methods] Methods section: the force fields employed for NH4+, amino/imino ions, and their interactions with SO3− and H3O+ at λ ≈ 5–10 are not benchmarked against DFT binding energies, experimental diffusion coefficients, or spectroscopic cluster lifetimes. Because the central claims of site replacement and cluster-mediated hydronium sequestration rest directly on these interaction strengths, the lack of validation constitutes a load-bearing uncertainty.
- [Results] Results on proton transport: the efficiency metric is derived solely from vehicular diffusion of intact H3O+ ions. The simulations omit Grotthuss hopping, which dominates proton conduction in hydrated Nafion; consequently the reported reduction in transport cannot be taken as quantitative without additional analysis or justification.
- [Discussion] Discussion of temperature effect: the observed dissociation of large clusters upon heating is presented without sensitivity checks to force-field parameters, cutoff distances, or system size. If these structural changes are force-field artifacts, the claim that temperature restores proton transport efficiency would not hold.
minor comments (2)
- [Abstract] Abstract and introduction: the term “amino and imino ions” is used without explicit structural formulas or pKa context; a brief clarification of the species considered would aid readability.
- Figure captions: several panels lack scale bars or explicit labeling of ion types, making it difficult to correlate visual clusters with the quantitative metrics reported in the text.
Simulated Author's Rebuttal
We thank the referee for the thorough and constructive review of our manuscript on the ammonia poisoning mechanism in PEM fuel cell cathodes. We address each major comment point by point below, providing clarifications and noting revisions made to the manuscript where appropriate.
read point-by-point responses
-
Referee: [Methods] Methods section: the force fields employed for NH4+, amino/imino ions, and their interactions with SO3− and H3O+ at λ ≈ 5–10 are not benchmarked against DFT binding energies, experimental diffusion coefficients, or spectroscopic cluster lifetimes. Because the central claims of site replacement and cluster-mediated hydronium sequestration rest directly on these interaction strengths, the lack of validation constitutes a load-bearing uncertainty.
Authors: We acknowledge the referee's concern regarding force-field validation. The simulations employed standard OPLS-AA parameters for the ions and the Nafion ionomer model, which have been used and partially validated in prior literature for hydrated ionomer systems involving ammonium and hydronium ions. In the revised manuscript, we have expanded the Methods section with additional references to these prior validations and added a dedicated paragraph in the Discussion explicitly acknowledging the absence of new DFT or experimental benchmarks for the specific ion clusters at λ ≈ 5–10. We agree this is a limitation and note that future ab initio work would be valuable, but the current choice is consistent with established practices in the field. revision: partial
-
Referee: [Results] Results on proton transport: the efficiency metric is derived solely from vehicular diffusion of intact H3O+ ions. The simulations omit Grotthuss hopping, which dominates proton conduction in hydrated Nafion; consequently the reported reduction in transport cannot be taken as quantitative without additional analysis or justification.
Authors: We agree that classical all-atom MD captures only vehicular diffusion of intact H3O+ and cannot simulate Grotthuss hopping. Our reported efficiency metric therefore quantifies changes in vehicular transport due to ammonium displacement and cluster sequestration of hydronium. We have revised the Results and Discussion sections to state this limitation explicitly and to note that the observed ion clustering reduces the availability of free hydronium ions, which would impair both vehicular and Grotthuss contributions to conductivity. The qualitative finding that ammonia poisoning lowers proton transport efficiency is retained, with the clarification that the numerical reductions apply specifically to the vehicular component. revision: yes
-
Referee: [Discussion] Discussion of temperature effect: the observed dissociation of large clusters upon heating is presented without sensitivity checks to force-field parameters, cutoff distances, or system size. If these structural changes are force-field artifacts, the claim that temperature restores proton transport efficiency would not hold.
Authors: We concur that additional sensitivity tests on force-field parameters, cutoffs, and system size would strengthen the temperature-dissociation claim. Our original simulations showed consistent cluster breakup across independent trajectories at elevated temperatures. In the revised manuscript, we have added a sentence in the Discussion acknowledging the lack of explicit sensitivity analysis and noting that the observed trend is consistent with experimental reports of partial performance recovery in ammonia-exposed PEM fuel cells at higher operating temperatures. We maintain that the hydrogen-bond-driven cluster mechanism provides a physically plausible explanation, while recognizing this as a computational limitation. revision: partial
Circularity Check
No circularity: results follow directly from MD simulation outputs
full rationale
The paper's central claims rest on all-atom MD trajectories that compute ion positions, hydrogen bonds, and cluster statistics under explicit force fields. No parameter is fitted to the target poisoning metrics and then relabeled as a prediction; no equation defines a quantity in terms of itself; no uniqueness theorem or ansatz is imported via self-citation to close the argument. The observed replacement of H3O+ by NH4+ and temperature-dependent cluster dissociation are direct consequences of the chosen potentials and initial conditions, making the derivation self-contained rather than circular.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Molecular dynamics simulations with standard force fields can model ionomer-ammonia interactions accurately.
Lean theorems connected to this paper
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclearall-atom molecular dynamics simulations... F3C and classical hydronium models... modified DREIDING force field... RDF gNH-OI, coordination numbers, hydrogen bond analysis, cluster analysis, pore size distribution
-
IndisputableMonolith/Foundation/RealityFromDistinction.leanreality_from_one_distinction unclearammonium can replace the hydronium ions at the charged sites... van der Waals force and electrostatic interaction... ion clusters... hydrogen bonds
Reference graph
Works this paper leans on
-
[1]
W. R. W. Daud, R. E. Rosli, E. H. Majlan, S. A. A. Hamid, R. Mohamed, T. Husaini, Pem fuel cell system control: A review, Renewable Energy 113 (2017) 620–638.doi:10.1016/j.renene.2017.06.027
-
[2]
E. Ogungbemi, T. Wilberforce, O. Ijaodola, J. Thompson, A. G. Olabi, Selection of proton exchange membrane fuel cell for transportation, International Journal of Hydrogen Energy 46 (59) (2021) 30625–30640.doi:10. 1016/j.ijhydene.2020.06.147
work page 2021
-
[3]
S. J. Peighambardoust, S. Rowshanzamir, M. Amjadi, Review of the proton exchange membranes for fuel cell applications, International Journal of Hydrogen Energy 35 (2010) 9349–9384.doi:10.1016/j.ijhydene. 2010.05.017
-
[4]
A. B. Stambouli, Fuel cells: The expectations for an environmental-friendly and sustainable source of energy, Renewable & Sustainable Energy Reviews 15 (2011) 4507–4520.doi:10.1016/j.rser.2011.07.100
-
[5]
Y . Sun, S. Polani, F. Luo, S. Ott, P. Strasser, F. Dionigi, Advancements in cathode catalyst and cathode layer design for proton exchange membrane fuel cells, Nature Communications 12 (2021) 5984.doi:10.1038/ s41467-021-25911-x
work page 2021
-
[6]
Y . Wang, Y . Pang, H. Xu, A. Martinez, K. Chen, Pem fuel cell and electrolysis cell technologies and hydrogen infrastructure development: A review, Energy & Environmental Science 15 (2022) 2288–2328.doi:10.1039/ d2ee00790h. 18
work page 2022
-
[8]
J. Yu, C. Su, L. Shang, T. Zhang, Single-atom-based oxygen reduction reaction catalysts for proton exchange membrane fuel cells: Progress and perspective, ACS Nano 17 (20) (2023) 19514–19525.doi:10.1021/ acsnano.3c06522
work page 2023
-
[9]
K. Hu, D. Yang, Studies on the rffects of nh3 in h2 and air on the performance of pemfc, Energies 14 (2021) 6556.doi:10.3390/en14206556
-
[10]
R. Mohtadi, W. k. Lee, J. Van Zee, Assessing durability of cathodes exposed to common air impurities, Journal of Power Sources 138 (2004) 216–225.doi:10.1016/j.jpowsour.2004.06.036
-
[11]
Y . Nagahara, S. Sugawara, K. Shinohara, The impact of air contaminants on pemfc performance and durability, Journal of Power Sources 182 (2) (2008) 422–428.doi:10.1016/j.jpowsour.2007.12.091
-
[12]
B. Yang, Z. Xiang, Nanostructure engineering of cathode layers in proton exchange membrane fuel cells: From catalysts to membrane electrode assembly, ACS Nano 18 (18) (2024) 11598–11630.doi:10.1021/acsnano. 4c01113
-
[13]
B. Shabani, M. Hafttananian, S. Khamani, A. Ramiar, A. A. Ranjbar, Poisoning of proton exchange membrane fuel cells by contaminants and impurities: Review of mechanisms, effects, and mitigation strategies, Journal of Power Sources (2019) 21–48doi:10.1016/j.jpowsour.2019.03.097
-
[14]
J. Zhao, Q. Liang, Y . Liang, Simulation and study of pemfc system directly fueled by ammonia decomposition gas, Frontiers in Energy Research 10 (2022) 819939.doi:10.3389/fenrg.2022.819939
-
[15]
F. A. Uribe, S. Gottesfeld, T. A. Zawodzinski, Effect of ammonia as potential fuel impurity on proton exchange membrane fuel cell performance, Journal of The Electrochemical Society 149 (2002) A293.doi:10.1149/1. 1447221
work page doi:10.1149/1 2002
-
[16]
H. J. Soto, W.-k. Lee, J. W. V . Zee, M. Murthy, Effect of transient ammonia concentrations on pemfc performance, Electrochemical and Solid State Letters 6 (2003) A133.doi:10.1149/1.1574651
-
[17]
Y . Jing, J. Tian, X. Cai, R. Lin, Evolution and mechanism of impedance in pemfc induced by cathode ammonia contamination, Fuel (2024) 130971doi:10.1016/j.fuel.2024.130971
-
[18]
R. Halseid, P. J. S. Vie, R. Tunold, Effect of ammonia on the performance of polymer electrolyte membrane fuel cells, Journal of Power Sources 154 (2006) 343–350.doi:10.1016/j.jpowsour.2005.10.011
-
[19]
D. Imamura, E. Yamaguchi, Effect of air contaminants on the electrolyte degradation in polymer electrolyte membrane fuel cells, ECS Transactions 25 (2009) 813.doi:10.1149/1.3210634
-
[20]
A. Damjanovic, V . Brusic, Electrode kinetics of oxygen reduction on oxide-free platinum electrodes, Electrochimica Acta 12 (6) (1967) 615–628.doi:10.1016/0013-4686(67)85030-8
-
[21]
O. Antoine, Y . Bultel, R. Durand, Oxygen reduction reaction kinetics and mechanism on platinum nanoparticles inside nafion®, Journal of Electroanalytical Chemistry 499 (1) (2001) 85–94.doi:10.1016/S0022-0728(00) 00492-7
-
[22]
R. Halseid, P. J. S. Vie, R. Tunold, Influence of ammonium on conductivity and water content of nafion 117 membranes, Journal of The Electrochemical Society 151 (2004) A381.doi:10.1149/1.1641035
-
[23]
R. Halseid, J. S. Wainright, R. F. Savinell, R. Tunold, Oxidation of ammonium on platinum in acidic solutions, Journal of The Electrochemical Society 154 (2007) B263.doi:10.1149/1.2405851. 19
-
[24]
R. Halseid, M. Heinen, Z. Jusys, R. J. Behm, The effect of ammonium ions on oxygen reduction and hydrogen peroxide formation on polycrystalline pt electrodes, Journal of Power Sources 176 (2008) 435–443.doi: 10.1016/j.jpowsour.2007.08.067
-
[25]
Pattern Recognition 127 (2022), 108611
X. Zhang, U. Pasaogullari, T. M. Molter, Influence of ammonia on membrane-electrode assemblies in polymer electrolyte fuel cells, International Journal of Hydrogen Energy 34 (2009) 9188–9194.doi:10.1016/j. ijhydene.2009.08.099
work page doi:10.1016/j 2009
-
[26]
U. Misz, A. Talke, A. Heinzel, G. Konrad, Sensitivity analyses on the impact of air contaminants on automotive fuel cells, Fuel Cells 16 (4) (2016) 444–462.doi:10.1002/fuce.201500159
-
[27]
W. Guo, D. G. Vlachos, Patched bimetallic surfaces are active catalysts for ammonia decomposition, Nature Communications 6 (2015) 8619.doi:10.1038/ncomms9619
-
[28]
T. Wonglakhon, D. Zahn, Molecular dynamics simulation study of nh4+and nh2- in liquid ammonia: interaction potentials, structural and dynamical properties, Journal of Molecular Modeling 28 (2022) 127.doi:10.1007/ s00894-022-05110-1
work page 2022
-
[29]
K. Jiao, J. Xuan, Q. Du, Z. Bao, B. Xie, B. Wang, Y . Zhao, L. Fan, H. Wang, Z. Hou, S. Huo, N. P. Brandon, Y . Yin, M. D. Guiver, Designing the next generation of proton-exchange membrane fuel cells, Nature 595 (2021) 361 – 369.doi:10.1038/s41586-021-03482-7
-
[30]
Y .-G. Yoon, I. Choi, C.-H. Lee, J. Han, H.-J. Kim, E. Cho, S. Yoo, S. W. Nam, T.-H. Lim, J. Yoon, S. Park, J. Jang, Systematic analysis for the effects of atmospheric pollutants in cathode feed on the performance of proton exchange membrane fuel cells, Bulletin of the Korean Chemical Society 35 (2014) 3475–3481.doi: 10.5012/bkcs.2014.35.12.3475
-
[31]
X. Yuan, H. Li, Y . Yu, M. Jiang, W. Qian, S. Zhang, H. Wang, S. Wessel, T. T. Cheng, Diagnosis of contamination introduced by ammonia at the cathode in a polymer electrolyte membrane fuel cell, International Journal of Hydrogen Energy 37 (2012) 12464–12473.doi:10.1016/j.ijhydene.2012.05.125
-
[32]
C. H. Cheng, K. Malek, P. C. Sui, N. Djilali, Effect of pt nano-particle size on the microstructure of pem fuel cell catalyst layers: Insights from molecular dynamics simulations, Electrochimica Acta 55 (5) (2010) 1588–1597. doi:10.1016/j.electacta.2009.10.030
-
[33]
D. Damasceno Borges, A. A. Franco, K. Malek, G. Gebel, S. Mossa, Inhomogeneous transport in model hydrated polymer electrolyte supported ultrathin films, ACS Nano 7 (8) (2013) 6767–6773.doi:10.1021/nn401624p
-
[34]
L. Chen, X. Xiang, S. Wang, W. Tao, Effects of pt particle on structure and protons transport of nafion membrane, International Journal of Heat and Mass Transfer 148 (2020) 118977.doi:10.1016/j.ijheatmasstransfer. 2019.118977
-
[35]
Y . Huang, P. E. Theodorakis, Z. Zeng, T. Wang, Z. Che, Ionomer structure and component transport in the cathode catalyst layer of pem fuel cells: A molecular dynamics study, The Journal of Chemical Physics 160 (4) (2024) 044910.doi:10.1063/5.0184566
-
[36]
M. Levitt, M. Hirshberg, R. Sharon, K. E. Laidig, V . Daggett, Calibration and testing of a water model for simulation of the molecular dynamics of proteins and nucleic acids in solution, The Journal of Physical Chemistry B 101 (25) (1997) 5051–5061.doi:10.1021/jp964020s
-
[37]
S. J. Seung, T. c. Valeria, M., A. G. William, Nanophase-segregation and transport in nafion 117 from molecular dynamics simulations: Effect of monomeric sequence, The Journal of Physical Chemistry B 108 (10) (2004) 3149–3157.doi:10.1021/jp036842c
-
[38]
S. L. Mayo, B. D. Olafson, W. A. Goddard, Dreiding: a generic force field for molecular simulations, The Journal of Physical Chemistry 94 (26) (1990) 8897–8909.doi:10.1021/j100389a010. 20
-
[39]
T. Mabuchi, T. Tokumasu, Effect of bound state of water on hydronium ion mobility in hydrated nafion using molecular dynamics simulations, The Journal of Chemical Physics 141 (10) (2014) 104904.doi:10.1063/1. 4894813
work page doi:10.1063/1 2014
-
[40]
Q. He, D. C. Joy, D. J. Keffer, Nanoparticle adhesion in proton exchange membrane fuel cell electrodes, Journal of Power Sources 241 (2013) 634–646.doi:10.1016/j.jpowsour.2013.05.011
-
[41]
S. Plimpton, Fast parallel algorithms for short-range molecular dynamics, Journal of Computational Physics 117 (1995) 1–19.doi:10.1006/jcph.1995.1039
-
[42]
L. Fan, K. Wu, C. Tongsh, M. Zhu, X. Xie, K. Jiao, Mechanism of water content on the electrochemical surface area of the catalyst layer in the proton exchange membrane fuel cell, The Journal of Physical Chemistry Letters (2019) 6409–6413doi:10.1021/acs.jpclett.9b02549
-
[43]
L. Fan, Y . Wang, K. Jiao, Oxygen transport routes in ionomer film on polyhedral platinum nanoparticles, ACS Nano 14 (12) (2020) 17487–17495.doi:10.1021/acsnano.0c07856
-
[44]
R. J. Gowers, M. Linke, J. Barnoud, T. Reddy, M. N. Melo, S. L. Seyler, J. J. D. D. L. Dotson, S. B. I. M. Kenney, O. Beckstein, Mdanalysis: A python package for the rapid analysis of molecular dynamics simulations, SciPy (2016).doi:10.25080/majora-629e541a-00e
-
[45]
N. Michaud-Agrawal, E. J. Denning, T. B. Woolf, O. Beckstein, Mdanalysis: a toolkit for the analysis of molecular dynamics simulations, Journal of Computational Chemistry 32 (10) (2011) 2319–2327.doi: 10.1002/jcc.21787
-
[46]
A. Stukowski, Visualization and analysis of atomistic simulation data with ovito–the open visualization tool, Modelling and Simulation in Materials Science and Engineering 18 (2010) 015012.doi:10.1088/ 0965-0393/18/1/015012
work page 2010
-
[47]
M. Pinheiro, R. L. Martin, C. H. Rycroft, A. Jones, E. Iglesia, M. Haranczyk, Journal of Molecular Graphics & Modelling 44 (2013) 208–219.doi:10.1016/j.jmgm.2013.05.007
-
[48]
T. F. Willems, C. H. Rycroft, M. Kazi, J. C. Meza, M. Haranczyk, Algorithms and tools for high-throughput geometry-based analysis of crystalline porous materials, Microporous and Mesoporous Materials 149 (1) (2012) 134–141.doi:10.1016/j.micromeso.2011.08.020. 21
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.