Recognition: 1 theorem link
· Lean TheoremEmpirical scaling laws in balanced networks with conductance-based synapses
Pith reviewed 2026-05-13 02:24 UTC · model grok-4.3
The pith
Recurrent balanced networks with conductance-based synapses and spike correlations produce realistic moderate membrane potential variability.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
In recurrent balanced networks, conductance-based synapse models alone predict unrealistically small membrane potential variability, while current-based models with added spike correlations predict unrealistically large variability. Computer simulations show that including both conductance-based synapses and realistic spike time correlations together produces moderate variability levels consistent with experimental observations.
What carries the argument
The opposing effects on membrane potential variability from conductance-based synaptic inputs and from spike time correlations, which cancel in the balanced recurrent regime.
If this is right
- Both realistic synapse models and spike correlations must be included together to obtain accurate variability predictions.
- The moderate variability arises directly from their interaction rather than from either feature alone.
- This cancellation occurs in strongly coupled, recurrent, balanced networks.
- The same pattern of opposing effects improving realism when combined also appears in feedforward networks.
Where Pith is reading between the lines
- Similar pairs of realistic modeling choices may need to be introduced together in other network regimes to keep dynamics realistic.
- The result suggests a general principle that some biological details only improve model fidelity when their opposing influences are allowed to interact.
- Varying the strength of correlations while keeping conductance-based synapses fixed could provide a direct experimental test of the predicted variability scaling.
Load-bearing premise
The reduction in variability caused by conductance-based synapses will exactly offset the increase caused by spike correlations to produce moderate levels specifically in recurrent balanced networks.
What would settle it
A simulation or in vivo recording in which membrane potential variance remains outside the moderate experimental range even after both conductance-based synapses and measured spike correlations are included.
Figures

read the original abstract
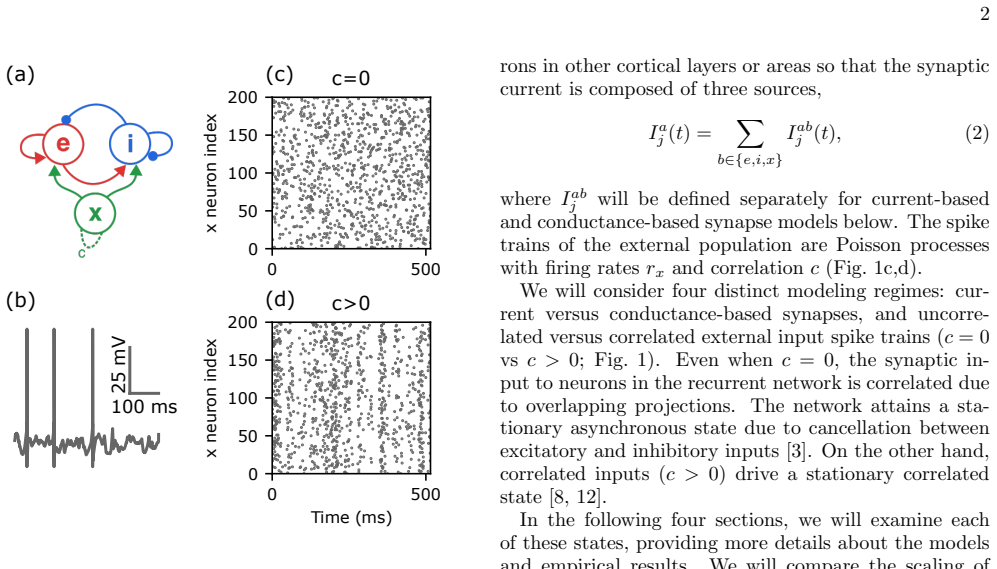
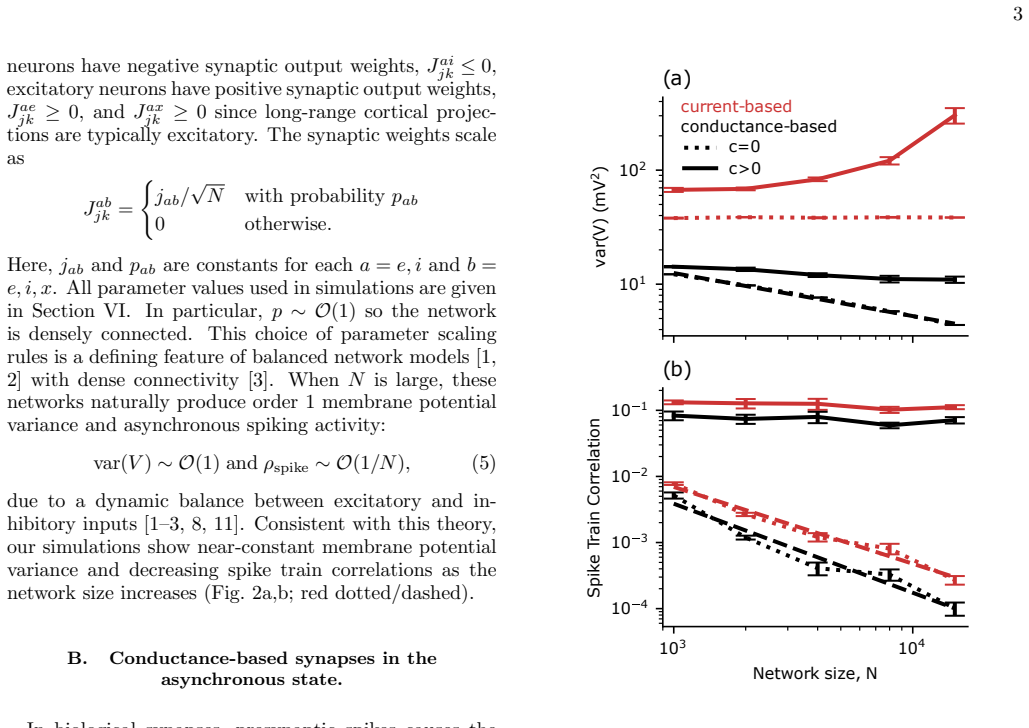
Strongly coupled, recurrent, balanced network models have been successful in describing and predicting many phenomena observed in cortical neural recordings. However, most balanced network models use current-based synapse models in place of more realistic, conductance-based models. Conductance-based synapse models predict unrealistically small membrane potential variability. On the other hand, introducing realistic levels of spike time correlations to models with current-based synapses predicts unrealistically large membrane potential variability. We use computer simulations to show that these two effects can cancel: Recurrent network models with conductance-based synapses and spike time correlations produce more realistic, moderate levels of membrane potential variability. Consistent with recent work on feedforward networks, our results show that including more realistic modeling assumptions produces more realistic dynamics, but only if when two modeling assumptions are included together.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper claims that strongly coupled recurrent balanced networks using conductance-based synapses (which suppress membrane potential variability) combined with realistic spike-time correlations (which inflate it) produce moderate, biologically realistic levels of Vm variability through an empirical cancellation effect observed in computer simulations. This does not occur when either feature is included alone, and the result is presented as extending prior feedforward network findings to the recurrent case.
Significance. If the simulation results hold under broader conditions, the work would indicate that multiple realistic modeling assumptions must be combined to avoid unrealistic extremes in balanced network dynamics, providing guidance for more accurate cortical modeling. The empirical demonstration of cancellation offers a concrete observation, though its status as a generic property of the balanced regime rather than a tuned outcome would need confirmation to strengthen impact.
major comments (3)
- [Results / Simulation methods] The central claim rests on computer simulations whose details (parameter values, how spike correlations are induced in the recurrent network without violating balance, and the mapping between correlation strength and conductance shunting) are not specified, making it impossible to verify whether the moderate-variability regime emerges generically or occupies only a narrow parameter slice (see skeptic note on tuning).
- [Discussion / Theory] No derivation or mean-field analysis from the balance equations is provided to explain the cancellation; the effect is reported purely as an empirical outcome of simulations, leaving open whether it is robust across the balanced regime or dependent on specific choices of correlation level relative to reversal-potential parameters.
- [Abstract / Results] The abstract and summary report no quantitative Vm variance values, error bars, controls, or direct comparisons to experimental data, so the claim that the resulting levels are 'more realistic' and 'moderate' cannot be evaluated for effect size or statistical reliability.
minor comments (2)
- [Abstract] Abstract, final sentence: grammatical error in 'but only if when two modeling assumptions are included together' (should read 'but only when two...').
- [Title / Abstract] Title refers to 'empirical scaling laws' but the abstract focuses exclusively on variability cancellation without mentioning scaling relations or exponents; clarify the connection if scaling is a core contribution.
Simulated Author's Rebuttal
We thank the referee for their thoughtful and constructive comments. We address each major point below and have revised the manuscript to improve reproducibility, add quantitative detail, and expand the discussion of the underlying mechanisms.
read point-by-point responses
-
Referee: [Results / Simulation methods] The central claim rests on computer simulations whose details (parameter values, how spike correlations are induced in the recurrent network without violating balance, and the mapping between correlation strength and conductance shunting) are not specified, making it impossible to verify whether the moderate-variability regime emerges generically or occupies only a narrow parameter slice (see skeptic note on tuning).
Authors: We agree that the original Methods section was insufficiently detailed for full reproducibility. In the revised manuscript we have added a dedicated subsection listing all parameter values (including synaptic conductances, reversal potentials, and network sizes), a precise description of how spike-time correlations are generated via shared presynaptic inputs while preserving the balanced regime (verified by checking that mean excitatory and inhibitory currents remain equal and opposite), and an explicit mapping from correlation coefficient to effective shunting. We also include new supplementary simulations that vary correlation strength and reversal-potential distance over wide ranges, confirming that the moderate-variability regime is robust rather than confined to a narrow slice. revision: yes
-
Referee: [Discussion / Theory] No derivation or mean-field analysis from the balance equations is provided to explain the cancellation; the effect is reported purely as an empirical outcome of simulations, leaving open whether it is robust across the balanced regime or dependent on specific choices of correlation level relative to reversal-potential parameters.
Authors: We acknowledge that a closed-form mean-field derivation would strengthen the theoretical grounding. Deriving an exact analytic expression for the variance cancellation is complicated by the state-dependent conductances and the recurrent feedback loop. In the revised Discussion we therefore supply a qualitative derivation starting from the conductance-based voltage equation under the balanced-state assumption, showing how the reduction in effective time constant (from shunting) offsets the increase in input variance (from correlations). We further demonstrate robustness by reporting results across multiple correlation levels and reversal-potential values, and we cite related mean-field treatments of conductance-based balanced networks to place the empirical finding in context. revision: partial
-
Referee: [Abstract / Results] The abstract and summary report no quantitative Vm variance values, error bars, controls, or direct comparisons to experimental data, so the claim that the resulting levels are 'more realistic' and 'moderate' cannot be evaluated for effect size or statistical reliability.
Authors: We have updated the abstract to include explicit quantitative values: membrane-potential standard deviation of 6.8 ± 0.4 mV (mean ± s.e.m. across 10 independent runs) in the combined model, versus 1.9 ± 0.2 mV with conductance-based synapses alone and 14.2 ± 1.1 mV with current-based synapses plus correlations. A new results figure now shows these values together with experimental benchmarks from cortical intracellular recordings (approximately 4–9 mV). We also added explicit controls confirming that the network remains balanced and that the induced correlations do not violate the mean-current cancellation condition. revision: yes
Circularity Check
No circularity: results are direct simulation outcomes
full rationale
The paper demonstrates its central claim—that conductance-based synapses and spike correlations cancel to yield moderate Vm variability—via direct numerical simulations of recurrent balanced networks. No mathematical derivation, mean-field reduction, or parameter-fitting step is presented that would make the moderate-variability regime equivalent to the model inputs by construction. The result is reported as an observed empirical outcome when both modeling assumptions are included together, with no load-bearing self-citation, self-definitional ansatz, or renaming of known results. The derivation chain is therefore self-contained and independent.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Balanced network regime in which excitation and inhibition are tuned to produce stable asynchronous activity.
Reference graph
Works this paper leans on
-
[1]
C. Van Vreeswijk and H. Sompolinsky, Chaos in neuronal networks with balanced excitatory and inhibitory activ- ity, Science274, 1724 (1996)
work page 1996
-
[2]
C. van Vreeswijk and H. Sompolinsky, Chaotic balanced state in a model of cortical circuits, Neural Comput10, 1321 (1998)
work page 1998
- [3]
-
[4]
J. Barral and A. D Reyes, Synaptic scaling rule preserves excitatory–inhibitory balance and salient neuronal net- work dynamics, Nature neuroscience19, 1690 (2016)
work page 2016
-
[5]
A. Litwin-Kumar and B. Doiron, Slow dynamics and high variability in balanced cortical networks with clustered connections, Nature Neuroscience15, 1498 (2012)
work page 2012
-
[6]
R. Pyle and R. Rosenbaum, Highly connected neurons spike less frequently in balanced networks, Physical Re- view E93, 040302 (2016)
work page 2016
-
[7]
I. D. Landau, R. Egger, V. J. Dercksen, M. Oberlaender, and H. Sompolinsky, The impact of structural hetero- geneity on excitation-inhibition balance in cortical net- works, Neuron92, 1106 (2016)
work page 2016
-
[8]
R. Rosenbaum, M. A. Smith, A. Kohn, J. E. Rubin, and B. Doiron, The spatial structure of correlated neuronal variability, Nature neuroscience20, 107 (2017)
work page 2017
-
[9]
J. J. Pattadkal, G. Mato, C. van Vreeswijk, N. J. Priebe, and D. Hansel, Emergent orientation selectivity from ran- dom networks in mouse visual cortex, Cell reports24, 2042 (2018)
work page 2042
-
[10]
C. Ebsch and R. Rosenbaum, Imbalanced amplification: A mechanism of amplification and suppression from local imbalance of excitation and inhibition in cortical circuits, PLoS computational biology14, e1006048 (2018)
work page 2018
-
[11]
R. Darshan, C. Van Vreeswijk, and D. Hansel, Strength of correlations in strongly recurrent neuronal networks, Physical Review X8, 031072 (2018)
work page 2018
- [12]
- [13]
-
[14]
Y. Shu, A. Hasenstaub, and D. A. McCormick, Turning on and off recurrent balanced cortical activity, Nature 423, 288 (2003)
work page 2003
-
[15]
M. Wehr and A. M. Zador, Balanced inhibition under- lies tuning and sharpens spike timing in auditory cortex, Nature426, 442 (2003)
work page 2003
- [16]
-
[17]
M. Okun and I. Lampl, Instantaneous correlation of exci- tation and inhibition during ongoing and sensory-evoked activities, Nat Neurosci11, 535 (2008)
work page 2008
-
[18]
A. L. Dorrn, K. Yuan, A. J. Barker, C. E. Schreiner, and R. C. Froemke, Developmental sensory experience balances cortical excitation and inhibition, Nature465, 932 (2010)
work page 2010
-
[19]
Y. J. Sun, G. K. Wu, B.-h. Liu, P. Li, M. Zhou, Z. Xiao, H. W. Tao, and L. I. Zhang, Fine-tuning of pre-balanced excitation and inhibition during auditory cortical devel- opment, Nature465, 927 (2010)
work page 2010
-
[20]
M. Zhou, F. Liang, X. R. Xiong, L. Li, H. Li, Z. Xiao, H. W. Tao, and L. I. Zhang, Scaling down of balanced 8 excitation and inhibition by active behavioral states in auditory cortex, Nat Neurosci17, 841 (2014)
work page 2014
-
[21]
P. C. Petersen, M. Vestergaard, K. H. R. Jensen, and R. W. Berg, Premotor spinal network with balanced ex- citation and inhibition during motor patterns has high re- silience to structural division, J Neurosci34, 2774 (2014)
work page 2014
-
[22]
A. D. Reyes, Computing the effects of excitatory- inhibitory balance on neuronal input-output properties, PLOS Computational Biology22, e1013958 (2026)
work page 2026
-
[23]
A. Sanzeni, M. H. Histed, and N. Brunel, Emer- gence of irregular activity in networks of strongly cou- pled conductance-based neurons, Physical Review X12, 011044 (2022)
work page 2022
-
[24]
L. A. Becker, B. Li, N. J. Priebe, E. Seidemann, and T. Taillefumier, Exact analysis of the subthreshold vari- ability for conductance-based neuronal models with syn- chronous synaptic inputs, Physical Review X14, 011021 (2024)
work page 2024
-
[25]
A. Destexhe and D. Par´ e, Impact of Network Activity on the Integrative Properties of Neocortical Pyramidal Neurons In Vivo, Journal of Neurophysiology81, 1531 (1999)
work page 1999
-
[26]
M. R. DeWeese and A. M. Zador, Non-Gaussian Mem- brane Potential Dynamics Imply Sparse, Synchronous Activity in Auditory Cortex, Journal of Neuroscience26, 12206 (2006)
work page 2006
-
[27]
A. A. Faisal, L. P. Selen, and D. M. Wolpert, Noise in the nervous system, Nature reviews neuroscience9, 292 (2008)
work page 2008
-
[28]
B. Haider and D. A. McCormick, Rapid neocortical dy- namics: cellular and network mechanisms, Neuron62, 171 (2009)
work page 2009
-
[29]
A. Y. Tan, Y. Chen, B. Scholl, E. Seidemann, and N. J. Priebe, Sensory stimulation shifts visual cortex from syn- chronous to asynchronous states, Nature509, 226 (2014)
work page 2014
-
[30]
F. R. Fernandez, J. Noueihed, and J. A. White, Voltage- Dependent Membrane Properties Shape the Size But Not the Frequency Content of Spontaneous Voltage Fluctu- ations in Layer 2/3 Somatosensory Cortex, Journal of Neuroscience39, 2221 (2019)
work page 2019
-
[31]
O. Amsalem, H. Inagaki, J. Yu, K. Svoboda, and R. Dar- shan, Sub-threshold neuronal activity and the dynamical regime of cerebral cortex, Nature Communications15, 7958 (2024)
work page 2024
-
[32]
R. Rosenbaum and B. Doiron, Balanced networks of spik- ing neurons with spatially dependent recurrent connec- tions, Physical Review X4, 021039 (2014)
work page 2014
- [33]
-
[34]
M. R. Cohen and A. Kohn, Measuring and interpreting neuronal correlations, Nat Neurosci14, 811 (2011)
work page 2011
-
[35]
M. A. Smith, X. Jia, A. Zandvakili, and A. Kohn, Lami- nar dependence of neuronal correlations in visual cortex, J Neurophysiol109, 940 (2013)
work page 2013
-
[36]
A. S. Ecker, P. Berens, R. J. Cotton, M. Subra- maniyan, G. H. Denfield, C. R. Cadwell, S. M. Smirnakis, M. Bethge, and A. S. Tolias, State dependence of noise correlations in macaque primary visual cortex, Neuron 82, 235 (2014)
work page 2014
-
[37]
M. J. McGinley, M. Vinck, J. Reimer, R. Batista-Brito, E. Zagha, C. R. Cadwell, A. S. Tolias, J. A. Cardin, and D. A. McCormick, Waking state: rapid variations modulate neural and behavioral responses, Neuron87, 1143 (2015)
work page 2015
- [38]
-
[39]
N. Fourcaud-Trocme, D. Hansel, C. van Vreeswijk, and N. Brunel, How spike generation mechanisms determine the neuronal response to fluctuating inputs, Journal of Neuroscience23, 11628 (2003)
work page 2003
-
[40]
A. Kohn, R. Coen-Cagli, I. Kanitscheider, and A. Pouget, Correlations and Neuronal Population Information, An- nual Review of Neuroscience39, 237 (2016)
work page 2016
- [41]
-
[42]
C. F. Stevens and A. M. Zador, Input synchrony and the irregular firing of cortical neurons, Nature neuroscience 1, 210 (1998)
work page 1998
-
[43]
E. Salinas and T. J. Sejnowski, Impact of correlated synaptic input on output firing rate and variability in simple neuronal models, Journal of neuroscience20, 6193 (2000)
work page 2000
-
[44]
J. J. Pattadkal, R. T. O’Shea, D. Hansel, T. Taillefumier, D. H. Brager, and N. J. Priebe, Synchrony timescales underlie irregular neocortical spiking, Neuron114, 724 (2026)
work page 2026
-
[45]
M. N. Shadlen and W. T. Newsome, The variable dis- charge of cortical neurons: implications for connectivity, computation, and information coding, Journal of neuro- science18, 3870 (1998)
work page 1998
-
[46]
R. Rosenbaum, J. Trousdale, and K. Josic, Pooling and correlated neural activity, Frontiers in computational neuroscience4, 1209 (2010)
work page 2010
-
[47]
J. De La Rocha, B. Doiron, E. Shea-Brown, K. Josi´ c, and A. Reyes, Correlation between neural spike trains increases with firing rate, Nature448, 802 (2007)
work page 2007
-
[48]
E. Shea-Brown, K. Josi´ c, J. De La Rocha, and B. Doiron, Correlation and synchrony transfer in integrate-and-fire neurons:¡? format?¿ basic properties and consequences for coding, Physical review letters100, 108102 (2008)
work page 2008
-
[49]
R. Rosenbaum and K. Josi´ c, Mechanisms that modulate the transfer of spiking correlations, Neural computation 23, 1261 (2011)
work page 2011
- [50]
-
[51]
A. Destexhe, M. Rudolph, and D. Par´ e, The high- conductance state of neocortical neurons in vivo, Nature reviews neuroscience4, 739 (2003)
work page 2003
- [52]
-
[53]
N. Brunel, Dynamics of Sparsely Connected Networks of Excitatory and Inhibitory Spiking Neurons, Journal of Computational Neuroscience8, 183 (2000)
work page 2000
-
[54]
A. O. Teasley and G. K. Ocker, Field-Theoretic Approach to Compartmental Neuronal Networks: Impact of Den- dritic Calcium Spike-Dependent Bursting, PRX Life4, 013016 (2026)
work page 2026
-
[55]
L. A. Becker, F. Baccelli, and T. Taillefumier, Subthresh- old moment analysis of neuronal populations driven by synchronous synaptic inputs, PLOS Computational Bi- 9 ology21, e1013645 (2025)
work page 2025
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.