Recognition: 2 theorem links
· Lean TheoremOptimal Scheduling of Dengue Vector Control
Pith reviewed 2026-05-13 02:49 UTC · model grok-4.3
The pith
Optimally timed combinations of dengue vector controls substantially suppress transmission risk in seasonal temperature models.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
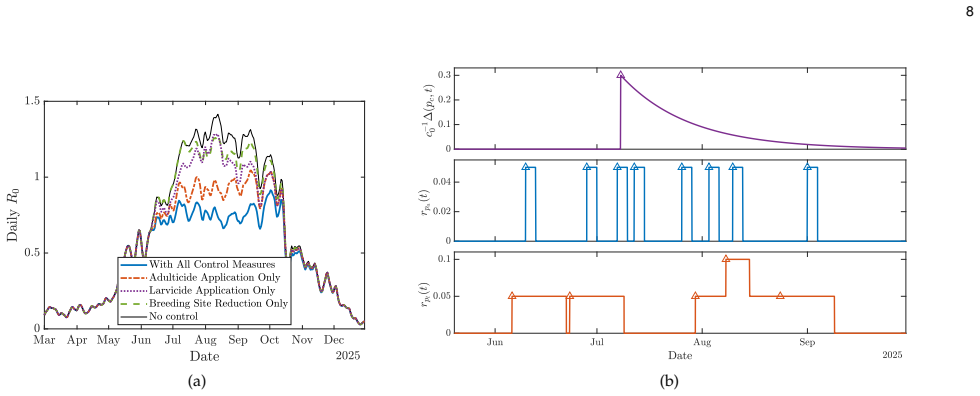
By extending the non-Markovian mechanistic Aedes life-cycle model with intervention-specific temporal profiles and applying an adjoint-based gradient descent procedure to minimize the time-dependent dengue reproduction number R0, the resulting optimal schedules for combined larvicide, adulticide, and breeding-site reduction produce substantial suppression of transmission risk, with the degree of suppression strongly modulated by seasonal temperature variation and the length of each intervention; the same optimization can be placed inside a model predictive control architecture to enable closed-loop, surveillance-driven vector management.
What carries the argument
Adjoint-based gradient descent optimization of intervention temporal profiles to minimize the time-dependent basic reproduction number R0 inside an extended high-fidelity non-Markovian stage-structured temperature-dependent Aedes aegypti life-cycle model.
If this is right
- Appropriately timed sequences of multiple interventions lower transmission risk more effectively than individual or untimed measures.
- Seasonal temperature fluctuations strongly determine the optimal windows and overall effectiveness of each control type.
- Longer intervention durations increase the achievable level of risk reduction for a given schedule.
- Placing the optimizer inside a model predictive control loop produces closed-loop schedules that adapt to real-time surveillance and uncertainty.
Where Pith is reading between the lines
- The same timing-optimization approach could be repurposed for other Aedes-borne diseases that share the same vector.
- Feeding climate-change temperature projections into the model could reveal how optimal control windows shift in future decades.
- Direct comparison of model-predicted population reductions against outcomes from small-scale field trials would test whether the optimization translates to operational settings.
Load-bearing premise
The high-fidelity non-Markovian mechanistic model of the Aedes life cycle, once extended with control effects, accurately represents real mosquito population dynamics under the temperature regimes and interventions considered.
What would settle it
Field measurements of Aedes aegypti population densities and dengue incidence in Miami after deployment of the computed optimal intervention schedule that show no substantial reduction relative to baseline or that deviate markedly from the simulated trajectories would falsify the claim that the optimized timings suppress risk.
Figures



read the original abstract
Dengue transmission is shaped by the population dynamics of the Aedes aegypti mosquito, making vector control a central strategy for disease mitigation. The impact of interventions such as larvicide, adulticide, and breeding-site reduction depends critically on their timing under fluctuating environmental conditions. We build on a high-fidelity, non-Markovian mechanistic model of the Aedes life cycle that captures stage-structured, temperature-dependent developmental delays, and mortality, and extend it to incorporate multiple vector control measures. Rather than using continuous abstract control amplitudes as in standard optimal control formulations, we introduce intervention-specific temporal profiles that better reflect operational practice. We then develop an adjoint-based gradient descent framework to compute the optimal timing of a sequence of interventions by minimizing the time-dependent dengue reproduction number, R0. Numerical simulations based on seasonal temperature data from Miami, Florida, show that appropriately timed combinations of interventions can substantially suppress transmission risk, with outcomes strongly influenced by seasonal temperature variation and intervention duration. We further propose embedding the resulting optimization framework within a Model Predictive Control architecture, yielding a closed-loop approach for real-time, surveillance-driven vector management under environmental and operational uncertainty.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript constructs a high-fidelity non-Markovian stage-structured model of the Aedes aegypti life cycle that incorporates temperature-dependent developmental delays and mortality rates. It extends the model to include three vector-control interventions (larvicide, adulticide, and breeding-site reduction) with operationally realistic temporal profiles rather than abstract continuous controls. An adjoint-based gradient-descent procedure is derived to optimize the timing of a sequence of interventions by minimizing a time-dependent basic reproduction number R0(t). Numerical simulations driven by seasonal temperature records from Miami, Florida, are used to illustrate that suitably timed intervention combinations can substantially lower transmission risk; the outcomes are shown to depend on seasonal temperature variation and intervention duration. The optimization framework is additionally embedded in a Model Predictive Control architecture to enable closed-loop, surveillance-driven management under uncertainty.
Significance. If the numerical results are reproducible and the underlying model is representative, the work supplies a mathematically systematic method for scheduling dengue vector interventions that respects operational constraints and environmental seasonality. The adjoint derivation for gradient-based minimization on a detailed mechanistic model is a technical contribution that could be reused in related vector-borne disease problems. The MPC embedding addresses practical needs for adaptive control. Significance is limited, however, by the absence of direct quantitative comparisons to standard or baseline schedules and by the lack of any empirical validation of the mosquito model against field observations.
major comments (2)
- [Numerical Experiments] Numerical Experiments section: the reported suppression levels are presented without any comparison to baseline intervention schedules (constant-rate application, random timing, or no intervention). Without such controls it is impossible to determine whether the optimized timing yields a genuine improvement over conventional practice or merely reflects the chosen objective and model structure.
- [Model formulation] Model formulation and parameter section: the non-Markovian stage-structured Aedes model relies on literature-derived parameters with no calibration or validation against Aedes population time series from Miami or comparable subtropical sites. Because the central claim rests on the quantitative outcomes of this specific model, the lack of empirical grounding is load-bearing for the assertion that the optimized schedules will suppress transmission risk in the field.
minor comments (2)
- [Adjoint derivation] The notation for the time-dependent R0(t) and its adjoint equations should be introduced with a clear statement of the underlying assumptions (e.g., constant human population, no spatial heterogeneity).
- [Figures] Figure captions for the Miami temperature-driven simulations should explicitly state the intervention durations, amplitudes, and number of applications used in each scenario.
Simulated Author's Rebuttal
We thank the referee for the detailed and constructive report. We address each major comment below and indicate the revisions we will make to strengthen the manuscript.
read point-by-point responses
-
Referee: [Numerical Experiments] Numerical Experiments section: the reported suppression levels are presented without any comparison to baseline intervention schedules (constant-rate application, random timing, or no intervention). Without such controls it is impossible to determine whether the optimized timing yields a genuine improvement over conventional practice or merely reflects the chosen objective and model structure.
Authors: We agree that explicit comparisons to baseline schedules are necessary to demonstrate the practical advantage of the optimized timings. In the revised manuscript we will add a new set of numerical experiments that apply the same objective function and Miami temperature forcing to three baselines: (i) constant-rate intervention at the mean intensity used in the optimized case, (ii) randomly timed interventions with the same total effort, and (iii) no intervention. The resulting R0(t) trajectories and cumulative transmission risk will be reported side-by-side with the optimized schedules so that the improvement attributable to timing can be quantified directly. revision: yes
-
Referee: [Model formulation] Model formulation and parameter section: the non-Markovian stage-structured Aedes model relies on literature-derived parameters with no calibration or validation against Aedes population time series from Miami or comparable subtropical sites. Because the central claim rests on the quantitative outcomes of this specific model, the lack of empirical grounding is load-bearing for the assertion that the optimized schedules will suppress transmission risk in the field.
Authors: The stage-specific developmental rates, mortalities, and temperature dependencies are taken from the peer-reviewed literature on Aedes aegypti (primarily from controlled laboratory studies and meta-analyses). This is the standard approach when constructing mechanistic models for optimization studies. We will add a dedicated paragraph in the Discussion that (a) states the provenance of every parameter, (b) reports a local sensitivity analysis showing that the optimal intervention windows remain qualitatively stable under ±20 % perturbations of the most influential rates, and (c) explicitly notes the absence of site-specific calibration as a limitation. Because the manuscript’s primary contribution is the adjoint-based scheduling framework and its MPC embedding rather than a fitted predictive model, we do not claim field-validated quantitative forecasts; the reported results illustrate the method under realistic seasonal forcing. revision: partial
Circularity Check
No significant circularity detected
full rationale
The derivation proceeds from an established non-Markovian stage-structured Aedes model (parameters drawn from prior literature), extends it with explicit intervention profiles, derives adjoint equations via standard optimal-control calculus to minimize a time-dependent R0, and evaluates the resulting schedules on Miami temperature forcing. None of the load-bearing steps—model construction, adjoint derivation, or numerical optimization—reduces by definition or self-citation to the target suppression outcomes; the reported suppression is an output of the optimization rather than an input. The framework is self-contained against external benchmarks and contains no self-definitional loops, fitted-input predictions, or uniqueness theorems imported from the authors' prior work.
Axiom & Free-Parameter Ledger
Lean theorems connected to this paper
-
IndisputableMonolith/Foundation/RealityFromDistinction.leanreality_from_one_distinction unclearadjoint-based gradient descent framework to compute the optimal timing of a sequence of interventions by minimizing the time-dependent dengue reproduction number, R0
-
IndisputableMonolith/Cost/FunctionalEquation.leanwashburn_uniqueness_aczel unclearnon-Markovian mechanistic model... auxiliary sub-states... temperature-dependent development rates
Reference graph
Works this paper leans on
-
[1]
The global distribution and burden of dengue,
S. Bhatt, P . W. Gething, O. J. Brady, J. P . Messina, A. W. Farlow, C. L. Moyes, J. M. Drake, J. S. Brownstein, A. G. Hoen, O. Sankohet al., “The global distribution and burden of dengue,”Nature, vol. 496, no. 7446, pp. 504–507, 2013
work page 2013
-
[2]
L. R. Bowman, S. Donegan, and P . J. McCall, “Is dengue vector control deficient in effectiveness or evidence?: Systematic review and meta-analysis,”PLoS neglected tropical diseases, vol. 10, no. 3, p. e0004551, 2016
work page 2016
-
[3]
A critical assessment of vector control for dengue prevention,
N. L. Achee, F. Gould, T. A. Perkins, R. C. Reiner Jr, A. C. Morrison, S. A. Ritchie, D. J. Gubler, R. Teyssou, and T. W. Scott, “A critical assessment of vector control for dengue prevention,”PLoS neglected tropical diseases, vol. 9, no. 5, p. e0003655, 2015
work page 2015
-
[4]
S. Kalimuddin, P . Y. Chia, J. G. Low, and E. E. Ooi, “Dengue and severe dengue,”Clinical microbiology reviews, vol. 38, no. 4, pp. e00 244–24, 2025
work page 2025
-
[5]
V . Alvarado-Castro, S. Paredes-Sol´ıs, E. Nava-Aguilera, A. Morales- P´erez, L. Alarc´on-Morales, N. A. Balderas-Vargas, and N. Ander- sson, “Assessing the effects of interventions for aedes aegypti control: systematic review and meta-analysis of cluster randomised controlled trials,”BMC public health, vol. 17, no. Suppl 1, p. 384, 2017
work page 2017
-
[6]
Community effectiveness of indoor spraying as a dengue vector control method: A systematic review,
M. Samuel, D. Maoz, P . Manrique, T. Ward, S. Runge-Ranzinger, J. Toledo, R. Boyce, and O. Horstick, “Community effectiveness of indoor spraying as a dengue vector control method: A systematic review,”PLoS neglected tropical diseases, vol. 11, no. 8, p. e0005837, 2017
work page 2017
-
[7]
A. B. Wilke, C. Vasquez, A. Carvajal, M. Ramirez, G. Cardenas, W. D. Petrie, and J. C. Beier, “Effectiveness of adulticide and larvicide in controlling high densities of aedes aegypti in urban environments,” PLoS One, vol. 16, no. 1, p. e0246046, 2021
work page 2021
-
[8]
Building the evidence base for dengue vector control: searching for certainty in an uncertain world,
O. Horstick, R. Boyce, and S. Runge-Ranzinger, “Building the evidence base for dengue vector control: searching for certainty in an uncertain world,”Pathogens and Global Health, vol. 112, no. 8, pp. 395–403, 2018
work page 2018
-
[9]
A. Jaffal, J. Fite, T. Baldet, P . Delaunay, F. Jourdain, R. Mora-Castillo, M.-M. Olive, and D. Roiz, “Current evidences of the efficacy of mosquito mass-trapping interventions to reduce aedes aegypti and aedes albopictus populations and aedes-borne virus transmission,” PLoS Neglected Tropical Diseases, vol. 17, no. 3, p. e0011153, 2023
work page 2023
-
[10]
D. A. Focks, D. G. Haile, E. Daniels, and G. A. Mount, “Dynamic life table model for aedes aegypti (diptera: Culicidae): analysis of the literature and model development,”Journal of medical entomology, vol. 30, no. 6, pp. 1003–1017, 1993
work page 1993
-
[11]
Follow up estimation of aedes aegypti entomological parameters and mathematical modellings,
H. M. Yang, M. d. L. da Graca Macoris, K. C. Galvani, and M. T. M. Andrighetti, “Follow up estimation of aedes aegypti entomological parameters and mathematical modellings,”Biosystems, vol. 103, no. 3, pp. 360–371, 2011. 11 (a) (b) Fig. A.2. Panels (a) and (b) show the time-dependent larvicide-induced larval mortality rate associated with ten optimally sc...
work page 2011
-
[12]
Assessing the effects of temperature on dengue transmission,
H. Yang, M. Macoris, K. Galvani, M. Andrighetti, and D. Wanderley, “Assessing the effects of temperature on dengue transmission,” Epidemiology & Infection, vol. 137, no. 8, pp. 1179–1187, 2009
work page 2009
-
[13]
Assessing the effects of temperature on the population of aedes aegypti, the vector of dengue,
——, “Assessing the effects of temperature on the population of aedes aegypti, the vector of dengue,”Epidemiology & Infection, vol. 137, no. 8, pp. 1188–1202, 2009
work page 2009
-
[14]
E. A. Mordecai, J. M. Cohen, M. V . Evans, P . Gudapati, L. R. Johnson, C. A. Lippi, K. Miazgowicz, C. C. Murdock, J. R. Rohr, S. J. Ryan et al., “Detecting the impact of temperature on transmission of zika, dengue, and chikungunya using mechanistic models,”PLoS neglected tropical diseases, vol. 11, no. 4, p. e0005568, 2017
work page 2017
-
[15]
E. P . Pliego, J. Vel´azquez-Castro, and A. F. Collar, “Seasonality on the life cycle of aedes aegypti mosquito and its statistical relation with dengue outbreaks,”Applied Mathematical Modelling, vol. 50, pp. 484–496, 2017
work page 2017
-
[16]
A. Kilicmanet al., “The development of a deterministic dengue epidemic model with the influence of temperature: a case study in malaysia,”Applied Mathematical Modelling, vol. 90, pp. 547–567, 2021
work page 2021
-
[17]
Managing disease outbreaks: The importance of vector mobility and spatially heterogeneous control,
J. Demers, S. Bewick, F. Agusto, K. A. Caillou ¨et, W. F. Fagan, and S. L. Robertson, “Managing disease outbreaks: The importance of vector mobility and spatially heterogeneous control,”PLoS computational biology, vol. 16, no. 8, p. e1008136, 2020
work page 2020
-
[18]
Stochastic modeling, analysis, and simulation of dengue in valle del cauca: A case study,
D. A. Becerra-Becerra, J. Rangel, and V . Arunachalam, “Stochastic modeling, analysis, and simulation of dengue in valle del cauca: A case study,”Mathematical Biosciences and Engineering, vol. 23, no. 2, pp. 266–290, 2026
work page 2026
-
[19]
Bayesian spatial and spatio-temporal approaches to modelling dengue fever: a systematic review,
A. Aswi, S. Cramb, P . Moraga, and K. Mengersen, “Bayesian spatial and spatio-temporal approaches to modelling dengue fever: a systematic review,”Epidemiology & Infection, vol. 147, p. e33, 2019
work page 2019
-
[20]
N. C. Dom, N. A. M. H. Abdullah, R. Dapari, and S. A. Salleh, “Fine-scale predictive modeling of aedes mosquito abundance and dengue risk indicators using machine learning algorithms with microclimatic variables,”Scientific Reports, vol. 15, no. 1, p. 37017, 2025
work page 2025
-
[21]
F. P . Rocha and M. Giesbrecht, “Machine learning algorithms for dengue risk assessment: a case study for s ˜ao lu´ıs do maranh˜ao: Fp rocha, m. giesbrecht,”Computational and Applied Mathematics, vol. 41, no. 8, p. 393, 2022
work page 2022
-
[22]
J. Chen, X. Huo, A. B. Wilke, J. C. Beier, C. Vasquez, W. Petrie, R. S. Cantrell, C. Cosner, and S. Ruan, “Linking mathematical models and trap data to infer the proliferation, abundance, and control of aedes aegypti,”Acta tropica, vol. 239, p. 106837, 2023
work page 2023
-
[23]
Assessing dengue risk globally using non-markovian models,
A. Vajdi, L. W. Cohnstaedt, and C. M. Scoglio, “Assessing dengue risk globally using non-markovian models,”Journal of Theoretical Biology, vol. 591, p. 111865, 2024
work page 2024
-
[24]
Modeling and optimal control applied to a vector borne disease,
H. S. Rodrigues, M. T. T. Monteiro, and D. F. Torres, “Modeling and optimal control applied to a vector borne disease,”arXiv preprint arXiv:1207.1949, 2012
-
[25]
E. Pliego-Pliego, O. Vasilieva, J. Vel´azquez-Castro, and A. F. Collar, “Control strategies for a population dynamics model of aedes aegypti with seasonal variability and their effects on dengue incidence,”Applied Mathematical Modelling, vol. 81, pp. 296–319, 2020
work page 2020
-
[26]
A. S. V . de Vasconcelos, J. S. de Lima, and R. T. N. Cardoso, “Multiobjective optimization to assess dengue control costs using a climate-dependent epidemiological model,”Scientific Reports, vol. 13, no. 1, p. 10271, 2023
work page 2023
-
[27]
A. Abidemi, O. J. Peteret al., “An optimal control model for dengue dynamics with asymptomatic, isolation, and vigilant compartments,”Decision Analytics Journal, vol. 10, p. 100413, 2024
work page 2024
-
[28]
Biological and chemical control of mosquito population by optimal control approach,
J. H. Arias-Castro, H. J. Martinez-Romero, and O. Vasilieva, “Biological and chemical control of mosquito population by optimal control approach,”Games, vol. 11, no. 4, p. 62, 2020
work page 2020
-
[29]
Optimal control strategies for dengue transmission in pakistan,
F. Agusto and M. Khan, “Optimal control strategies for dengue transmission in pakistan,”Mathematical biosciences, vol. 305, pp. 102–121, 2018
work page 2018
-
[30]
Optimal-control techniques for managing dengue outbreaks: An advanced mathematical modeling,
M. S. Miah, M. A. Hye, M. S. Hossain, and M. M. Rahman, “Optimal-control techniques for managing dengue outbreaks: An advanced mathematical modeling,”Natural Sciences, vol. 5, no. 4, p. e70035, 2025
work page 2025
-
[31]
F. F. Herdicho, F. Fatmawati, C. Alfiniyah, M. A. Rois, S. Martini, D. Aldila, and F. Nyabadza, “Optimal control of dengue hem- orrhagic fever model by classifying sex in west java province, indonesia,”Scientific reports, vol. 15, no. 1, p. 17127, 2025
work page 2025
-
[32]
An optimal control model of mosquito reduction management in a dengue endemic region,
K. P . Wijaya, T. G¨otz, and E. Soewono, “An optimal control model of mosquito reduction management in a dengue endemic region,” International Journal of Biomathematics, vol. 7, no. 05, p. 1450056, 2014
work page 2014
-
[33]
A. E. Bryson,Applied optimal control: optimization, estimation and control. Routledge, 2018
work page 2018
-
[34]
L. S. Pontryagin,Mathematical theory of optimal processes. Routledge, 2018
work page 2018
-
[35]
H. Hersbach, B. Bell, P . Berrisford, S. Hirahara, A. Hor ´anyi, J. Mu ˜noz-Sabater, J. Nicolas, C. Peubey, R. Radu, D. Schepers et al., “The era5 global reanalysis,”Quarterly Journal of the Royal Meteorological Society, vol. 146, no. 730, pp. 1999–2049, 2020
work page 1999
-
[36]
Geneva: World Health Organization, 2012
World Health Organization,Handbook for Integrated Vector Manage- ment. Geneva: World Health Organization, 2012
work page 2012
-
[37]
L. C. Farnesi, A. J. Martins, D. Valle, and G. L. Rezende, “Embryonic development of aedes aegypti (diptera: Culicidae): influence of different constant temperatures,”Mem´ orias do Instituto Oswaldo Cruz, vol. 104, pp. 124–126, 2009
work page 2009
-
[38]
R. Barrera, “Competition and population regulation inAedes aegypti (Diptera: Culicidae): the role of density-dependent intraspecific larval competition,”Journal of Medical Entomology, vol. 33, no. 5, pp. 820–826, 1996
work page 1996
-
[39]
Surveillance and control ofAedes aegyptiandAedes albopictusin the United States,
Centers for Disease Control and Prevention, “Surveillance and control ofAedes aegyptiandAedes albopictusin the United States,” https://www.cdc.gov/mosquitoes/pdfs/ mosquito-control-508.pdf, 2017, accessed June 2025
work page 2017
-
[40]
Robust and optimal predictive control of the COVID- 19 outbreak,
J. K ¨ohler, L. Schwenkel, A. Koch, J. Berberich, P . Pauli, and F. Allg¨ower, “Robust and optimal predictive control of the COVID- 19 outbreak,”Annual Reviews in Control, vol. 51, pp. 525–539, 2021
work page 2021
-
[41]
Nonlinear model predictive control with logic constraints for COVID-19 management,
T. P ´eni, B. Csutak, G. Szederk ´enyi, and G. R ¨ost, “Nonlinear model predictive control with logic constraints for COVID-19 management,”Nonlinear Dynamics, vol. 102, no. 4, pp. 1965–1986, 2020. 12
work page 1965
-
[42]
A Bayesian hierarchical model for estimation of abundance and spatial density ofAedes aegypti,
D. A. M. Villela, C. T. Codec ¸o, F. Figueiredo, G. A. Garcia, R. Maciel- de Freitas, and C. J. Struchiner, “A Bayesian hierarchical model for estimation of abundance and spatial density ofAedes aegypti,”PLoS ONE, vol. 10, no. 4, p. e0123794, 2015
work page 2015
-
[43]
L. Eisen, A. J. Monaghan, S. Lozano-Fuentes, D. F. Steinhoff, M. H. Hayden, and P . E. Bieringer, “The impact of temperature on the bionomics of aedes (stegomyia) aegypti, with special reference to the cool geographic range margins,”Journal of medical entomology, vol. 51, no. 3, pp. 496–516, 2014
work page 2014
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.