Recognition: unknown
Astrocytic resource diffusion stabilizes persistent activity in neural fields
Pith reviewed 2026-05-10 16:23 UTC · model grok-4.3
The pith
Astrocytic resource diffusion stabilizes persistent activity bumps by smoothing asymmetries and suppressing drift.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
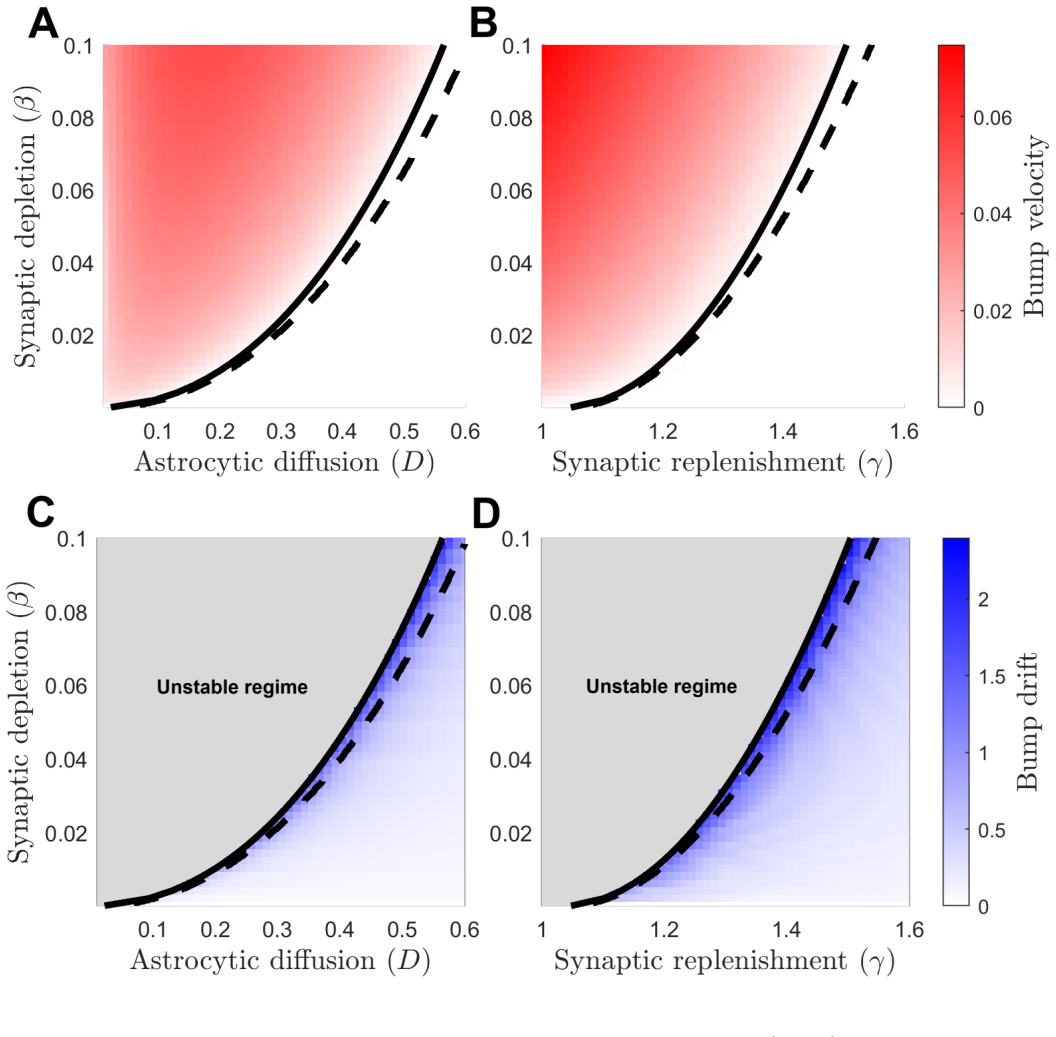
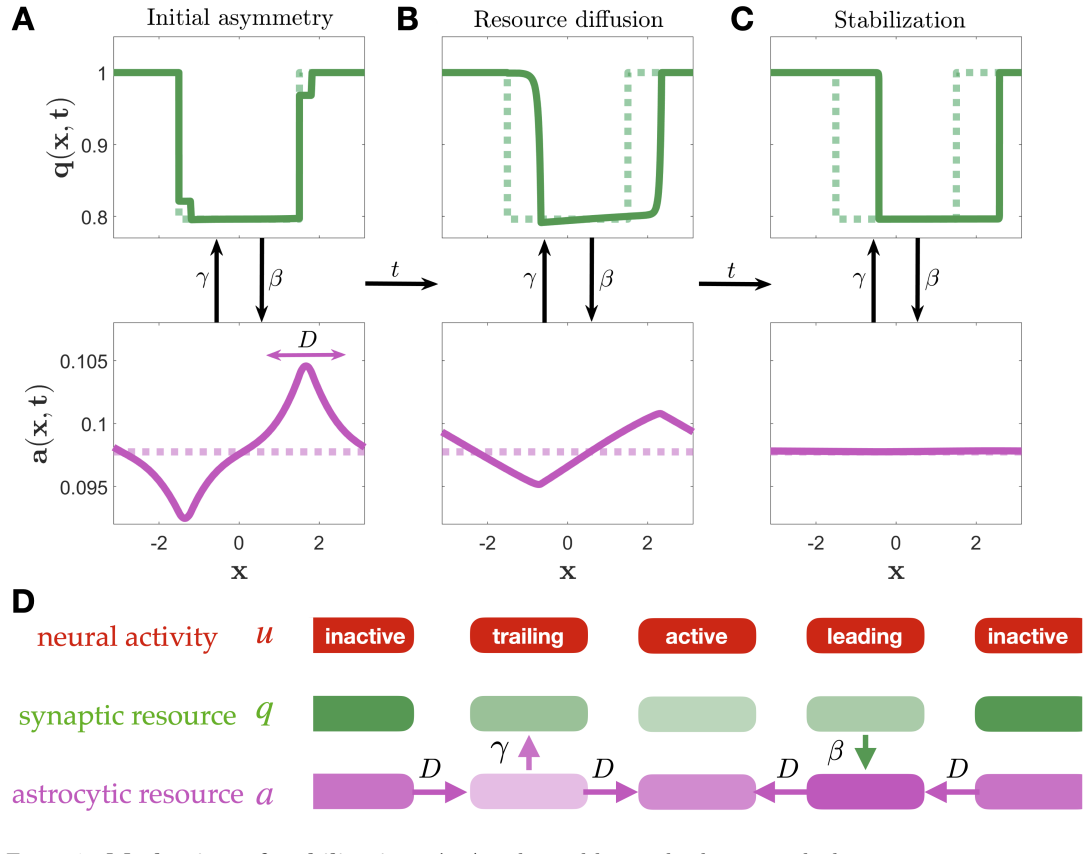
In the coupled astrocyte-neural field system, astrocytic diffusion smooths resource asymmetries created by small bump displacements while synaptic replenishment transfers that smoothing back into the synaptic pool; together, sufficiently strong diffusion and replenishment suppress drift instabilities and enlarge the parameter regime in which stationary bumps persist.
What carries the argument
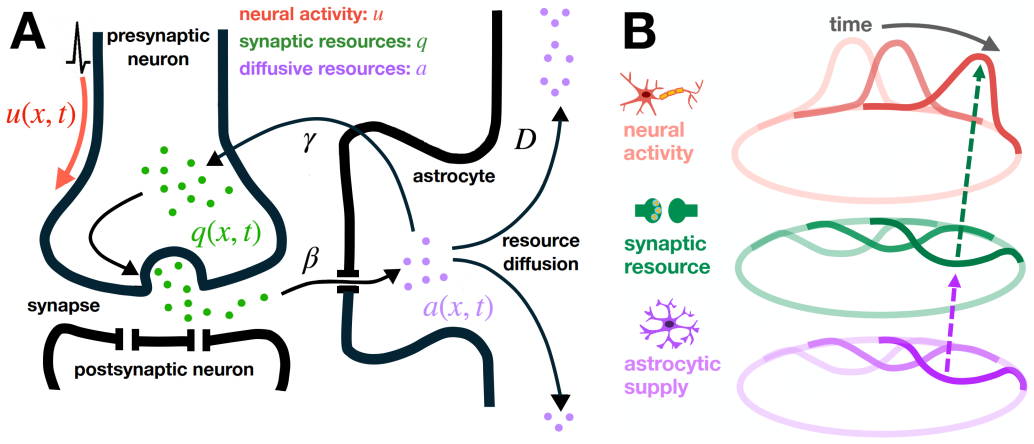
The two-stage stabilization mechanism: astrocytic diffusion smooths resource asymmetries from bump displacements, and synaptic replenishment feeds the result back to the synaptic efficacy pool.
If this is right
- Stationary activity bumps remain fixed over a wider range of synaptic and resource parameters once astrocytic diffusion and replenishment exceed threshold values.
- Drift instabilities that otherwise cause bumps to translate are eliminated by the resource-redistribution loop.
- The conserved resource pool must be both depleted locally and redistributed globally for the stabilization to occur.
- Low-dimensional Fourier truncations of the system reproduce the same stability thresholds found by the full spectral analysis.
Where Pith is reading between the lines
- Disrupting astrocyte gap junctions or resource recycling could cause memory bumps to drift or collapse even when neural connectivity remains intact.
- The ring geometry result suggests that similar stabilization may operate in cortical sheets if local diffusion lengths are comparable to bump widths.
- Manipulating astrocyte calcium waves or metabolic coupling in slice experiments could test whether resource diffusion directly controls bump lifetime.
- The model implies that working-memory deficits linked to glial pathology may arise from loss of this spatial smoothing rather than from purely neuronal changes.
Load-bearing premise
Linearization about the stationary bump solutions, with perturbations at the bump edges treated carefully, accurately determines the stability of the full nonlinear system on the ring.
What would settle it
A direct numerical simulation or experiment in which raising the astrocytic diffusion coefficient fails to reduce bump drift speed or restore stability would falsify the proposed mechanism.
Figures




read the original abstract
Persistent neural activity underlying working memory requires sustained synaptic transmission, yet the metabolic and neurotransmitter support provided by astrocyte networks is largely absent from spatially extended neural circuit models. We introduce a coupled astrocyte-neural field model in which synaptic efficacy is regulated by depletion and recovery of a conserved resource pool recycled and spatially redistributed through diffusively coupled astrocytes. We obtain explicit stationary bump profiles and self-consistency conditions for bump width and amplitude on a canonical ring architecture. Linearizing about these solutions while carefully accounting for perturbations at bump boundaries, we analyze the resulting spectral problem governing stability. Our analysis, supported by numerical simulations and low-dimensional Fourier truncations, reveals a two-stage stabilization mechanism: astrocytic diffusion smooths resource asymmetries created by small bump displacements, and synaptic replenishment transfers this smoothing back to the synaptic pool. Together, sufficiently strong diffusion and replenishment suppress drift instabilities and enlarge the parameter regime in which stationary bumps persist.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript introduces a coupled neural field and astrocyte model incorporating a conserved resource pool that is depleted by synaptic activity and redistributed via astrocytic diffusion. Explicit stationary bump profiles are obtained on a ring architecture, along with self-consistency conditions for their width and amplitude. The system is linearized about these profiles, carefully accounting for perturbations at the bump boundaries, and the resulting spectral problem is analyzed. The analysis, supported by numerical simulations and low-dimensional Fourier truncations, identifies a two-stage stabilization mechanism whereby astrocytic diffusion smooths resource asymmetries induced by small bump displacements, and synaptic replenishment feeds this smoothing back to the synaptic pool, thereby suppressing drift instabilities and expanding the parameter range for persistent stationary bumps.
Significance. This result, if the linear stability analysis holds under nonlinear perturbations, would be significant for models of working memory by providing a biologically plausible way for astrocyte-mediated resource diffusion to stabilize bump attractors against drift. The derivation of explicit stationary profiles and the identification of the two-stage mechanism are notable strengths, as is the use of simulations to corroborate the spectral findings. The work bridges neural field theory with glial biology in a mathematically tractable manner.
major comments (2)
- [Stability analysis (linearization about stationary bumps)] The central stability claim depends on the linearized spectral problem obtained by accounting for perturbations at bump boundaries. However, the explicit form of the linearized operator, the boundary matching conditions, and the resulting eigenvalue spectrum as functions of the astrocytic diffusion coefficient and synaptic replenishment rate are not provided in sufficient detail to allow verification that all relevant eigenvalues (particularly the translational mode) lie in the left half-plane for the claimed parameter regimes.
- [Numerical simulations and Fourier truncations] The manuscript states that the stabilization is confirmed by numerical simulations and low-dimensional Fourier truncations, but lacks details on the integration scheme, spatial discretization, time horizons, or quantitative measures of drift suppression (e.g., bump position variance over long times). This is load-bearing because the linear analysis alone may not preclude nonlinear re-emergence of instabilities.
minor comments (2)
- [Abstract] The abstract could more explicitly state the model equations or key parameters to improve accessibility.
- [Notation] Ensure consistent use of symbols for the resource pool, diffusion coefficients, and replenishment rates throughout the text and figures.
Simulated Author's Rebuttal
We thank the referee for their detailed and constructive review of our manuscript. Their comments highlight important aspects of the stability analysis and numerical validation that merit clarification. We address each major comment point by point below and will revise the manuscript accordingly to improve verifiability and reproducibility.
read point-by-point responses
-
Referee: The central stability claim depends on the linearized spectral problem obtained by accounting for perturbations at bump boundaries. However, the explicit form of the linearized operator, the boundary matching conditions, and the resulting eigenvalue spectrum as functions of the astrocytic diffusion coefficient and synaptic replenishment rate are not provided in sufficient detail to allow verification that all relevant eigenvalues (particularly the translational mode) lie in the left half-plane for the claimed parameter regimes.
Authors: We acknowledge that while the manuscript outlines the linearization procedure around the stationary bump profiles (including boundary perturbations via a moving-frame approach), the explicit operator expressions, matching conditions, and parameter-dependent eigenvalue spectra are presented at a summary level rather than in full expanded form. To address this, we will add a new appendix in the revised manuscript that derives the linearized operator in detail, specifies the boundary matching conditions, and includes explicit eigenvalue calculations (or numerical spectra) for representative values of the astrocytic diffusion coefficient and synaptic replenishment rate. This will allow direct verification that the translational mode is stabilized in the claimed regimes. revision: yes
-
Referee: The manuscript states that the stabilization is confirmed by numerical simulations and low-dimensional Fourier truncations, but lacks details on the integration scheme, spatial discretization, time horizons, or quantitative measures of drift suppression (e.g., bump position variance over long times). This is load-bearing because the linear analysis alone may not preclude nonlinear re-emergence of instabilities.
Authors: We agree that the current description of the numerical methods is insufficient for full reproducibility and for rigorously supporting the claim that linear stability extends to nonlinear regimes. In the revised manuscript, we will include a dedicated methods subsection (or appendix) specifying the integration scheme (e.g., explicit Euler or Runge-Kutta with spectral or finite-difference spatial discretization), grid resolution, time-step size, total simulation horizons, and quantitative diagnostics such as bump centroid variance and drift velocity over long times. These additions will demonstrate drift suppression and help rule out nonlinear instability re-emergence. revision: yes
Circularity Check
No circularity: derivation chain is self-contained
full rationale
The paper derives explicit stationary bump profiles and self-consistency conditions directly from the coupled neural-astrocyte PDE system on the ring, then linearizes about those profiles (accounting for boundary perturbations) to obtain the spectral problem whose eigenvalues determine stability. The two-stage mechanism (astrocytic diffusion smoothing resource asymmetries, followed by synaptic replenishment) is extracted from the structure of this linear operator and confirmed via Fourier truncations and simulations. No step reduces a claimed prediction or stability result to a fitted parameter, self-definition, or self-citation chain; the stationary solutions and eigenvalue conditions are obtained from the model equations without presupposing the final stability conclusion. The analysis therefore remains independent of its inputs.
Axiom & Free-Parameter Ledger
free parameters (2)
- astrocytic diffusion coefficient
- synaptic replenishment rate
axioms (2)
- domain assumption The resource pool is conserved and recycled through diffusively coupled astrocytes.
- domain assumption Stationary bump solutions exist on a canonical ring architecture.
invented entities (1)
-
conserved resource pool
no independent evidence
Reference graph
Works this paper leans on
-
[1]
L. F. Abbott, Juan A. Varela, Kamal Sen, and Sacha B. Nelson. Synaptic depression and cortical gain control.Science, 275(5297):221–224, 1997
1997
-
[2]
S. Amari. Dynamics of pattern formation in lateral-inhibition type neural fields.Biological Cybernetics, 27:77–87, 1977
1977
-
[3]
Interplay between persistent 20 activity and activity-silent dynamics in the prefrontal cortex underlies serial biases in working memory
JoaoBarbosa, HeikeStein, RebeccaLMartinez, AdriàGalan-Gadea, SihaiLi, JosepDalmau, KirstenCS Adam, Josep Valls-Solé, Christos Constantinidis, and Albert Compte. Interplay between persistent 20 activity and activity-silent dynamics in the prefrontal cortex underlies serial biases in working memory. Nature neuroscience, 23(8):1016–1024, 2020
2020
-
[4]
Benna and Stefano Fusi
Marcus K. Benna and Stefano Fusi. Computational principles of synaptic memory consolidation. Nature Neuroscience, 19(12):1697–1706, 2016
2016
-
[5]
Springer Science & Business Media, 2008
Mario Bernardo, Chris Budd, Alan Richard Champneys, and Piotr Kowalczyk.Piecewise-smooth dynamical systems: theory and applications, volume 163. Springer Science & Business Media, 2008
2008
-
[6]
P. C. Bressloff and Z. P. Kilpatrick. Two-dimensional bumps in piecewise smooth neral fields with synaptic depression.SIAM Journal of Applied Math, 71(2):379–408, 2011
2011
-
[7]
Spatiotemporal dynamics of continuum neural fields.Journal of Physics A: Mathematical and Theoretical, 45(3):033001, 2012
Paul C Bressloff. Spatiotemporal dynamics of continuum neural fields.Journal of Physics A: Mathematical and Theoretical, 45(3):033001, 2012
2012
-
[8]
Two-dimensional bumps in piecewise smooth neural fields with synaptic depression.SIAM Journal on Applied Mathematics, 71(2):379–408, 2011
Paul C Bressloff and Zachary P Kilpatrick. Two-dimensional bumps in piecewise smooth neural fields with synaptic depression.SIAM Journal on Applied Mathematics, 71(2):379–408, 2011
2011
-
[9]
N. F. Britton. Spatial structures and periodic travelling waves in an integro-differential reaction-diffusion population model.SIAM Journal on Applied Mathematics, 50(6):1663–1688, 1990
1990
-
[10]
Multiscale motion and deformation of bumps in stochastic neural fields with dynamic connectivity.Multiscale Modeling & Simulation, 22(1):178–203, 2024
Heather L Cihak and Zachary P Kilpatrick. Multiscale motion and deformation of bumps in stochastic neural fields with dynamic connectivity.Multiscale Modeling & Simulation, 22(1):178–203, 2024
2024
-
[11]
Compte, N
A. Compte, N. Brunel, P.S. Goldman-Rakic, and X.J. Wang. Synaptic mechanisms and network dynamics underlying spatial working memory in a cortical network model.Cerebral Cortex, 10(9), 2000
2000
-
[12]
Constantinidis and T
C. Constantinidis and T. Klingberg. The neuroscience of working memory capacity and training. Nature Reviews Neuroscience, 17:438–449, 2016
2016
-
[13]
Springer, 2014
Stephen Coombes, Peter beim Graben, Roland Potthast, and James Wright.Neural fields: theory and applications. Springer, 2014
2014
-
[14]
Waves and bumps in neuronal networks with axo-dendritic synaptic interactions.Physica D: Nonlinear Phenomena, 178(3-4):219–241, 2003
Stephen Coombes, Gabriel J Lord, and Markus R Owen. Waves and bumps in neuronal networks with axo-dendritic synaptic interactions.Physica D: Nonlinear Phenomena, 178(3-4):219–241, 2003
2003
-
[15]
Evans functions for integral neural field equations with heaviside firing rate function.SIAM Journal on Applied Dynamical Systems, 3(4):574–600, 2004
Stephen Coombes and Markus R Owen. Evans functions for integral neural field equations with heaviside firing rate function.SIAM Journal on Applied Dynamical Systems, 3(4):574–600, 2004
2004
-
[16]
Bumps, breathers, and waves in a neural network with spike frequency adaptation.Physical Review Letters, 94(14):148102, 2005
Stephen Coombes and Markus R Owen. Bumps, breathers, and waves in a neural network with spike frequency adaptation.Physical Review Letters, 94(14):148102, 2005
2005
-
[17]
Astrocytes: orchestrating synaptic plasticity? Neuroscience, 323:43–61, 2016
Maurizio De Pittà, Nicolas Brunel, and Andrea Volterra. Astrocytes: orchestrating synaptic plasticity? Neuroscience, 323:43–61, 2016
2016
-
[18]
De Pitttá and N
M. De Pitttá and N. Brunel. Multiple forms of working memory emerge from synapse–astrocyte interactions in a neuron–glia network model.Neuroscience, 119(43), 2022
2022
-
[19]
Folias and P.C
S.E. Folias and P.C. Bressloff. Breathing pulses in an excitatory neural network.SIAM Journal on Applied Dynamical Systems, 3(3):478–407, 2004
2004
-
[20]
Funahashi, C.J
S. Funahashi, C.J. Bruce, and P.S. Goldman-Rakic. Mnemonic coding of visual space in the monkey’s dorsolateral prefrontal cortex.Sci Rep, 61:331–349, 1989. 21
1989
-
[21]
Astroglial networks: a step further in neuroglial and gliovascular interactions.Nature Reviews Neuroscience, 11(2):87–99, 2010
Christian Giaume, Annette Koulakoff, Lisa Roux, David Holcman, and Nathalie Rouach. Astroglial networks: a step further in neuroglial and gliovascular interactions.Nature Reviews Neuroscience, 11(2):87–99, 2010
2010
-
[22]
Goldman-Rakic
P.S. Goldman-Rakic. Cellular basis of working memory.Neuron, 14:477–485, 1995
1995
-
[23]
Existence and stability of standing pulses in neural networks: Ii
Yixin Guo and Carson C Chow. Existence and stability of standing pulses in neural networks: Ii. stability.SIAM Journal on Applied Dynamical Systems, 4(2):249–281, 2005
2005
-
[24]
Gregory Handy and Sean D. Lawley. Revising Berg–Purcell for finite receptor kinetics.Biophysical Journal, 120(11):2237–2248, 2021
2021
-
[25]
Z. P. Kilpatrick and P. C. Bressloff. Effects of synaptic depression and adaptation on spatiotemporal dynamics of an excitatory neuronal network.Physica D, 239:547–560, 2010
2010
-
[26]
Z. P. Kilpatrick and P. C. Bressloff. Stability of bumps in piecewise smooth neural fields with nonlinear adaptation.Physica D, 239(12):1048–1060, 2010
2010
-
[27]
Ring attractor dynamics in the drosophila central brain.Science, 356(6340):849–853, 2017
Sung Soo Kim, Hervé Rouault, Shaul Druckmann, and Vivek Jayaraman. Ring attractor dynamics in the drosophila central brain.Science, 356(6340):849–853, 2017
2017
-
[28]
Neuron–astrocyte associative memory
Leo Kozachkov, Jean-Jacques Slotine, and Dmitry Krotov. Neuron–astrocyte associative memory. Proceedings of the National Academy of Sciences, 122(21):e2417788122, 2025
2025
-
[29]
Lawley, Janet Best, and Michael C
Sean D. Lawley, Janet Best, and Michael C. Reed. Neurotransmitter concentrations in the presence of neural switching in one dimension.Discrete and Continuous Dynamical Systems – Series B, 21(7):2255–2273, 2016
2016
-
[30]
Changing concepts of working memory.Nature neuroscience, 17(3):347–356, 2014
Wei Ji Ma, Masud Husain, and Paul M Bays. Changing concepts of working memory.Nature neuroscience, 17(3):347–356, 2014
2014
-
[31]
Cholin- ergic neuromodulation of prefrontal attractor dynamics controls performance in spatial working memory
Alexandre Mahrach, David Bestue, Xue-Lian Qi, Christos Constantinidis, and Albert Compte. Cholin- ergic neuromodulation of prefrontal attractor dynamics controls performance in spatial working memory. Journal of Neuroscience, 44(23), 2024
2024
-
[32]
Spreading disease: integro-differential equations old and new.Mathematical Biosciences, 184(2):201–222, 2003
Jan Medlock and Mark Kot. Spreading disease: integro-differential equations old and new.Mathematical Biosciences, 184(2):201–222, 2003
2003
-
[33]
Stable population coding for working memory coexists with heterogeneous neural dynamics in prefrontal cortex.Proceedings of the National Academy of Sciences, 114(2):394–399, 2017
John D Murray, Alberto Bernacchia, Nicholas A Roy, Christos Constantinidis, Ranulfo Romo, and Xiao-Jing Wang. Stable population coding for working memory coexists with heterogeneous neural dynamics in prefrontal cortex.Proceedings of the National Academy of Sciences, 114(2):394–399, 2017
2017
-
[34]
The role of astrocytes in the regulation of synaptic plasticity and memory formation.Neural plasticity, 2013(1):185463, 2013
Yusuke Ota, Alexander T Zanetti, and Robert M Hallock. The role of astrocytes in the regulation of synaptic plasticity and memory formation.Neural plasticity, 2013(1):185463, 2013
2013
-
[35]
Emerging role for astroglial networks in information processing: from synapse to behavior.Trends in neurosciences, 36(7):405–417, 2013
Ulrike Pannasch and Nathalie Rouach. Emerging role for astroglial networks in information processing: from synapse to behavior.Trends in neurosciences, 36(7):405–417, 2013
2013
-
[36]
Pinto and G.B
D.J. Pinto and G.B. Ermentrout. Spatially structured activity in synaptically coupled neuronal networks: I. traveling fronts and pulses.SIAM Journal on Applied Mathematics, 62(1):206–225, 2001
2001
-
[37]
Spatial-temporal dynamics in nonlocal epidemiological models
Shigui Ruan. Spatial-temporal dynamics in nonlocal epidemiological models. In Yasuhiro Takeuchi, Yoh Iwasa, and Kazunori Sato, editors,Mathematics for Life Science and Medicine, pages 97–122. Springer, Berlin, 2007. 22
2007
-
[38]
Schouseboe, L.K
A. Schouseboe, L.K. Bak, and H.S. Waagepetersen. Astrocytic control of biosynthesis and turnover of the neurotransmitters glutamate and gaba.Frontiers in Endocrinology, 4, 2013
2013
-
[39]
VladimirShustermanandWilliamCTroy. Frombaselinetoepileptiformactivity: apathtosynchronized rhythmicity in large-scale neural networks.Physical Review E—Statistical, Nonlinear, and Soft Matter Physics, 77(6):061911, 2008
2008
-
[40]
Stable and dynamic coding for working memory in primate prefrontal cortex.Journal of neuroscience, 37(27):6503–6516, 2017
Eelke Spaak, Kei Watanabe, Shintaro Funahashi, and Mark G Stokes. Stable and dynamic coding for working memory in primate prefrontal cortex.Journal of neuroscience, 37(27):6503–6516, 2017
2017
-
[41]
Decoding working memory information from neurons with and without persistent activity in the primate prefrontal cortex.Journal of Neurophysiology, 130(6):1392–1402, 2023
Lilianna Thrower, Wenhao Dang, Rye G Jaffe, Jasmine D Sun, and Christos Constantinidis. Decoding working memory information from neurons with and without persistent activity in the primate prefrontal cortex.Journal of Neurophysiology, 130(6):1392–1402, 2023
2023
-
[42]
Neural networks with dynamic synapses.Neural computation, 10(4):821–835, 1998
Misha Tsodyks, Klaus Pawelzik, and Henry Markram. Neural networks with dynamic synapses.Neural computation, 10(4):821–835, 1998
1998
-
[43]
Tsodyks and Henry Markram
Misha V. Tsodyks and Henry Markram. The neural code between neocortical pyramidal neurons depends on neurotransmitter release probability.Proceedings of the National Academy of Sciences, 94(2):719–723, 1997
1997
-
[44]
X.J. Wang. Synaptic reverberation underlying mnemonic persistent activity.Trends in Neurosciences, 24:455–463, 2001
2001
-
[45]
The astrocyte: powerhouse and recycling center.Cold Spring Harbor perspectives in biology, 7(12):a020396, 2015
Bruno Weber and L Felipe Barros. The astrocyte: powerhouse and recycling center.Cold Spring Harbor perspectives in biology, 7(12):a020396, 2015
2015
-
[46]
Learning-associated astrocyte ensembles regulate memory recall.Nature, 637(8045):478–486, 2025
Michael R Williamson, Wookbong Kwon, Junsung Woo, Yeunjung Ko, Ehson Maleki, Kwanha Yu, Sanjana Murali, Debosmita Sardar, and Benjamin Deneen. Learning-associated astrocyte ensembles regulate memory recall.Nature, 637(8045):478–486, 2025
2025
-
[47]
Wimmer, D.Q
K. Wimmer, D.Q. Nykamp, C. Constantinidis, and A. Compte. Bump attractor dynamics in prefrontal cortex explains behavioral precision in spatial working memory.Nature Neuroscience, 17:431–439, 2014. 23
2014
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.