Recognition: unknown
Role of chloride concentration in modulating seizure transitions in excitatory and inhibitory networks
Pith reviewed 2026-05-10 07:59 UTC · model grok-4.3
The pith
A fraction of inhibitory synaptic conductance acts as a control parameter organizing seizure dynamics into pre-ictal, ictal-tonic, and ictal-clonic stages.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
The fraction of inhibitory synaptic conductance contributing to channel-mediated influx acts as a control parameter that organizes seizure dynamics into distinct stages—pre-ictal, ictal-tonic, and ictal-clonic—distinguished by characteristic amplitude and frequency signatures. Decreasing this fraction shortens ictal activity and suppresses seizure initiation, whereas high fractions promote the emergence of ictal-tonic and ictal-clonic stages and spiral-wave dynamics, rendering seizure dynamics largely insensitive to inhibition. At intermediate values, seizures bypass the ictal-tonic stage and emerge directly as the ictal-clonic stage. Joint variation with synaptic strengths shows that recur
What carries the argument
The fraction of inhibitory synaptic conductance contributing to channel-mediated chloride influx, which sets intracellular chloride levels and thereby tunes the excitation-inhibition balance that drives stage transitions.
If this is right
- Decreasing the influx fraction shortens ictal activity and suppresses seizure initiation.
- High influx fractions promote ictal-tonic and ictal-clonic stages plus spiral-wave dynamics and render seizures insensitive to inhibition.
- Intermediate influx fractions cause seizures to skip the ictal-tonic stage and emerge directly as ictal-clonic.
- Recurrent excitation expands the tonic-clonic regime while recurrent inhibition prolongs pre-ictal states and suppresses ictal-clonic activity.
Where Pith is reading between the lines
- If the influx fraction truly governs stage order, then selective blockade of chloride-permeable channels could be tested as a way to truncate seizures at the pre-ictal phase.
- The model's prediction of inhibition insensitivity at high fractions offers a possible explanation for why some seizures persist despite enhanced GABA signaling.
- Joint manipulation of the fraction and synaptic weights suggests experiments that vary both transporter activity and recurrent connectivity to map the boundaries between seizure regimes.
Load-bearing premise
The simulated network dynamics produced by chloride influx and extrusion map directly onto the amplitude, frequency, and sequence of biological seizure stages.
What would settle it
Simultaneous recordings of intracellular chloride levels and local field potentials during induced seizures in brain slices, testing whether the measured influx fraction predicts the same stage sequence and waveform signatures as in the model.
Figures














read the original abstract
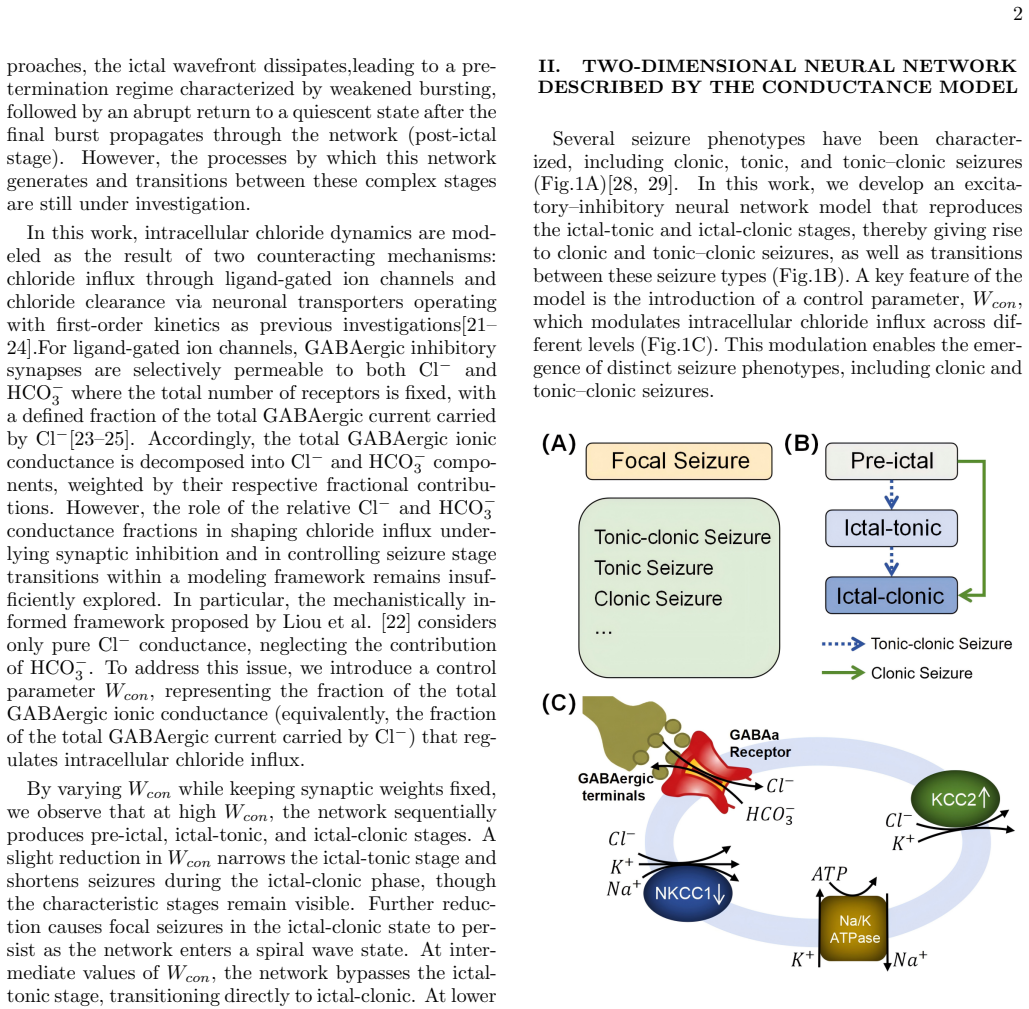
Experimental evidence indicates that intracellular chloride concentration regulates the excitation and inhibition (EI) balance, yet the mechanisms by which activity-dependent chloride dynamics drive seizure evolution and stage transitions remain unclear. We present a conductance-based neuronal network in which EI balance emerges from chloride homeostasis via channel-mediated influx and transporter-mediated extrusion. We show that the fraction of inhibitory synaptic conductance contributing to channel-mediated influx acts as a control parameter that organizes seizure dynamics into distinct stages,pre-ictal, ictal-tonic, and ictal-clonic,distinguished by characteristic amplitude and frequency signatures. Decreasing this fraction shortens ictal activity and suppresses seizure initiation, whereas high fraction promotes the emergence of ictal-tonic and ictal-clonic stages and spiral-wave dynamics, rendering seizure dynamics largely insensitive to inhibition. At intermediate values, seizures bypass the ictal-tonic stage and emerge directly as the icta,clonic stage. Moreover, joint variation of fractions with synaptic strengths reveals that recurrent excitation expands the tonic-clonic seizure, while recurrent inhibition prolongs pre-ictal states and suppresses ictal-clonic activity.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript presents a conductance-based excitatory-inhibitory neuronal network model in which intracellular chloride concentration is dynamically regulated by channel-mediated influx and transporter-mediated extrusion. The central claim is that the fraction of inhibitory synaptic conductance contributing to Cl- influx functions as a control parameter that organizes seizure-like dynamics into three distinct stages—pre-ictal, ictal-tonic, and ictal-clonic—each characterized by specific amplitude and frequency signatures. Decreasing this fraction shortens ictal episodes and suppresses initiation, higher values promote tonic-clonic transitions and spiral-wave activity, and intermediate values cause direct emergence of the clonic stage; joint variation with recurrent synaptic strengths further modulates stage durations and transitions.
Significance. If the simulated stage organization and parameter dependence prove robust and quantitatively consistent with experimental recordings, the work would offer a mechanistic account of how activity-dependent chloride shifts can drive seizure evolution beyond static EI-balance models. The explicit incorporation of chloride homeostasis is a conceptual strength, and the systematic exploration of the influx fraction together with synaptic weights provides a clear, falsifiable parameter map. However, the absence of direct, quantitative matching to in-vivo or in-vitro EEG signatures and the lack of reported sensitivity or validation checks limit the immediate biological interpretability.
major comments (2)
- [Abstract] Abstract: the claim that the fraction 'organizes seizure dynamics into distinct stages... distinguished by characteristic amplitude and frequency signatures' is load-bearing, yet no quantitative criteria (spectral peaks, amplitude thresholds, duration rules, or statistical tests) are supplied for stage assignment. Without these definitions it is impossible to determine whether the three-stage structure emerges from the chloride dynamics or is imposed by the chosen parameter ranges and post-hoc labeling.
- [Abstract] Abstract and model description: the fraction of inhibitory conductance driving channel-mediated Cl- influx is introduced as the organizing control parameter, but the manuscript supplies neither the explicit equations governing chloride influx/extrusion nor any sensitivity analysis showing that the stage sequence persists when other parameters (e.g., reversal potentials, transporter rates) are varied within physiological bounds. This leaves open the possibility that the reported organization is an artifact of the specific implementation rather than a general consequence of chloride homeostasis.
minor comments (2)
- [Abstract] Abstract contains typographical errors: missing space after the comma in 'stages,pre-ictal' and the malformed 'icta,clonic' (should read 'ictal-clonic').
- [Abstract] The abstract reports simulation outcomes but references neither figures, tables, nor any quantitative metrics (e.g., mean stage durations, dominant frequencies, or error bars), making it difficult for readers to assess the strength of the reported effects.
Simulated Author's Rebuttal
We thank the referee for the constructive comments on our manuscript. We address each major comment below and describe the revisions that will be made to strengthen the presentation of the results.
read point-by-point responses
-
Referee: [Abstract] Abstract: the claim that the fraction 'organizes seizure dynamics into distinct stages... distinguished by characteristic amplitude and frequency signatures' is load-bearing, yet no quantitative criteria (spectral peaks, amplitude thresholds, duration rules, or statistical tests) are supplied for stage assignment. Without these definitions it is impossible to determine whether the three-stage structure emerges from the chloride dynamics or is imposed by the chosen parameter ranges and post-hoc labeling.
Authors: We agree that explicit quantitative criteria are needed to make the stage classification reproducible and to demonstrate that the organization arises from the underlying chloride dynamics. In the revised manuscript we will add precise definitions in the Results and Methods sections: pre-ictal episodes are identified by low-amplitude, low-frequency (<4 Hz) activity lasting at least 5 s; tonic stages by sustained high-amplitude (>2× baseline) oscillations in the 8–20 Hz band; and clonic stages by progressively decreasing amplitude with dominant frequency shifting to 3–8 Hz. These criteria will be applied uniformly across all simulations, and we will include a supplementary analysis showing that the same stage sequence is recovered when the criteria are varied within reasonable bounds, confirming that the structure is not an artifact of post-hoc labeling. revision: yes
-
Referee: [Abstract] Abstract and model description: the fraction of inhibitory conductance driving channel-mediated Cl- influx is introduced as the organizing control parameter, but the manuscript supplies neither the explicit equations governing chloride influx/extrusion nor any sensitivity analysis showing that the stage sequence persists when other parameters (e.g., reversal potentials, transporter rates) are varied within physiological bounds. This leaves open the possibility that the reported organization is an artifact of the specific implementation rather than a general consequence of chloride homeostasis.
Authors: The governing equations for intracellular chloride (including the term for channel-mediated influx proportional to the inhibitory conductance fraction and the KCC2-mediated extrusion term) are stated in the Methods section. To address the request for robustness, we will add a dedicated sensitivity analysis (new figure and text) in which we systematically vary the chloride reversal potential (±10 mV around the nominal value) and the transporter rate constant (±30 % of the baseline value) while keeping the influx fraction fixed. The results show that the three-stage organization and its dependence on the influx fraction remain intact across the tested physiological range, indicating that the reported control-parameter behavior is a general consequence of the chloride-homeostasis mechanism rather than an artifact of the specific parameter choice. revision: yes
Circularity Check
No significant circularity; parameter sweep yields observed regimes
full rationale
The paper constructs a conductance-based EI network with explicit chloride influx/extrusion equations. It then treats the fraction of inhibitory conductance driving channel-mediated Cl- influx as an explicit, independently varied control parameter and reports the resulting dynamical regimes (pre-ictal, ictal-tonic, ictal-clonic) distinguished by amplitude/frequency features. These regimes are not defined in terms of the fraction itself; they are simulation outcomes. No equations reduce to tautology, no fitted quantity is relabeled as a prediction, and no load-bearing self-citation chain is invoked. The derivation chain from model equations to stage organization is therefore independent and self-contained.
Axiom & Free-Parameter Ledger
free parameters (1)
- fraction of inhibitory synaptic conductance contributing to channel-mediated influx
axioms (1)
- domain assumption EI balance emerges from chloride homeostasis via channel-mediated influx and transporter-mediated extrusion
Reference graph
Works this paper leans on
-
[1]
On the nature of seizure dynamics.Brain, 137(8):2210–2230, 2014
Viktor K Jirsa, William C Stacey, Pascale P Quilichini, Anton I Ivanov, and Christophe Bernard. On the nature of seizure dynamics.Brain, 137(8):2210–2230, 2014
2014
-
[2]
Noise ampli- fication precedes extreme epileptic events on human eeg
Oleg E Karpov, Vadim V Grubov, Vladimir A Maksi- menko, Nikita Utaschev, Viachaslav E Semerikov, De- nis A Andrikov, and Alexander E Hramov. Noise ampli- fication precedes extreme epileptic events on human eeg. Physical Review E, 103(2):022310, 2021
2021
-
[3]
The secondarily general- ized tonic-clonic seizure: a videotape analysis.Neurology, 44(8):1403–1403, 1994
WH Theodore, RJ Porter, P Albert, K Kelley, E Brom- field, O Devinsky, and S Sato. The secondarily general- ized tonic-clonic seizure: a videotape analysis.Neurology, 44(8):1403–1403, 1994
1994
-
[4]
Expansion of epileptogenic networks via neu- roplasticity in neural mass models.PLOS Computational Biology, 20(12):e1012666, 2024
Elif K¨ oksal-Ers¨ oz, Pascal Benquet, and Fabrice Wendling. Expansion of epileptogenic networks via neu- roplasticity in neural mass models.PLOS Computational Biology, 20(12):e1012666, 2024
2024
-
[5]
Multimodal wearable eeg, emg and accelerometry measurements improve the accuracy of tonic-clonic seizure detection.Physiological Measure- ment, 45(6):065005, 2024
Jingwei Zhang, Lauren Swinnen, Christos Chatzichristos, Victoria Broux, Renee Proost, Katrien Jansen, Benno Mahler, Nicolas Zabler, Nino Epitashvilli, Matthias Duempelmann, et al. Multimodal wearable eeg, emg and accelerometry measurements improve the accuracy of tonic-clonic seizure detection.Physiological Measure- ment, 45(6):065005, 2024
2024
-
[6]
Epilepsy: a clinical overview.The American Journal of Medicine, 134(7):840–847, 2021
Tracey A Milligan. Epilepsy: a clinical overview.The American Journal of Medicine, 134(7):840–847, 2021
2021
-
[7]
Biomolecular mechanisms of epileptic seizures and epilepsy: a review.Acta Epilep- tologica, 5(1):28, 2023
Komang Trisna Sumadewi, Saktivi Harkitasari, and David Christopher Tjandra. Biomolecular mechanisms of epileptic seizures and epilepsy: a review.Acta Epilep- tologica, 5(1):28, 2023
2023
-
[8]
Astrocyte chloride, excitatory-inhibitory balance and epilepsy.Neural regeneration research, 14 19(9):1887, 2024
Verena Untiet, Maiken Nedergaard, and Alexei Verkhratsky. Astrocyte chloride, excitatory-inhibitory balance and epilepsy.Neural regeneration research, 14 19(9):1887, 2024
2024
-
[9]
Simple model for the prediction of seizure durations.Physical Review E, 110(1):014401, 2024
Tyler Salners, Karin A Dahmen, and John Beggs. Simple model for the prediction of seizure durations.Physical Review E, 110(1):014401, 2024
2024
-
[10]
Chaos shapes transient synchrony activi- ties and switchings in the excitatory-inhibitory networks
Gaobiao Zhu, Yan Zhang, Jiao Wu, Muhua Zheng, and Kesheng Xu. Chaos shapes transient synchrony activi- ties and switchings in the excitatory-inhibitory networks. Nonlinear Dynamics, 112(9):7555–7570, 2024
2024
-
[11]
The role of inflam- mation in the development of epilepsy.Journal of neu- roinflammation, 15(1):144, 2018
Amna Rana and Alberto E Musto. The role of inflam- mation in the development of epilepsy.Journal of neu- roinflammation, 15(1):144, 2018
2018
-
[12]
Springer Nature, 2023
Richard M Kostrzewa.Handbook of neurotoxicity. Springer Nature, 2023
2023
-
[13]
Molecular mechanisms of epilepsy.Nature neuroscience, 18(3):367–372, 2015
Kevin Staley. Molecular mechanisms of epilepsy.Nature neuroscience, 18(3):367–372, 2015
2015
-
[14]
New epilepsy therapies in develop- ment.Nature Reviews Drug Discovery, 23(9):682–708, 2024
Pavel Klein, Rafal M Kaminski, Matthias Koepp, and Wolfgang L¨ oscher. New epilepsy therapies in develop- ment.Nature Reviews Drug Discovery, 23(9):682–708, 2024
2024
-
[15]
Excitatory/inhibitory balance in epilep- sies and neurodevelopmental disorders: Depolarizingγ- aminobutyric acid as a common mechanism.Epilepsia, 64(8):1975–1990, 2023
Eline JH van van Hugte, Dirk Schubert, and Nael Nadif Kasri. Excitatory/inhibitory balance in epilep- sies and neurodevelopmental disorders: Depolarizingγ- aminobutyric acid as a common mechanism.Epilepsia, 64(8):1975–1990, 2023
1975
-
[16]
Neuronal chloride and excitability—the big im- pact of small changes.Current opinion in neurobiology, 43:35–42, 2017
Joseph V Raimondo, Blake A Richards, and Melanie A Woodin. Neuronal chloride and excitability—the big im- pact of small changes.Current opinion in neurobiology, 43:35–42, 2017
2017
-
[17]
Chloride-dependent cation transport via slc12 carriers at atomic resolution.Annual Review of Physiology, 87(1):397–419, 2025
Eric Delpire. Chloride-dependent cation transport via slc12 carriers at atomic resolution.Annual Review of Physiology, 87(1):397–419, 2025
2025
-
[18]
Chloride trans- porters controlling neuronal excitability.Physiological Reviews, 103(2):1095–1135, 2023
Jessica C Pressey, Miranda de Saint-Rome, Vineeth A Raveendran, and Melanie A Woodin. Chloride trans- porters controlling neuronal excitability.Physiological Reviews, 103(2):1095–1135, 2023
2023
-
[19]
In- hibitory neurons and the asymmetric shape of neuronal avalanches.Physical Review E, 111(2):024133, 2025
Roberto Zaccariello, Hans J Herrmann, Alessandro Sar- racino, Stefano Zapperi, and Lucilla de Arcangelis. In- hibitory neurons and the asymmetric shape of neuronal avalanches.Physical Review E, 111(2):024133, 2025
2025
-
[20]
Role of nkcc1 and kcc2 in epilepsy: from expression to function.Frontiers in neurology, 10:1407, 2020
Ru Liu, Junling Wang, Shuli Liang, Guojun Zhang, and Xiaofeng Yang. Role of nkcc1 and kcc2 in epilepsy: from expression to function.Frontiers in neurology, 10:1407, 2020
2020
-
[21]
Chloride dynamics alter the input-output properties of neurons.PLoS computa- tional biology, 16(5):e1007932, 2020
Christopher B Currin, Andrew J Trevelyan, Colin J Ak- erman, and Joseph V Raimondo. Chloride dynamics alter the input-output properties of neurons.PLoS computa- tional biology, 16(5):e1007932, 2020
2020
-
[22]
A model for focal seizure onset, propagation, evo- lution, and progression.Elife, 9:e50927, 2020
Jyun-you Liou, Elliot H Smith, Lisa M Bateman, Samuel L Bruce, Guy M McKhann, Robert R Goodman, Ronald G Emerson, Catherine A Schevon, and LF Ab- bott. A model for focal seizure onset, propagation, evo- lution, and progression.Elife, 9:e50927, 2020
2020
-
[23]
Components of neuronal chloride transport in rat and human neocortex
Rudolf A Deisz, Thomas-N Lehmann, Peter Horn, Christoph Dehnicke, and Robert Nitsch. Components of neuronal chloride transport in rat and human neocortex. The Journal of physiology, 589(6):1317–1347, 2011
2011
-
[24]
Christopher Brian Currin and Joseph Valentino Rai- mondo. Computational models reveal how chloride dy- namics determine the optimal distribution of inhibitory synapses to minimise dendritic excitability.PLoS Com- putational Biology, 18(9):e1010534, 2022
2022
-
[25]
Biophysical models reveal the relative impor- tance of transporter proteins and impermeant anions in chloride homeostasis.Elife, 7:e39575, 2018
Kira M D¨ usterwald, Christopher B Currin, Richard J Burman, Colin J Akerman, Alan R Kay, and Joseph V Raimondo. Biophysical models reveal the relative impor- tance of transporter proteins and impermeant anions in chloride homeostasis.Elife, 7:e39575, 2018
2018
-
[26]
Enrico Cherubini, Graziella Di Cristo, and Massimo Avoli. Dysregulation of gabaergic signaling in neu- rodevelomental disorders: targeting cation-chloride co- transporters to re-establish a proper e/i balance.Fron- tiers in cellular neuroscience, 15:813441, 2022
2022
-
[27]
The role of gabaergic signalling in neurodevelopmental disorders
Xin Tang, Rudolf Jaenisch, and Mriganka Sur. The role of gabaergic signalling in neurodevelopmental disorders. Nature Reviews Neuroscience, 22(5):290–307, 2021
2021
-
[28]
Au- tonomic manifestations of epilepsy: emerging pathways to sudden death?Nature Reviews Neurology, 17(12):774– 788, 2021
Roland D Thijs, Philippe Ryvlin, and Rainer Surges. Au- tonomic manifestations of epilepsy: emerging pathways to sudden death?Nature Reviews Neurology, 17(12):774– 788, 2021
2021
-
[29]
Robert S Fisher, J Helen Cross, Jacqueline A French, Norimichi Higurashi, Edouard Hirsch, Floor E Jansen, Lieven Lagae, Solomon L Mosh´ e, Jukka Peltola, Eliane Roulet Perez, et al. Operational classification of seizure types by the international league against epilepsy: Posi- tion paper of the ilae commission for classification and terminology.Epilepsia,...
2017
-
[30]
Dynamics of pattern formation in lateral-inhibition type neural fields.Biological cybernet- ics, 27(2):77–87, 1977
Shun-ichi Amari. Dynamics of pattern formation in lateral-inhibition type neural fields.Biological cybernet- ics, 27(2):77–87, 1977
1977
-
[31]
Waves, bumps, and patterns in neural field theories.Biological cybernetics, 93(2):91–108, 2005
Stephen Coombes. Waves, bumps, and patterns in neural field theories.Biological cybernetics, 93(2):91–108, 2005
2005
-
[32]
Bayesian estimation inherent in a mexican-hat-type neural network.Physical Review E, 93(5):052303, 2016
Ken Takiyama. Bayesian estimation inherent in a mexican-hat-type neural network.Physical Review E, 93(5):052303, 2016
2016
-
[33]
Plastic systemic inhibition controls amplitude while allowing phase pattern in a stochastic neural field model.Physical Review E, 103(3):032311, 2021
Conor L Morrison, Priscilla E Greenwood, and Lawrence M Ward. Plastic systemic inhibition controls amplitude while allowing phase pattern in a stochastic neural field model.Physical Review E, 103(3):032311, 2021
2021
-
[34]
Control mecha- nisms in cortical epileptogenic foci*: surround inhibition
David A Prince and B Joe Wilder. Control mecha- nisms in cortical epileptogenic foci*: surround inhibition. Archives of neurology, 16(2):194–202, 1967
1967
-
[35]
Neuroelectro: a window to the world’s neuron electrophysiology data
Shreejoy J Tripathy, Judith Savitskaya, Shawn D Burton, Nathaniel N Urban, and Richard C Gerkin. Neuroelectro: a window to the world’s neuron electrophysiology data. Frontiers in neuroinformatics, 8:40, 2014
2014
-
[36]
Modeling spontaneous activity in the developing spinal cord using activity- dependent variations of intracellular chloride.Journal of Neuroscience, 25(14):3601–3612, 2005
Cristina Marchetti, Joel Tabak, Nikolai Chub, Michael J O’Donovan, and John Rinzel. Modeling spontaneous activity in the developing spinal cord using activity- dependent variations of intracellular chloride.Journal of Neuroscience, 25(14):3601–3612, 2005
2005
-
[37]
Epileptiform burst af- terhyperpolarization: calcium-dependent potassium po- tential in hippocampal ca1 pyramidal cells.Science, 210(4474):1122–1124, 1980
B Eo Alger and R Ao Nicoll. Epileptiform burst af- terhyperpolarization: calcium-dependent potassium po- tential in hippocampal ca1 pyramidal cells.Science, 210(4474):1122–1124, 1980
1980
-
[38]
Episodic activity in a heterogeneous excitatory network, from spiking neurons to mean field
Boris B Vladimirski, Jo¨ el Tabak, Michael J O’Donovan, and John Rinzel. Episodic activity in a heterogeneous excitatory network, from spiking neurons to mean field. Journal of computational neuroscience, 25:39–63, 2008
2008
-
[39]
Mexican hat wavelet transform and its applications
Abhishek Singh, Aparna Rawat, and Nikhila Raghuthaman. Mexican hat wavelet transform and its applications. InMethods of mathematical modelling and computation for complex systems, pages 299–317. Springer, 2021
2021
-
[40]
Cross-scale effects of neural interactions during human neocortical 15 seizure activity.Proceedings of the National Academy of Sciences, 114(40):10761–10766, 2017
Tahra L Eissa, Koen Dijkstra, Christoph Brune, Ronald G Emerson, Michel JAM van Putten, Robert R Goodman, Guy M McKhann Jr, Catherine A Schevon, Wim van Drongelen, and Stephan A van Gils. Cross-scale effects of neural interactions during human neocortical 15 seizure activity.Proceedings of the National Academy of Sciences, 114(40):10761–10766, 2017
2017
-
[41]
Role of inhibitory control in modulating focal seizure spread.Brain, 141(7):2083–2097, 2018
Jyun-you Liou, Hongtao Ma, Michael Wenzel, Mingrui Zhao, Eliza Baird-Daniel, Elliot H Smith, Andy Daniel, Ronald Emerson, Rafael Yuste, Theodore H Schwartz, et al. Role of inhibitory control in modulating focal seizure spread.Brain, 141(7):2083–2097, 2018
2083
-
[42]
Multivariate regression methods for estimating velocity of ictal discharges from human microelectrode recordings.Journal of neural engineering, 14(4):044001, 2017
Jyun-you Liou, Elliot H Smith, Lisa M Bateman, Guy M McKhann, Robert R Goodman, Bradley Greger, Tyler S Davis, Spencer S Kellis, Paul A House, and Catherine A Schevon. Multivariate regression methods for estimating velocity of ictal discharges from human microelectrode recordings.Journal of neural engineering, 14(4):044001, 2017
2017
-
[43]
Recovery of activation propagation and self-sustained os- cillation abilities in stroke brain networks.Physical Re- view E, 111(3):034309, 2025
Yingpeng Liu, Jiao Wu, Kesheng Xu, and Muhua Zheng. Recovery of activation propagation and self-sustained os- cillation abilities in stroke brain networks.Physical Re- view E, 111(3):034309, 2025
2025
-
[44]
Cation-chloride cotrans- porters in neuronal development, plasticity and disease
Kai Kaila, Theodore J Price, John A Payne, Martin Puskarjov, and Juha Voipio. Cation-chloride cotrans- porters in neuronal development, plasticity and disease. Nature reviews neuroscience, 15(10):637–654, 2014
2014
-
[45]
Chloride regulation: a dynamic equilibrium crucial for synaptic inhibition.Neuron, 89(6):1157–1172, 2016
Nicolas Doyon, Laurent Vinay, Steven A Prescott, and Yves De Koninck. Chloride regulation: a dynamic equilibrium crucial for synaptic inhibition.Neuron, 89(6):1157–1172, 2016
2016
-
[46]
Spiral wave dynamics in neocortex.Neuron, 68(5):978–990, 2010
Xiaoying Huang, Weifeng Xu, Jianmin Liang, Kentaroh Takagaki, Xin Gao, and Jian-young Wu. Spiral wave dynamics in neocortex.Neuron, 68(5):978–990, 2010
2010
-
[47]
Semiology of epileptic seizures: a critical review.Epilepsy & Behavior, 15(1):2–9, 2009
Soheyl Noachtar and Astrid S Peters. Semiology of epileptic seizures: a critical review.Epilepsy & Behavior, 15(1):2–9, 2009
2009
-
[48]
Chloride dysregulation, seizures, and cerebral edema: a relationship with therapeutic potential.Trends in neu- rosciences, 40(5):276–294, 2017
Joseph Glykys, Volodymyr Dzhala, Kiyoshi Egawa, Kristopher T Kahle, Eric Delpire, and Kevin Staley. Chloride dysregulation, seizures, and cerebral edema: a relationship with therapeutic potential.Trends in neu- rosciences, 40(5):276–294, 2017
2017
-
[49]
Sin- auer, 2001
Bertil Hille.Ion channels of excitable membranes. Sin- auer, 2001
2001
-
[50]
Cambridge Univer- sity Press, 2014
Wulfram Gerstner, Werner M Kistler, Richard Naud, and Liam Paninski.Neuronal dynamics: From single neurons to networks and models of cognition. Cambridge Univer- sity Press, 2014
2014
-
[51]
Focal seizures are organized by feedback between neural activity and ion concentration changes.elife, 11:e68541, 2022
Damiano Gentiletti, Marco de Curtis, Vadym Gnatkovsky, and Piotr Suffczynski. Focal seizures are organized by feedback between neural activity and ion concentration changes.elife, 11:e68541, 2022
2022
-
[52]
Preictal dysfunctions of inhibitory interneurons para- doxically lead to their rebound hyperactivity and to low-voltage-fast onset seizures in dravet syndrome
Fabrizio Capitano, Mathieu Kuchenbuch, Jennifer Lav- igne, Hava Chaptoukaev, Maria A Zuluaga, Marco Lorenzi, Rima Nabbout, and Massimo Mantegazza. Preictal dysfunctions of inhibitory interneurons para- doxically lead to their rebound hyperactivity and to low-voltage-fast onset seizures in dravet syndrome. Proceedings of the National Academy of Sciences, 1...
2024
-
[53]
Duo Li, Qiang Li, and Rui Zhang. Dynamical modeling and analysis of epileptic discharges transition caused by glutamate release with metabolism processes regulation from astrocyte.Chaos: An Interdisciplinary Journal of Nonlinear Science, 34(12), 2024
2024
-
[54]
Maxime Guye, Gaelle Bettus, Fabrice Bartolomei, and Patrick J Cozzone. Graph theoretical analysis of struc- tural and functional connectivity mri in normal and pathological brain networks.Magnetic Resonance Ma- terials in Physics, Biology and Medicine, 23(5):409–421, 2010
2010
-
[55]
Computer modelling of epilepsy.Na- ture Reviews Neuroscience, 9(8):626–637, 2008
William W Lytton. Computer modelling of epilepsy.Na- ture Reviews Neuroscience, 9(8):626–637, 2008
2008
-
[56]
A quantitative description of membrane current and its application to conduction and excitation in nerve.The Journal of phys- iology, 117(4):500, 1952
Alan L Hodgkin and Andrew F Huxley. A quantitative description of membrane current and its application to conduction and excitation in nerve.The Journal of phys- iology, 117(4):500, 1952
1952
-
[57]
The dynamic brain: from spiking neurons to neural masses and cor- tical fields.PLoS computational biology, 4(8):e1000092, 2008
Gustavo Deco, Viktor K Jirsa, Peter A Robinson, Michael Breakspear, and Karl Friston. The dynamic brain: from spiking neurons to neural masses and cor- tical fields.PLoS computational biology, 4(8):e1000092, 2008
2008
-
[58]
Zayneb Brari and Safya Belghith. A new algorithm for largest lyapunov exponent determination for noisy chaotic signal studies with application to electroen- cephalographic signals analysis for epilepsy and epilep- tic seizures detection.Chaos, Solitons & Fractals, 165:112757, 2022
2022
-
[59]
Noise-induced coexisting firing patterns in hybrid-synaptic interacting networks.Physica A: Sta- tistical Mechanics and its Applications, 615:128591, 2023
Xinyi Wang, Xiyun Zhang, Muhua Zheng, Leijun Xu, and Kesheng Xu. Noise-induced coexisting firing patterns in hybrid-synaptic interacting networks.Physica A: Sta- tistical Mechanics and its Applications, 615:128591, 2023
2023
-
[60]
The neurobiology of interoception and affect.Trends in Cog- nitive Sciences, 28(7):643–661, 2024
MJ Feldman, E Bliss-Moreau, and KA Lindquist. The neurobiology of interoception and affect.Trends in Cog- nitive Sciences, 28(7):643–661, 2024
2024
-
[61]
Short-term plasticity as a mech- anism to regulate and retain multistability.Chaos, Soli- tons&Fractals, 165:112891, 2022
Xinjia Zhou, Changhai Tian, Xiyun Zhang, Muhua Zheng, and Kesheng Xu. Short-term plasticity as a mech- anism to regulate and retain multistability.Chaos, Soli- tons&Fractals, 165:112891, 2022
2022
-
[62]
Formation and maintenance of neu- ronal collective dynamics through local perturbation and intrinsic node dynamics.Chaos, Solitons&Fractals, 199:116648, 2025
Runzhou Liu, Yan Zhang, Jijun Wang, Muhua Zheng, and Kesheng Xu. Formation and maintenance of neu- ronal collective dynamics through local perturbation and intrinsic node dynamics.Chaos, Solitons&Fractals, 199:116648, 2025
2025
-
[63]
Kun Shan, Changhai Tian, Zhigang Zheng, Muhua Zheng, and Kesheng Xu. Short-term plasticity pro- motes synchronization of coupled chaotic oscillators in excitatory–inhibitory networks.Chaos: An Interdisci- plinary Journal of Nonlinear Science, 35(7), 2025
2025
-
[64]
Low-calcium epileptiform activity in the hippocampus in vivo.Journal of neurophysiology, 90(4):2253–2260, 2003
Zhouyan Feng and Dominique M Durand. Low-calcium epileptiform activity in the hippocampus in vivo.Journal of neurophysiology, 90(4):2253–2260, 2003
2003
-
[65]
Propagation of non-synaptic epilep- tiform activity across a lesion in rat hippocampal slices
Jun Lian, Marom Bikson, Jianwei Shuai, and Do- minique M Durand. Propagation of non-synaptic epilep- tiform activity across a lesion in rat hippocampal slices. The Journal of physiology, 537(Pt 1):191, 2001
2001
-
[66]
Chemical synaptic transmission is not necessary for epileptic seizures to persist in the baboon papio papio.Experimental neurology, 89(1):250–258, 1985
R Pumain, Ch Menini, U Heinemann, J Louvel, and C Silva-Barrat. Chemical synaptic transmission is not necessary for epileptic seizures to persist in the baboon papio papio.Experimental neurology, 89(1):250–258, 1985
1985
-
[67]
Tresk channel contributes to depolarization-induced shunting inhibition and mod- ulates epileptic seizures.Cell Reports, 36(3), 2021
Weiyuan Huang, Yue Ke, Jianping Zhu, Shuai Liu, Jin Cong, Hailin Ye, Yanwu Guo, Kewan Wang, Zhenhai Zhang, Wenxiang Meng, et al. Tresk channel contributes to depolarization-induced shunting inhibition and mod- ulates epileptic seizures.Cell Reports, 36(3), 2021
2021
-
[68]
Pelkey, Ramesh Chittajallu, Michael T
Kenneth A. Pelkey, Ramesh Chittajallu, Michael T. Craig, Ludovic Tricoire, Jason C. Wester, and Chris J. McBain. Hippocampal gabaergic inhibitory interneurons. Physiological Reviews, 97(4):1619–1747, 2017
2017
-
[69]
Fast activity at seizure onset is mediated by inhibitory circuits in the entorhinal cortex 16 in vitro.Annals of Neurology, 64(6):674–686, 2008
Vadym Gnatkovsky, Laura Librizzi, Federica Trombin, and Marco de Curtis. Fast activity at seizure onset is mediated by inhibitory circuits in the entorhinal cortex 16 in vitro.Annals of Neurology, 64(6):674–686, 2008
2008
-
[70]
Eissa, Koen Dijkstra, Christoph Brune, Ronald G
Tahra L. Eissa, Koen Dijkstra, Christoph Brune, Ronald G. Emerson, Michel J. A. M. van Putten, Robert R. Goodman, Guy M. McKhann, Catherine A. Schevon, Wim van Drongelen, and Stephan A. van Gils. Cross-scale effects of neural interactions during human neocortical seizure activity.Proceedings of the National Academy of Sciences, 114(40):10761–10766, 2017
2017
-
[71]
Potassium dynamics and seizures: Why is potassium ictogenic?Epilepsy research, 143:50–59, 2018
Marco de Curtis, Laura Uva, Vadym Gnatkovsky, and Laura Librizzi. Potassium dynamics and seizures: Why is potassium ictogenic?Epilepsy research, 143:50–59, 2018
2018
-
[72]
In vivo optical mapping of epileptic foci and surround inhibition in ferret cerebral cortex, 2001
Theodore H Schwartz and Tobias Bonhoeffer. In vivo optical mapping of epileptic foci and surround inhibition in ferret cerebral cortex, 2001
2001
-
[73]
Microphysiology of epileptiform activity in human neocortex.Journal of Clinical Neuro- physiology, 25(6):321–330, 2008
Catherine A Schevon, Sau K Ng, Joshua Cappell, Robert R Goodman, Guy McKhann Jr, Allen Waziri, Al- mut Branner, Alexandre Sosunov, Charles E Schroeder, and Ronald G Emerson. Microphysiology of epileptiform activity in human neocortex.Journal of Clinical Neuro- physiology, 25(6):321–330, 2008
2008
-
[74]
Multiarmed spirals in excitable media.Physical review letters, 78(12):2489, 1997
Bakthier Vasiev, Florian Siegert, and Cornelis Weijer. Multiarmed spirals in excitable media.Physical review letters, 78(12):2489, 1997
1997
-
[75]
An experimental- ist’s approach to accurate localization of phase singular- ities during reentry.Annals of biomedical engineering, 29(1):47–59, 2001
Anand N Iyer and Richard A Gray. An experimental- ist’s approach to accurate localization of phase singular- ities during reentry.Annals of biomedical engineering, 29(1):47–59, 2001
2001
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.