Recognition: unknown
Skull-Conforming Acoustic Holographic Lenses for Transcranial Targeting
Pith reviewed 2026-05-08 13:17 UTC · model grok-4.3
The pith
Skull-conforming acoustic holographic lenses encode individualized wavefront corrections to achieve precise transcranial ultrasound focusing.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
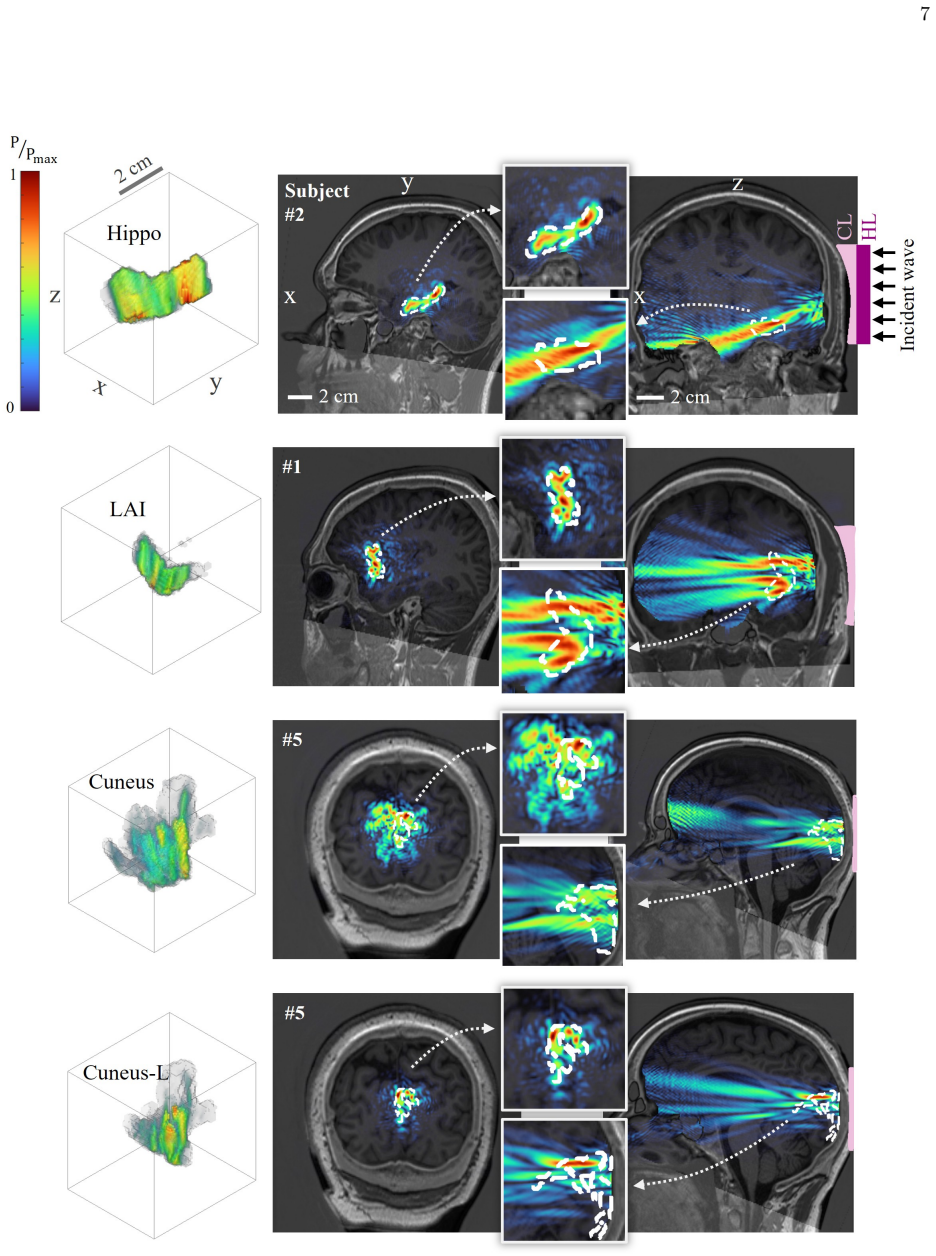
Within the subject-specific volumetric holography framework, cranial geometry and therapeutic constraints are embedded into a physics-based optimization pipeline that synthesizes holographic phase patterns. The resulting lens is paired with a skull- and skin-conforming coupling layer that enhances impedance continuity and reduces reflection losses, allowing simultaneous aberration mitigation and efficient transcranial transmission. Numerical simulations demonstrate consistent volumetric focusing and target coverage within safety limits, and ex vivo validation on a human skull confirms accurate fabrication and faithful reconstruction of the designed acoustic fields.
What carries the argument
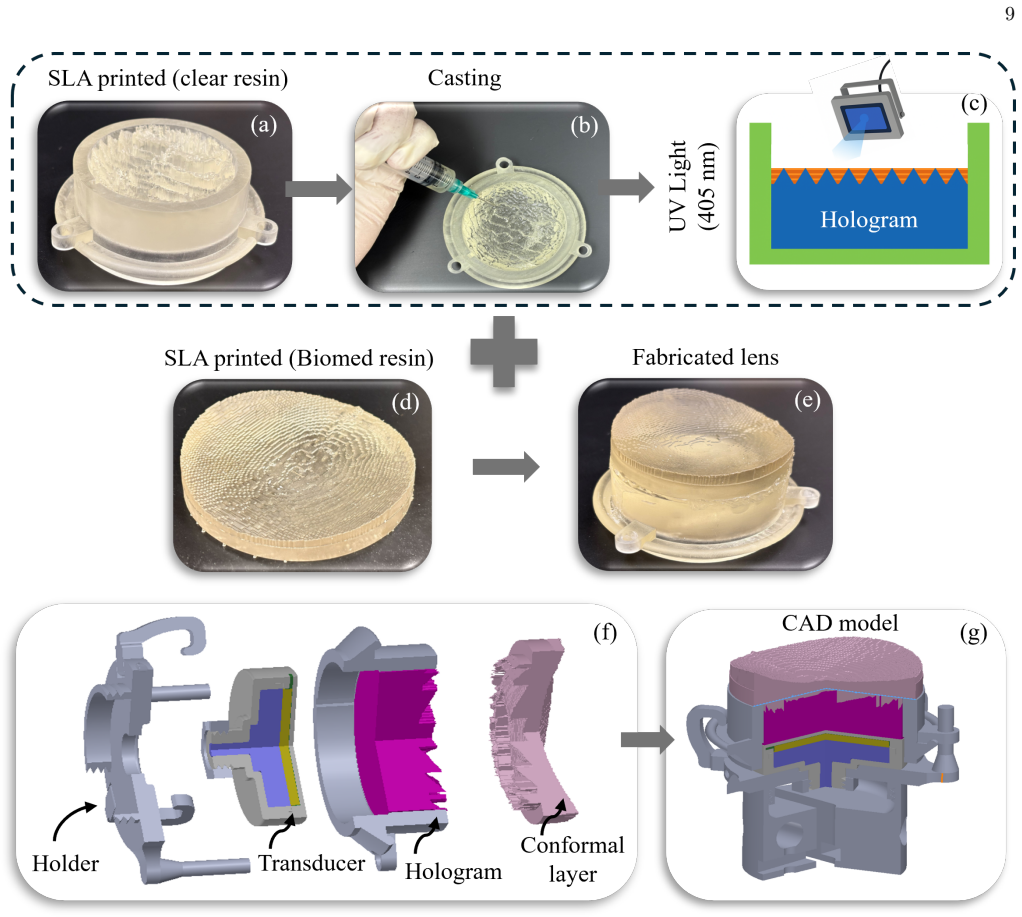
The subject-specific volumetric holography (SSVH) optimization pipeline, which embeds cranial geometry and therapeutic constraints into physics-based phase synthesis for the holographic lens, integrated with a conformal acoustic coupling layer.
If this is right
- Consistent volumetric focusing and reliable target coverage are maintained across multiple subjects and therapeutic targets.
- Pressure fields remain within established safety limits during transmission.
- Accurate fabrication of the lenses enables faithful reconstruction of the designed three-dimensional acoustic fields.
- The conformal coupling layer reduces reflection losses and stabilizes alignment for efficient energy delivery.
Where Pith is reading between the lines
- The physical encoding of corrections into a removable lens could simplify clinical workflows by reducing dependence on high-channel-count phased arrays.
- Adaptation to other irregular bony structures, such as the ribs or pelvis, may extend the same conformal holography principle to additional ultrasound therapies.
- Real-time recalibration of the lens design using intraoperative imaging could address slow anatomical shifts between planning and treatment.
Load-bearing premise
The optimization pipeline and single-skull ex vivo results will accurately predict performance once tissue properties, motion, and long-term coupling in living subjects introduce additional unmodeled effects.
What would settle it
A side-by-side in vivo measurement of the actual pressure field versus the simulated field, showing whether the focal position or amplitude deviates beyond acceptable clinical tolerances under realistic breathing and tissue variation.
Figures



read the original abstract
Transcranial focused ultrasound (tFUS) offers noninvasive access to deep brain circuits but remains limited by skull-induced phase aberration, acoustic impedance mismatch, and poor volumetric control of intracranial pressure fields. Conventional phased-array and planar holographic strategies compensate aberrations electronically or computationally, yet do not resolve geometric and coupling inconsistencies imposed by subject-specific cranial morphology. We introduce personalized skull-conforming acoustic holograms that physically encode individualized wavefront corrections into a conformal acoustic interface. Within a subject-specific volumetric holography (SSVH) framework, cranial geometry and therapeutic constraints are embedded into a physics-based optimization pipeline for holographic phase synthesis. The resulting lens is integrated with a skull- and skin-conforming coupling layer that enhances impedance continuity, reduces reflection losses, and stabilizes spatial alignment, enabling simultaneous aberration mitigation and efficient transcranial transmission. Numerical simulations across multiple subjects and targets demonstrate consistent volumetric focusing and reliable target coverage while maintaining pressure fields within safety limits. Experimental validation using an ex vivo human skull confirms accurate fabrication, effective acoustic coupling, and faithful reconstruction of designed three-dimensional acoustic fields. By unifying wavefront engineering with anatomical conformity, this work establishes skull-conforming acoustic holography as a scalable strategy for high-fidelity, anatomically adaptive transcranial ultrasound targeting.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript introduces personalized skull-conforming acoustic holographic lenses that physically encode subject-specific wavefront corrections via a subject-specific volumetric holography (SSVH) framework. The approach combines physics-based optimization of holographic phase synthesis with a conformal coupling layer to mitigate skull aberrations, reduce reflections, and stabilize alignment for transcranial focused ultrasound. Numerical simulations across multiple subjects and targets demonstrate consistent volumetric focusing within safety limits, while experimental validation on a single ex vivo human skull confirms fabrication accuracy and faithful reconstruction of the designed 3D pressure fields. The authors conclude that unifying wavefront engineering with anatomical conformity establishes skull-conforming acoustic holography as a scalable strategy for high-fidelity, anatomically adaptive transcranial targeting.
Significance. If the central claims hold, the work offers a practical physical alternative to electronic phased arrays for subject-specific tFUS, potentially improving targeting precision, transmission efficiency, and safety by addressing both phase aberrations and geometric/coupling mismatches in one conformal device. The multi-subject simulation campaign and ex vivo fabrication/pressure-field test provide concrete evidence of feasibility and are strengths that could accelerate translation if extended.
major comments (2)
- Abstract: the claims of 'consistent volumetric focusing' and 'faithful reconstruction of designed three-dimensional acoustic fields' are presented without any quantitative metrics (e.g., focal pressure error, targeting accuracy, or standard deviation across subjects), error bars, or statistical analysis; this directly limits evaluation of whether the data support the performance assertions.
- Experimental Validation and Conclusion: the scalability claim ('scalable strategy for high-fidelity, anatomically adaptive transcranial ultrasound targeting') rests on simulations plus a single ex vivo skull; no data or analysis address in vivo factors such as tissue-property variability, respiratory/cardiac motion, or time-varying acoustic coupling that would perturb the designed wavefronts, making the extrapolation load-bearing for the central claim.
Simulated Author's Rebuttal
We thank the referee for the constructive comments. We address each major point below, revising the manuscript where appropriate to strengthen clarity and accuracy.
read point-by-point responses
-
Referee: [—] Abstract: the claims of 'consistent volumetric focusing' and 'faithful reconstruction of designed three-dimensional acoustic fields' are presented without any quantitative metrics (e.g., focal pressure error, targeting accuracy, or standard deviation across subjects), error bars, or statistical analysis; this directly limits evaluation of whether the data support the performance assertions.
Authors: We agree that the abstract would benefit from explicit quantitative support. In the revised manuscript we have incorporated key metrics drawn from the multi-subject simulation campaign and ex vivo measurements, including focal-pressure error, targeting accuracy, and standard deviations across subjects, together with reference to error bars and statistical summaries already present in the results. These additions directly substantiate the claims of consistent volumetric focusing and faithful field reconstruction. revision: yes
-
Referee: [—] Experimental Validation and Conclusion: the scalability claim ('scalable strategy for high-fidelity, anatomically adaptive transcranial ultrasound targeting') rests on simulations plus a single ex vivo skull; no data or analysis address in vivo factors such as tissue-property variability, respiratory/cardiac motion, or time-varying acoustic coupling that would perturb the designed wavefronts, making the extrapolation load-bearing for the central claim.
Authors: The referee correctly notes that the experimental component is limited to one ex vivo skull and that dynamic in vivo effects are not examined. The multi-subject simulations already quantify robustness to anatomical variation, and the ex vivo test confirms fabrication fidelity and acoustic performance. We have revised the conclusion to qualify the scalability statement, presenting the work as a foundational demonstration whose translation to clinical settings will require dedicated in vivo studies of motion and coupling variability. This adjustment preserves the strength of the reported evidence while accurately delimiting its scope. revision: partial
- The manuscript contains no in vivo data addressing tissue-property variability, respiratory/cardiac motion, or time-varying acoustic coupling; these factors cannot be quantified without additional experiments.
Circularity Check
No significant circularity; claims rest on independent physics modeling and ex-vivo validation
full rationale
The derivation chain proceeds from subject-specific cranial geometry and therapeutic constraints through a physics-based optimization pipeline (SSVH) to generate holographic phase maps, followed by numerical simulations across multiple subjects and direct experimental pressure-field measurements on one ex-vivo skull. None of these steps reduce to self-definition, fitted parameters renamed as predictions, or load-bearing self-citations; the optimization is governed by external wave-propagation physics and the validation data are acquired independently of the final claims. The abstract and described framework contain no equations or premises that are tautological with their inputs.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Physics-based models of ultrasound propagation through heterogeneous skull geometry accurately predict phase aberrations and pressure fields.
Reference graph
Works this paper leans on
-
[1]
Transcranial focused ultrasound (tFUS) offers noninvasive access to deep brain circuits but remains limited by skull-induced phase aberration, acoustic impedance mismatch, and poor volumetric control of intracranial pressure fields. Conventional phased-array and planar holographic strategies compensate aberrations elec- tronically or computationally, yet ...
2014
-
[2]
Subject- specific frameworks may offer a practical pathway toward overcoming these limitations by explicitly incorporating anatomical and acoustic variability into ultrasound de- livery. The importance of such individualized strategies has been further underscored by the ITRUSST (Interna- tional Consortium for Transcranial Ultrasonic Stimula- tion Safety ...
work page internal anchor Pith review Pith/arXiv arXiv 2026
-
[3]
BioMed Elastic 50A Resin Manufacturing Guide,
Although computationally demanding, this method has been demonstrated to be applicable in heterogeneous me- dia for acoustic holography applications 25,31,55, and is therefore included in our SSVH framework. The optimal thickness map determined by the HHO method is used as input for the k-Wave simulations. Both the hologram and the secondary conforming la...
-
[4]
54B. E. Treeby and B. T. Cox, Journal of biomedical optics15, 021314 (2010). 55D. Andr´ es, N. Jim´ enez, J. M. Benlloch, and F. Camarena, Ul- trasound in Medicine & Biology48, 872 (2022)
2010
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.