Recognition: unknown
Hierarchical organization of critical brain dynamics
Pith reviewed 2026-05-08 13:03 UTC · model grok-4.3
The pith
Criticality signatures in the brain vary systematically along the anatomical hierarchy, but static and dynamic exponents point in opposite directions.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Signatures of criticality are not uniform, but instead vary systematically along the known anatomical hierarchy in both brain systems. The direction along this gradient is inconsistent across different criticality exponents, revealing a nontrivial, measure-dependent organization: exponents based on static properties point to a gradient in one direction, while the exponent based on dynamic properties points in the opposite direction. Moreover, the signatures across the visual system are strongly modulated by the engagement in a visual task. Correlations among criticality markers of different brain regions during active engagement are sufficient to reconstruct the anatomical hierarchy from the
What carries the argument
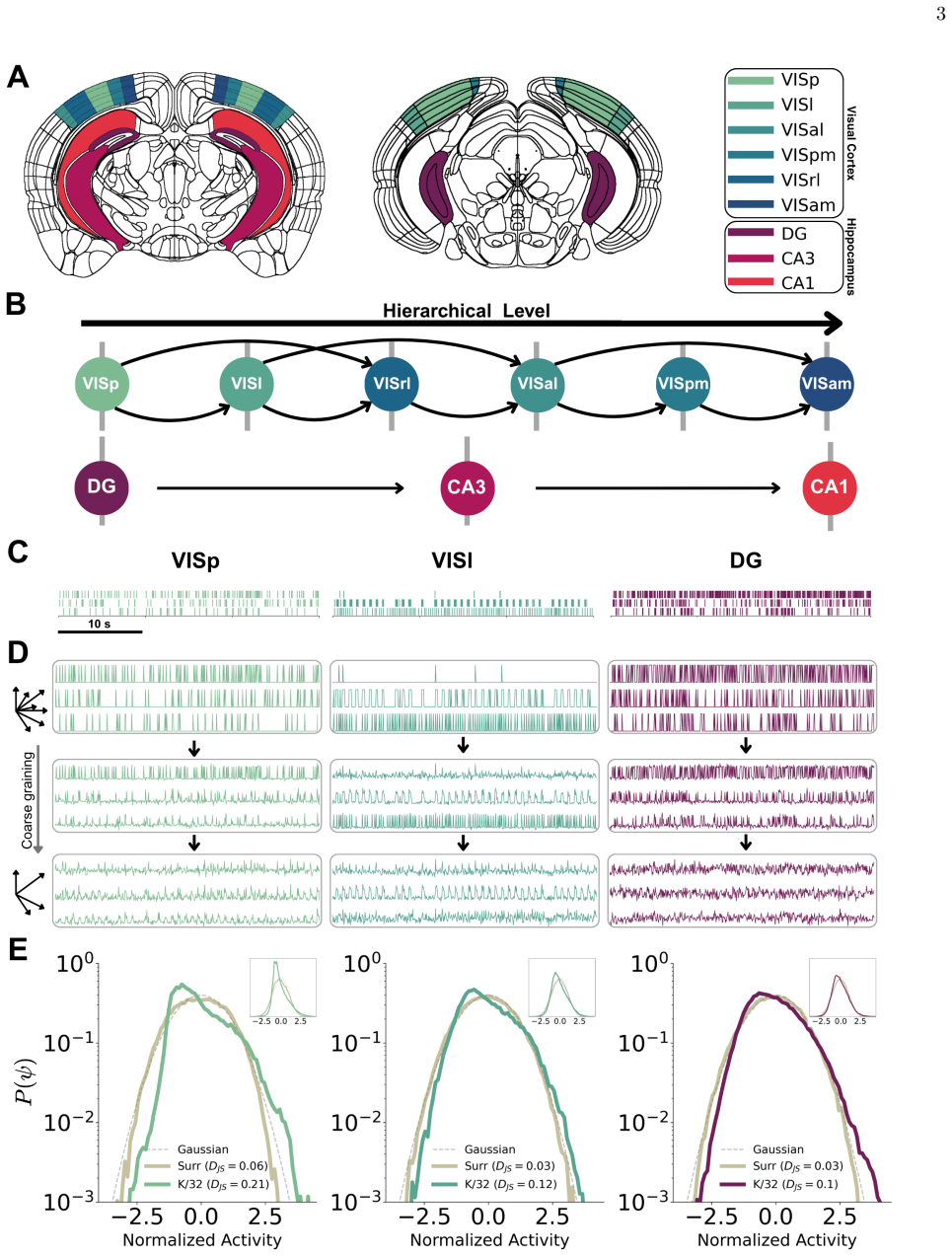
Phenomenological renormalization group methods applied to large-scale neuronal spiking activity to extract criticality exponents that vary with position in the anatomical hierarchy.
Load-bearing premise
The phenomenological renormalization group methods correctly extract comparable criticality exponents from spiking data across regions, and the anatomical hierarchy is known independently of those dynamic measures.
What would settle it
Applying the same renormalization-group analysis and finding that criticality exponents remain uniform across regions or that static and dynamic exponents align in the same direction along the hierarchy would falsify the claim of systematic, measure-dependent organization.
Figures





read the original abstract
The hierarchical organization of the brain is a fundamental structural principle, while brain criticality is a leading hypothesis for its collective dynamics. However, the connection between structure and signatures of criticality remains an open question. Here, we address this issue by applying phenomenological renormalization group approaches to large-scale neuronal spiking activity from the mouse visual cortex and hippocampus. We find that signatures of criticality are not uniform, but instead vary systematically along the known anatomical hierarchy in both brain systems. Strikingly, the direction along this gradient is inconsistent across different criticality exponents, revealing a nontrivial, measure-dependent organization: exponents based on static properties point to a gradient in one direction, while the exponent based on dynamic properties points in the opposite direction. Moreover, the signatures across the visual system are strongly modulated by the engagement in a visual task. We show that the correlations among criticality markers of different brain regions during active engagement are sufficient to reconstruct the anatomical hierarchy from the dynamics. Scaling exponents closely follow a theoretically predicted scaling relation among them, and covary with the hierarchical position. Our findings provide a direct link between the collective dynamics of neurons and the macroscopic architecture of the brain.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper applies phenomenological renormalization group (PRG) methods to large-scale spiking activity from mouse visual cortex and hippocampus. It claims that criticality signatures vary systematically along the known anatomical hierarchy in both systems, with static-property exponents and dynamic-property exponents exhibiting gradients in opposite directions. Task engagement modulates the signatures in the visual system, correlations among markers suffice to reconstruct the anatomical hierarchy from dynamics, and the exponents closely follow a theoretically predicted scaling relation while covarying with hierarchical position.
Significance. If the central claims hold after addressing methodological concerns, this work would provide a direct empirical link between collective neural dynamics and macroscopic brain architecture, demonstrating nontrivial measure-dependent organization of criticality signatures. The reported adherence of exponents to a theoretical scaling relation supplies a valuable internal consistency check. Such findings could inform models of how structural hierarchies shape brain function and enable inference of anatomical organization from dynamic measures.
major comments (2)
- The PRG procedure applied to spiking data assumes that extracted criticality exponents are comparable across regions despite substantial differences in neuron density, laminar organization, and sampling fraction (e.g., visual cortex versus hippocampal subregions). No validation is described for whether coarse-graining or scaling analysis is insensitive to effective system size or event rate, which is load-bearing for the claims of systematic hierarchical variation and opposite gradient directions between static and dynamic exponents.
- The abstract and main text supply no information on data preprocessing, statistical tests, error estimation, sample sizes, or controls for multiple comparisons. This absence prevents evaluation of the robustness of the reported gradients, task modulation, and hierarchy reconstruction from marker correlations.
minor comments (2)
- The abstract states clear findings but omits basic methodological context such as the number of animals or recordings, which would help readers assess the scale of the evidence.
- Notation distinguishing static versus dynamic criticality exponents should be introduced explicitly early in the text to prevent reader confusion when discussing their opposing gradients.
Simulated Author's Rebuttal
We thank the referee for the constructive feedback and for recognizing the potential significance of linking criticality signatures to brain architecture. We address each major comment below with clarifications and commitments to revision.
read point-by-point responses
-
Referee: The PRG procedure applied to spiking data assumes that extracted criticality exponents are comparable across regions despite substantial differences in neuron density, laminar organization, and sampling fraction (e.g., visual cortex versus hippocampal subregions). No validation is described for whether coarse-graining or scaling analysis is insensitive to effective system size or event rate, which is load-bearing for the claims of systematic hierarchical variation and opposite gradient directions between static and dynamic exponents.
Authors: We appreciate this methodological concern. The PRG implementation used identical coarse-graining rules, binning, and scaling analysis parameters across all regions to promote comparability, and the resulting gradients align closely with the known anatomical hierarchy, which serves as an external consistency check. Nevertheless, to strengthen the claims, we will add explicit validation analyses in the revision, including (i) subsampling of high-density regions to match lower-density event rates and neuron counts, (ii) systematic variation of coarse-graining thresholds to confirm exponent stability, and (iii) a supplementary table reporting effective system sizes and firing rates per region. These additions will be discussed in a new paragraph in the Methods and Results sections. revision: yes
-
Referee: The abstract and main text supply no information on data preprocessing, statistical tests, error estimation, sample sizes, or controls for multiple comparisons. This absence prevents evaluation of the robustness of the reported gradients, task modulation, and hierarchy reconstruction from marker correlations.
Authors: We agree that these details should be more accessible in the main text. The supplementary Methods already contain the full preprocessing pipeline (spike sorting with Kilosort2, quality metrics, and inclusion criteria), statistical procedures (linear mixed-effects models for gradients, Pearson correlations for hierarchy reconstruction, bootstrap-derived 95% confidence intervals for exponents), sample sizes (n = 5 mice for visual cortex, n = 3 for hippocampus), and multiple-comparison correction (FDR at q < 0.05). In the revised manuscript we will (i) insert a concise Methods paragraph summarizing these elements, (ii) add error bars or shaded confidence intervals to all relevant figures, and (iii) explicitly state the sample sizes and correction method in the figure legends and Results text. revision: yes
Circularity Check
No significant circularity; derivation relies on independent anatomical hierarchy and external theoretical scaling check.
full rationale
The paper applies phenomenological renormalization group methods to spiking data from known anatomical regions, reports gradients in criticality exponents along the independently established hierarchy, and notes that exponents follow a theoretically predicted scaling relation. The hierarchy reconstruction from marker correlations is presented as validation against an external structural reference, not as a self-derived input. No quoted steps reduce a claimed prediction or uniqueness result to a fitted parameter or self-citation by construction. The PRG procedure is treated as a standard tool whose cross-region comparability is assumed but not shown to be tautological within the paper's equations.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption Phenomenological renormalization group methods can be applied to neuronal spiking activity to extract comparable criticality exponents across brain regions
Reference graph
Works this paper leans on
-
[1]
Each recording session lasted approximately 2 hours and 25 minutes
Data For this study, we used a publicly available dataset pro- vided by the Allen Brain Institute [41], which consists of large-scale neuronal recordings from multiple brain ar- eas in mice using Neuropixels 1.0 probes. Each recording session lasted approximately 2 hours and 25 minutes. The experiment was divided into three main parts. In the first part, ...
-
[2]
This procedure eliminates correlations between spikes while preserving firing statistics, providing reference values for compari- son
Surrogate data To evaluate how our results differ from chance, we gen- erated surrogate data by shuffling the inter-spike intervals of each unit within the 30-s time window. This procedure eliminates correlations between spikes while preserving firing statistics, providing reference values for compari- son
-
[3]
Starting from the covariance matrix, Cij =⟨σ iσj⟩,(A1) whereσ i(t) denotes the time series of activity of uniti, we compute its ranked eigenvaluesλ 1 > λ2 >
Momentum-space phenomenological renormalization To coarse-grain the activity and obtain normalized dis- tributions, we employ the phenomenological renormaliza- tion procedure [27, 29, 45]. Starting from the covariance matrix, Cij =⟨σ iσj⟩,(A1) whereσ i(t) denotes the time series of activity of uniti, we compute its ranked eigenvaluesλ 1 > λ2 > . . . > λ N...
-
[4]
Jensen-Shannon distance correlations Applying the PRG procedure within 30-s windows yields a time series ofD JS values for each experiment. To investigate the dynamics of brain activity, we computed the Pearson correlation between theD JS time series of two different brain areas within the same session: Corr(Di JS (t), Dj JS (t)) = Cov(Di JS , Dj JS ) σDi...
-
[5]
Modularity and communities We employed thenetworkxPython library [58] to com- pute community structure in the network using the Lou- vain algorithm. The quality of a given partition was as- sessed using the modularity function [59, 60], defined as Q= 1 2M X i̸=j Aij −γ kikj 2M δ(ci, cj),(A9) whereA ij denotes the adjacency matrix,k i andk j are the degree...
-
[6]
5 and Fig
Phenomenological renormalization exponents To extract PRG scaling exponents shown in Fig. 5 and Fig. 6, we employ an alternative phenomenologi- cal renormalization scheme based on real-space coarse- graining [28]. The procedure proceeds by iteratively merging neural activity variables according to their pair- wise correlations. At each step, the two neuro...
-
[7]
Linkenkaer-Hansen, V
K. Linkenkaer-Hansen, V. V. Nikouline, J. M. Palva, and R. J. Ilmoniemi, Long-Range Temporal Correlations and Scaling Behavior in Human Brain Oscillations, J. Neu- rosci.21, 1370 (2001)
2001
-
[8]
J. M. Beggs and D. Plenz, Neuronal Avalanches in Neo- cortical Circuits, J. Neurosci.23, 11167 (2003)
2003
-
[9]
D. R. Chialvo, Emergent complex neural dynamics, Nat. Phys.6, 744 (2010)
2010
-
[10]
M. A. Mu˜ noz, Colloquium: Criticality and dynamical scaling in living systems, Rev. Mod. Phys.90, 031001 (2018)
2018
-
[11]
Tomen, M
N. Tomen, M. J. Herrmann, and U. Ernst,The Func- tional Role of Critical Dynamics in Neural Systems (Springer, Cham, Switzerland, 2019)
2019
-
[12]
O’Byrne and K
J. O’Byrne and K. Jerbi, How critical is brain criticality?, Trends Neurosci.45, 820 (2022)
2022
-
[13]
Bertschinger and T
N. Bertschinger and T. Natschl¨ ager, Real-Time Compu- tation at the Edge of Chaos in Recurrent Neural Net- works, Neural Comput.16, 1413 (2004)
2004
-
[14]
Haldeman and J
C. Haldeman and J. M. Beggs, Critical Branching Cap- tures Activity in Living Neural Networks and Maximizes the Number of Metastable States, Phys. Rev. Lett.94, 058101 (2005)
2005
-
[15]
Kinouchi and M
O. Kinouchi and M. Copelli, Optimal dynamical range of excitable networks at criticality, Nat. Phys.2, 348 (2006)
2006
-
[16]
J. M. Beggs, The criticality hypothesis: how local cortical networks might optimize information processing, Philos. Trans. R. Soc. London, Ser. A366, 329 (2008)
2008
-
[17]
W. L. Shew, H. Yang, T. Petermann, R. Roy, and D. Plenz, Neuronal Avalanches Imply Maximum Dy- namic Range in Cortical Networks at Criticality, J. Neu- rosci.29, 15595 (2009)
2009
-
[18]
W. L. Shew, H. Yang, S. Yu, R. Roy, and D. Plenz, In- formation Capacity and Transmission Are Maximized in Balanced Cortical Networks with Neuronal Avalanches, J. Neurosci.31, 55 (2011)
2011
-
[19]
Boedecker, O
J. Boedecker, O. Obst, J. T. Lizier, N. M. Mayer, and M. Asada, Information processing in echo state networks at the edge of chaos, Theor. Biosci.131, 205 (2012)
2012
-
[20]
S. H. Gautam, T. T. Hoang, K. McClanahan, S. K. Grady, and W. L. Shew, Maximizing Sensory Dynamic Range by Tuning the Cortical State to Criticality, PLoS Comput. Biol.11, e1004576 (2015)
2015
-
[21]
Avramiea, A
A.-E. Avramiea, A. Masood, H. D. Mansvelder, and K. Linkenkaer-Hansen, Long-Range Amplitude Coupling Is Optimized for Brain Networks That Function at Crit- icality, J. Neurosci.42, 2221 (2022)
2022
-
[22]
Barzon, D
G. Barzon, D. M. Busiello, and G. Nicoletti, Excitation- inhibition balance controls information encoding in neu- ral populations, Phys. Rev. Lett.134, 068403 (2025)
2025
-
[23]
T. L. Ribeiro, M. Copelli, F. Caixeta, H. Belchior, D. R. Chialvo, M. A. L. Nicolelis, and S. Ribeiro, Spike Avalanches Exhibit Universal Dynamics across the Sleep- Wake Cycle, PLoS ONE5, e14129 (2010)
2010
-
[24]
Lombardi, H
F. Lombardi, H. J. Herrmann, C. Perrone-Capano, D. Plenz, and L. de Arcangelis, Balance between Excita- tion and Inhibition Controls the Temporal Organization of Neuronal Avalanches, Phys. Rev. Lett.108, 228703 (2012)
2012
-
[25]
Tagliazucchi, P
E. Tagliazucchi, P. Balenzuela, D. Fraiman, and D. R. Chialvo, Criticality in Large-Scale Brain fMRI Dynam- ics Unveiled by a Novel Point Process Analysis, Front. Physiol.3(2012)
2012
-
[26]
J. M. Palva, A. Zhigalov, J. Hirvonen, O. Korhonen, K. Linkenkaer-Hansen, and S. Palva, Neuronal long- range temporal correlations and avalanche dynamics are correlated with behavioral scaling laws, Proc. Natl. Acad. Sci. USA110, 3585 (2013)
2013
-
[27]
Shriki, J
O. Shriki, J. Alstott, F. Carver, T. Holroyd, R. N. Hen- son, M. L. Smith, R. Coppola, E. Bullmore, and D. Plenz, Neuronal Avalanches in the Resting MEG of the Human Brain, J. Neurosci.33, 7079 (2013)
2013
-
[28]
Cocchi, L
L. Cocchi, L. L. Gollo, A. Zalesky, and M. Breakspear, Criticality in the brain: A synthesis of neurobiology, models and cognition, Prog. Neurobiol.158, 132 (2017)
2017
-
[29]
A. J. Fontenele, N. A. P. De Vasconcelos, T. Feli- ciano, L. A. A. Aguiar, C. Soares-Cunha, B. Coimbra, L. Dalla Porta, S. Ribeiro, A. J. Rodrigues, N. Sousa, P. V. Carelli, and M. Copelli, Criticality between Corti- cal States, Phys. Rev. Lett.122, 208101 (2019)
2019
-
[30]
S. A. Jones, J. H. Barfield, V. K. Norman, and W. L. Shew, Scale-free behavioral dynamics directly linked with scale-free cortical dynamics, eLife12, e79950 (2023)
2023
-
[31]
Touboul and A
J. Touboul and A. Destexhe, Power-law statistics and universal scaling in the absence of criticality, Phys. Rev. E95, 012413 (2017)
2017
-
[32]
T. T. A. Carvalho, A. J. Fontenele, M. Girardi-Schappo, T. Feliciano, L. A. A. Aguiar, T. P. L. Silva, N. A. P. de Vasconcelos, P. V. Carelli, and M. Copelli, Subsam- pled directed-percolation models explain scaling relations experimentally observed in the brain, Front. Neural Circ. 14, 83 (2021)
2021
-
[33]
Bradde and W
S. Bradde and W. Bialek, PCA Meets RG, J. Stat. Phys. 167, 462 (2017)
2017
-
[34]
Meshulam, J
L. Meshulam, J. L. Gauthier, C. D. Brody, D. W. Tank, and W. Bialek, Coarse graining, fixed points, and scaling in a large population of neurons, Phys. Rev. Lett.123, 178103 (2019)
2019
-
[35]
D. M. Castro, T. Feliciano, N. A. P. De Vasconcelos, C. Soares-Cunha, B. Coimbra, A. J. Rodrigues, P. V. Carelli, and M. Copelli, In and Out of Criticality? State- Dependent Scaling in the Rat Visual Cortex, PRX Life 2, 023008 (2024)
2024
-
[36]
G. G. Cambrainha, D. M. Castro, N. A. P. de Vascon- celos, P. V. Carelli, and M. Copelli, Criticality at work: Scaling in the mouse cortex enhances performance, PRX Life3, 033026 (2025)
2025
-
[38]
Morales, S
G. Morales, S. Di Santo, and M. Mu˜ noz, Quasiuniver- sal scaling in mouse-brain neuronal activity stems from edge-of-instability critical dynamics, Proc. Natl. Acad. Sci.120, e2208998120 (2023)
2023
-
[39]
Harris, L
B. Harris, L. L. Gollo, and B. D. Fulcher, Tracking the distance to criticality in systems with unknown noise, Phys. Rev. X14, 031021 (2024)
2024
-
[40]
Q. Wang, O. Sporns, and A. Burkhalter, Network analy- sis of corticocortical connections reveals ventral and dor- sal processing streams in mouse visual cortex, J. Neu- rosci.32, 4386 (2012). 13
2012
-
[41]
Bienkowski, I
M. Bienkowski, I. Bowman, M. Song, L. Gou, T. Ard, K. Cotter, M. Zhu, N. L. Benavidez, S. Yamashita, J. Abu-Jaber, S. Azam, D. Lo, N. N. Foster, H. Hin- tiryan, and H.-W. Dong, Integration of gene expression and brain-wide connectivity reveals the multiscale orga- nization of mouse hippocampal networks, Nat. Neurosci. 21, 1628–1643 (2018)
2018
-
[42]
R. D. D’Souza, Q. Wang, W. Ji, A. M. Meier, H. Kennedy, K. Knoblauch, and A. Burkhalter, Hierar- chical and nonhierarchical features of the mouse visual cortical network, Nat. Commun.13, 1 (2022)
2022
-
[43]
Vezoli, L
J. Vezoli, L. Magrou, R. Goebel, X. Wang, K. Knoblauch, M. Vinck, and H. Kennedy, Cortical hierarchy, dual coun- terstream architecture and the importance of top-down generative networks, Neuroimage225, 117479 (2021)
2021
-
[44]
P. M. M¨ uller and C. Meisel, Spatial and temporal cor- relations in human cortex are inherently linked and pre- dicted by functional hierarchy, vigilance state as well as antiepileptic drug load, PLoS Comput. Biol.19, 1 (2023)
2023
-
[45]
Ponce-Alvarez, Network mechanisms underlying the regional diversity of variance and time scales of the brain’s spontaneous activity fluctuations, J
A. Ponce-Alvarez, Network mechanisms underlying the regional diversity of variance and time scales of the brain’s spontaneous activity fluctuations, J. Neurosci.45, 1 (2025)
2025
-
[46]
D. M. Castro, E. P. Raposo, M. Copelli, and F. A. N. Santos, Interdependent scaling exponents in the human brain, Phys. Rev. Lett.135, 198401 (2025)
2025
-
[47]
DANDI Archive
Allen Brain Institute for Brain Science, Allen institute - visual behavior - neuropixels [data set]. DANDI Archive. (2024)
2024
-
[48]
B. R. Munn, E. J. M¨ uller, I. Favre-Bulle, E. Scott, J. T. Lizier, M. Breakspear, and J. M. Shine, Multiscale orga- nization of neuronal activity unifies scale-dependent the- ories of brain function, Cell187, 7303 (2024)
2024
- [49]
-
[50]
Kadanoff, More is the same; phase transitions and mean field theories., J
L. Kadanoff, More is the same; phase transitions and mean field theories., J. Stat. Phys.137, 777–797 (2009)
2009
-
[51]
Nicoletti, S
G. Nicoletti, S. Suweis, and A. Maritan, Scaling and criticality in a phenomenological renormalization group, Phys. Rev. Res.2, 023144 (2020)
2020
-
[52]
Ponce-Alvarez, M
A. Ponce-Alvarez, M. L. Kringelbach, and G. Deco, Crit- ical scaling of whole-brain resting-state dynamics, Com- mun. Biol.6, 627 (2023)
2023
-
[53]
P. ˇZivadinovi´ c, F. Lombardi, D. Dupret, C. Boc- cara, S. Taveira, J. Ramirez-Villegas, G. Tkaˇ cik, and J. Csicsvari, The scaling behavior of hippocampal activ- ity in sleep/rest predicts spatial memory performance, bioRxiv 10.64898/2026.03.11.711043 (2026)
-
[54]
K. B. Hengen and W. L. Shew, Is criticality a unified set-point of brain function?, Neuron113, 1 (2024)
2024
-
[55]
S. J. Kiebel, J. Daunizeau, and K. J. Friston, A hierar- chy of time-scales and the brain, PLoS Comput. Biol.4, e1000209 (2008)
2008
-
[56]
J. D. Murray, A. Bernacchia, D. J. Freedman, R. Romo, J. D. Wallis, X. Cai, C. Padoa-Schioppa, T. Pasternak, H. Seo, D. Lee,et al., A hierarchy of intrinsic timescales across primate cortex, Nat. Neurosci.17, 1661 (2014)
2014
-
[57]
L. L. Gollo, A. Zalesky, R. M. Hutchison, M. Van Den Heuvel, and M. Breakspear, Dwelling quietly in the rich club: brain network determinants of slow cortical fluctuations, Philos. Trans. R. Soc. B370, 20140165 (2015)
2015
-
[58]
T. L. Ribeiro, M. Copelli, F. Caixeta, H. Belchior, D. R. Chialvo, M. A. L. Nicolelis, and S. Ribeiro, Spike avalanches exhibit universal dynamics across the sleep- wake cycle, PLoS One5, e14129 (2010)
2010
-
[59]
C. J. Honey, O. Sporns, L. Cammoun, X. Gigandet, J.- P. Thiran, R. Meuli, and P. Hagmann, Predicting human resting-state functional connectivity from structural con- nectivity, Proc. Natl. Acad. Sci.106, 2035 (2009)
2035
-
[60]
Fotiadis, L
P. Fotiadis, L. Parkes, K. A. Davis, T. D. Satterthwaite, R. T. Shinohara, and D. S. Bassett, Structure–function coupling in macroscale human brain networks, Nat. Rev. Neurosci25, 688 (2024)
2024
-
[61]
Rabuffo, P
G. Rabuffo, P. Bozzo, B. Nguyen, D. Depannemaecker, M. N. Pompili, L. L. Gollo, T. Fukai, P. Sorrentino, and L. Dalla Porta, The connectome modulates critical brain dynamics across local and global scales, BioRxiv , 2025 (2025)
2025
-
[62]
Lin, Divergence measures based on the shannon en- tropy, IEEE Trans
J. Lin, Divergence measures based on the shannon en- tropy, IEEE Trans. Inf. Theory37, 145 (1991)
1991
-
[63]
Endres and J
D. Endres and J. Schindelin, A new metric for probability distributions, IEEE Trans. Inf. Theory49, 1858 (2003)
2003
-
[64]
A. A. Hagberg, D. A. Schult, and P. J. Swart, Explor- ing network structure, dynamics, and function using net- workx, inProceedings of the 7th Python in Science Con- ference, edited by G. Varoquaux, T. Vaught, and J. Mill- man (Pasadena, CA USA, 2008) pp. 11 – 15
2008
-
[65]
M. E. J. Newman and M. Girvan, Finding and evaluat- ing community structure in networks, Phys. Rev. E69, 026113 (2004)
2004
-
[66]
Reichardt and S
J. Reichardt and S. Bornholdt, Statistical mechanics of community detection, Phys. Rev. E74, 016110 (2006)
2006
-
[67]
L. Meshulam, J. L. Gauthier, C. D. Brody, D. W. Tank, and W. Bialek, Coarse–graining and hints of scaling in a population of 1000+ neurons (2018), arXiv:1812.11904 [physics.bio-ph]. 14 Supplementary Material HIPPOCAMPUS CAN PRESENT DIFFERENT SHAPE OF ACTIVITY DISTRIBUTION As discussed in the main text, the distribution of coarse-grained activity in the hi...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.