Recognition: unknown
Hydrodynamic interactions mask the true heterogeneity of a microscopic collective
Pith reviewed 2026-05-08 06:59 UTC · model grok-4.3
The pith
Hydrodynamic interactions mask true heterogeneity in microscopic collectives, making even uniform groups appear varied from measured speeds.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
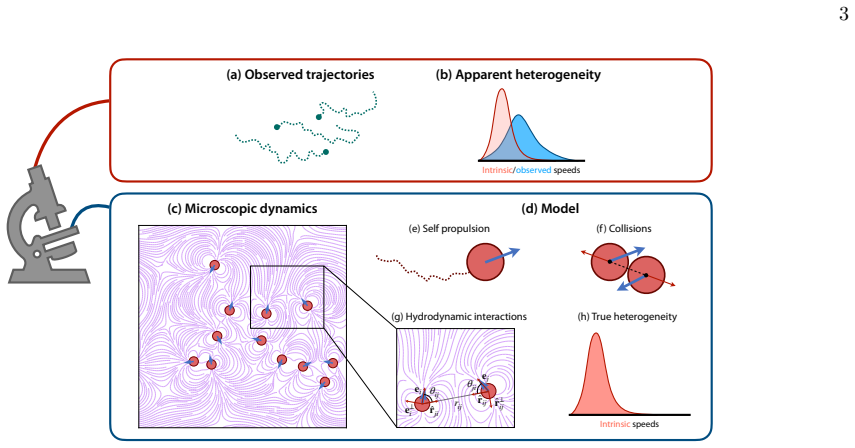
In a model system of agents undergoing purely physical interactions, the observed speeds result from competing effects where collisions reduce motion while hydrodynamic interactions increase it; consequently, when hydrodynamic interactions dominate, the heterogeneity inferred from those speeds no longer matches the true distribution of intrinsic motilities, so that a homogeneous collective registers as heterogeneous.
What carries the argument
The masking of intrinsic motility heterogeneity by dominant hydrodynamic flows in the presence of short-range collisions, which together determine measured speeds and alter collision isotropy.
Load-bearing premise
All interactions are purely physical with no biological sensing or active responses, and the chosen interaction strengths and particle densities represent real microscopic collectives.
What would settle it
Run the same simulation twice on a perfectly homogeneous collective: once with hydrodynamic interactions included and once without; if the version with hydrodynamics yields a non-zero inferred heterogeneity from speed statistics while the version without yields zero, the masking claim holds.
Figures





read the original abstract
Coordinated movement and self-organisation of active self-driven agents is common in nature and is seen across different scales, from herds of animals to collective motion in bacteria. Often, these systems are heterogeneous in composition, with different agents having different intrinsic motilities. Inferring these intrinsic characteristics and quantifying the level of heterogeneity in a collective system is crucial to understanding the observed emergent phenomena. However, when interaction effects dominate, i.e. the observed movement of an agent is strongly influenced by its interacting neighbours, inferring the intrinsic characteristics of agents becomes a challenge. We consider a collective system of agents that undergo purely physical interactions like collisions and long-range hydrodynamic interactions, which resembles a system of microswimmers immersed in a fluid medium. We incorporate heterogeneity into the system through variations in agent motility and examine how the perceived heterogeneity, inferred from measured speeds, depends on the strength of hydrodynamic interactions and the true intrinsic variability. The interplay between short-range collisions, long-range hydrodynamic interactions, and intrinsic heterogeneity makes the inference problem non-trivial. When hydrodynamic effects dominate, true heterogeneity is effectively masked, making even a homogeneous collective appear heterogeneous. The competing effects of collisions, which slow agents down, and hydrodynamic interactions, which enhance their motion, further complicate reliable inference. Hydrodynamic interactions also modify collision angles, rendering them more isotropic. Overall, the findings show highlight experimentally measured properties of microscopic collectives may not accurately reflect their true characteristics.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper uses agent-based simulations of microswimmers undergoing short-range collisions and long-range hydrodynamic interactions to show that, when hydrodynamic effects dominate, measured speed distributions make even intrinsically homogeneous collectives appear heterogeneous; collisions slow agents while hydrodynamics enhance motion and isotropize collision angles, complicating inference of true motility heterogeneity.
Significance. If the central masking result is robust, the work identifies a concrete mechanism by which interaction-dominated regimes in active matter can systematically bias experimental estimates of intrinsic heterogeneity, with direct implications for interpreting motility data in bacterial suspensions and other microscopic collectives.
major comments (3)
- [Simulation Methods] Simulation Methods: the manuscript provides no explicit values or ranges for the hydrodynamic interaction strength, particle density, or collision parameters, nor any sensitivity analysis; without these, it is impossible to determine whether the reported masking is generic or an artifact of the chosen regime.
- [Results] Results section on perceived vs. true heterogeneity: the claim that hydrodynamics 'mask' heterogeneity rests on qualitative comparison of speed histograms; no quantitative metric (e.g., Kullback-Leibler divergence or inferred variance ratio) is defined or reported, leaving the strength of the masking effect unquantified.
- [Discussion] Discussion: the extrapolation to real microswimmer systems assumes purely passive physical interactions dominate; no comparison is made to measured hydrodynamic strengths or densities in experimental bacterial or algal collectives, which is load-bearing for the stated experimental relevance.
minor comments (2)
- [Abstract] Abstract: 'show highlight' is a typographical error and should be corrected.
- [Figures] Figure captions: several panels lack explicit labels for the hydrodynamic strength parameter, reducing readability.
Simulated Author's Rebuttal
We thank the referee for the constructive and insightful comments, which have helped us improve the clarity and rigor of the manuscript. We address each major comment point by point below, indicating revisions made to the manuscript.
read point-by-point responses
-
Referee: [Simulation Methods] Simulation Methods: the manuscript provides no explicit values or ranges for the hydrodynamic interaction strength, particle density, or collision parameters, nor any sensitivity analysis; without these, it is impossible to determine whether the reported masking is generic or an artifact of the chosen regime.
Authors: We agree that explicit parameter values and sensitivity analysis were missing from the original submission. In the revised manuscript, we have added a new subsection in the Methods detailing the hydrodynamic interaction strength (via the Oseen tensor prefactor, with values from 0.1 to 10 in reduced units), particle densities (0.005 to 0.2), and collision parameters (hard-sphere radius and coefficient of restitution = 0.9). We have also included a sensitivity analysis (new Figure S1 and accompanying text) demonstrating that the masking effect is robust across regimes where hydrodynamic interactions dominate collisions, while noting the boundaries where collisions begin to dominate. revision: yes
-
Referee: [Results] Results section on perceived vs. true heterogeneity: the claim that hydrodynamics 'mask' heterogeneity rests on qualitative comparison of speed histograms; no quantitative metric (e.g., Kullback-Leibler divergence or inferred variance ratio) is defined or reported, leaving the strength of the masking effect unquantified.
Authors: We accept that the original presentation relied on qualitative visual comparison. To address this, we have defined and reported two quantitative metrics in the revised Results: (1) the ratio of perceived speed variance to true intrinsic motility variance, and (2) the Kullback-Leibler divergence between the measured speed distribution and the ground-truth motility distribution. These are plotted as functions of hydrodynamic strength and true heterogeneity level, providing a clear quantification of the masking effect and its dependence on interaction regime. revision: yes
-
Referee: [Discussion] Discussion: the extrapolation to real microswimmer systems assumes purely passive physical interactions dominate; no comparison is made to measured hydrodynamic strengths or densities in experimental bacterial or algal collectives, which is load-bearing for the stated experimental relevance.
Authors: We have revised the Discussion to include direct comparisons to experimental literature. We now reference measured hydrodynamic interaction strengths and densities from studies on E. coli and Chlamydomonas suspensions (e.g., densities ~10^7–10^9 cells/ml where long-range flows are significant), and map our simulation parameters onto these regimes. While our model remains idealized (purely passive interactions, no active sensing), the chosen parameter ranges align with dilute-to-moderate density experimental conditions where hydrodynamic masking would be expected. We note that full experimental validation lies beyond the scope of this theoretical work. revision: partial
Circularity Check
No significant circularity
full rationale
The paper reports results from direct numerical simulations of microswimmer collectives that incorporate collisions and hydrodynamic interactions as explicit physical forces. Heterogeneity is introduced by varying intrinsic motility parameters, and perceived heterogeneity is measured from simulated speed distributions; the masking effect is observed as an outcome of the dynamics rather than imposed by definition or by fitting a parameter to the target observable. No equations reduce the central claim to its inputs by construction, no self-citations supply load-bearing uniqueness theorems, and no ansatz is smuggled in via prior work. The model is self-contained as a parameter study whose conclusions follow from the simulated trajectories under stated physical rules.
Axiom & Free-Parameter Ledger
free parameters (1)
- hydrodynamic interaction strength
Reference graph
Works this paper leans on
-
[1]
T. Vicsek and A. Zafeiris, Collective motion, Physics Re- ports517, 71 (2012), arXiv: 1010.5017
-
[2]
Ramaswamy, The Mechanics and Statistics of Active Matter, Annual Review of Condensed Matter Physics1, 323 (2010)
S. Ramaswamy, The Mechanics and Statistics of Active Matter, Annual Review of Condensed Matter Physics1, 323 (2010)
2010
-
[3]
Alert and X
R. Alert and X. Trepat, Physical Models of Collective Cell Migration, Annual Review of Condensed Matter Physics11, 77 (2020)
2020
-
[4]
D. B. Kearns, A field guide to bacterial swarming motil- ity, Nature Reviews Microbiology8, 634 (2010)
2010
-
[5]
Be’er, B
A. Be’er, B. Ilkanaiv, R. Gross, D. B. Kearns, S. Heiden- reich, M. B¨ ar, and G. Ariel, A phase diagram for bacterial swarming, Communications Physics3, 66 (2020)
2020
-
[6]
Sumpter, The principles of collective animal be- haviour, Philosophical Transactions of the Royal Society B: Biological Sciences361, 5 (2006)
D. Sumpter, The principles of collective animal be- haviour, Philosophical Transactions of the Royal Society B: Biological Sciences361, 5 (2006)
2006
-
[7]
I. D. Couzin and J. Krause, Self-organization and col- lective behavior in vertebrates, inAdvances in the Study of Behavior, Advances in the Study of Behavior, Vol. 32 (Academic Press, 2003) pp. 1–75
2003
-
[8]
Helbing and P
D. Helbing and P. Moln´ ar, Social force model for pedes- trian dynamics, Physical Review E51, 4282 (1995)
1995
-
[9]
K. A. Bacik, B. S. Bacik, and T. Rogers, Lane nucleation in complex active flows, Science379, 923 (2023). 9
2023
-
[10]
Zadeh and B
P. Zadeh and B. A. Camley, Inferring Nonlinear Dynam- ics of Cell Migration, PRX Life2, 043020 (2024)
2024
- [11]
-
[12]
Lukeman, Y.-X
R. Lukeman, Y.-X. Li, and L. Edelstein-Keshet, Inferring individual rules from collective behavior, Proceedings of the National Academy of Sciences107, 12576 (2010)
2010
-
[13]
Y. Katz, K. Tunstrøm, C. C. Ioannou, C. Huepe, and I. D. Couzin, Inferring the structure and dynamics of interactions in schooling fish, Proceedings of the National Academy of Sciences108, 18720 (2011)
2011
-
[14]
J. E. Herbert-Read, A. Perna, R. P. Mann, T. M. Schaerf, D. J. T. Sumpter, and A. J. W. Ward, Inferring the rules of interaction of shoaling fish, Proceedings of the National Academy of Sciences108, 18726 (2011)
2011
-
[15]
D. S. Calovi, A. Litchinko, V. Lecheval, U. Lopez, A. P´ erez Escudero, H. Chat´ e, C. Sire, and G. Ther- aulaz, Disentangling and modeling interactions in fish with burst-and-coast swimming reveal distinct alignment and attraction behaviors, PLOS Computational Biology 14, e1005933 (2018)
2018
-
[16]
A. Nabeel and D. R. M, Disentangling intrinsic motion from neighbourhood effects in heterogeneous collective motion (2022), number: arXiv:2110.05864 arXiv:2110.05864 [nlin, physics:physics]
-
[17]
Goodale, H
E. Goodale, H. Sridhar, K. E. Sieving, P. Bangal, G. J. Colorado Z., D. R. Farine, E. W. Heymann, H. H. Jones, I. Krams, A. E. Mart´ ınez, F. Monta˜ no-Centellas, J. Mu˜ noz, U. Srinivasan, A. Theo, and K. Shanker, Mixed company: a framework for understanding the composi- tion and organization of mixed-species animal groups, Bi- ological Reviews95, 889 (2020)
2020
-
[18]
A. J. W. Ward, T. M. Schaerf, A. L. J. Burns, J. T. Lizier, E. Crosato, M. Prokopenko, and M. M. Webster, Cohe- sion, order and information flow in the collective motion of mixed-species shoals, Royal Society Open Science5, 181132 (2018)
2018
-
[19]
Reichhardt and C
C. Reichhardt and C. J. O. Reichhardt, Stripes, clus- ters, and nonequilibrium ordering for bidisperse colloids with repulsive interactions, Physical Review E75, 040402 (2007)
2007
-
[20]
Del Mar Delgado, M
M. Del Mar Delgado, M. Miranda, S. J. Alvarez, E. Gu- rarie, W. F. Fagan, V. Penteriani, A. Di Virgilio, and J. M. Morales, The importance of individual variation in the dynamics of animal collective movements, Philo- sophical Transactions of the Royal Society B: Biological Sciences373, 20170008 (2018)
2018
-
[21]
J. W. Jolles, A. J. King, and S. S. Killen, The Role of Individual Heterogeneity in Collective Animal Behaviour, Trends in Ecology & Evolution35, 278 (2020)
2020
-
[22]
Fern´ andez-L´ opez, D
P. Fern´ andez-L´ opez, D. Oro, R. Lloret-Cabot, M. Gen- ovart, J. Garriga, and F. Bartumeus, Foraging ants as liquid brains: Movement heterogeneity shapes collective efficiency, Proceedings of the National Academy of Sci- ences122, e2506930122 (2025)
2025
-
[23]
G. B. Blanchard, A. G. Fletcher, and L. J. Schumacher, The devil is in the mesoscale: Mechanical and be- havioural heterogeneity in collective cell movement, Sem- inars in Cell & Developmental Biology93, 46 (2019)
2019
-
[24]
Y. Chiu, J.-L. Hsu, C.-J. Lo, and Y. Jun, Length het- erogeneity of flexible bacteria enhances swarming, Soft Matter 10.1039/D5SM00798D (2026)
-
[25]
Tan and C
P. Tan and C. E. Miles, Intrinsic statistical separation of subpopulations in heterogeneous collective motion via dimensionality reduction, Physical Review E109, 014403 (2024)
2024
-
[26]
L. J. Schumacher, P. K. Maini, and R. E. Baker, Sem- blance of Heterogeneity in Collective Cell Migration, Cell Systems5, 119 (2017)
2017
-
[27]
Filella, F
A. Filella, F. Nadal, C. Sire, E. Kanso, and C. Eloy, Model of Collective Fish Behavior with Hydrody- namic Interactions, Physical Review Letters120, 198101 (2018)
2018
-
[28]
Tchieu, E
A. Tchieu, E. Kanso, and P. Newton, The finite-dipole dynamical system, Royal Society of London Proceedings Series A468, 3006 (2012)
2012
-
[29]
Newton, The dipole dynamical system, Conference Publications2005, 692 (2005)
P. Newton, The dipole dynamical system, Conference Publications2005, 692 (2005)
2005
-
[30]
Vishwakarma, B
M. Vishwakarma, B. Thurakkal, J. P. Spatz, and T. Das, Dynamic heterogeneity influences the leader–follower dy- namics during epithelial wound closure, Philosophical Transactions of the Royal Society B: Biological Sciences 375, 20190391 (2020)
2020
-
[31]
Tufoni, A
C. Tufoni, A. Battistella, S. Luppi, R. Boscolo, G. Ricci, M. Lazzarino, and L. Andolfi, Flagellar beating forces of human spermatozoa with different motility behaviors, Reproductive Biology and Endocrinology22, 28 (2024)
2024
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.