Recognition: unknown
On the role of crack electrolyte wetting in the degradation and performance of battery active particles
Pith reviewed 2026-05-09 16:17 UTC · model grok-4.3
The pith
Electrolyte wetting inside particle cracks redistributes reactions and raises delivered capacity beyond uniform flux predictions.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
Reaction redistribution inside cracked particles is governed mainly by local solid-state lithium concentration and stress fields rather than electrolyte potential gradients; the coupled model therefore predicts approximately 8x higher flux at crack tips, 25% higher delivered capacity at 1C, and 10% higher tensile stresses than the uniform-flux assumption.
What carries the argument
The controlled comparison between a fully coupled electro-chemo-mechanical model that resolves fields inside electrolyte-wetted cracks and a conventional single-particle model that imposes uniform interfacial flux.
If this is right
- Uniform flux models underpredict delivered capacity by 25% at 1C-rate, with the gap widening at higher rates.
- Tensile stresses throughout delithiation are underestimated by about 10%, altering predicted crack-driving forces.
- Neglecting crack-electrolyte coupling produces systematic underestimates of both utilisation limits and fatigue-relevant stress histories.
- Reaction rates become strongly heterogeneous, with local amplification at crack tips governed by concentration and stress rather than electrolyte potential.
Where Pith is reading between the lines
- Battery models used for lifetime prediction would need to track crack opening and electrolyte ingress to avoid optimistic capacity forecasts.
- Particle morphology or coating strategies that limit electrolyte penetration into cracks could reduce the capacity gain but also lower stress concentrations.
- The same coupling may appear in other intercalation materials where fracture exposes fresh surfaces to liquid electrolyte.
Load-bearing premise
Electrolyte potential gradients inside cracks remain secondary, and the chosen material properties, crack geometries, and rates are representative of real particles.
What would settle it
Direct measurement of delivered capacity and crack-tip stress in cathode particles containing known cracks, performed at 1C and higher rates, with and without electrolyte access to the crack interiors.
Figures







read the original abstract
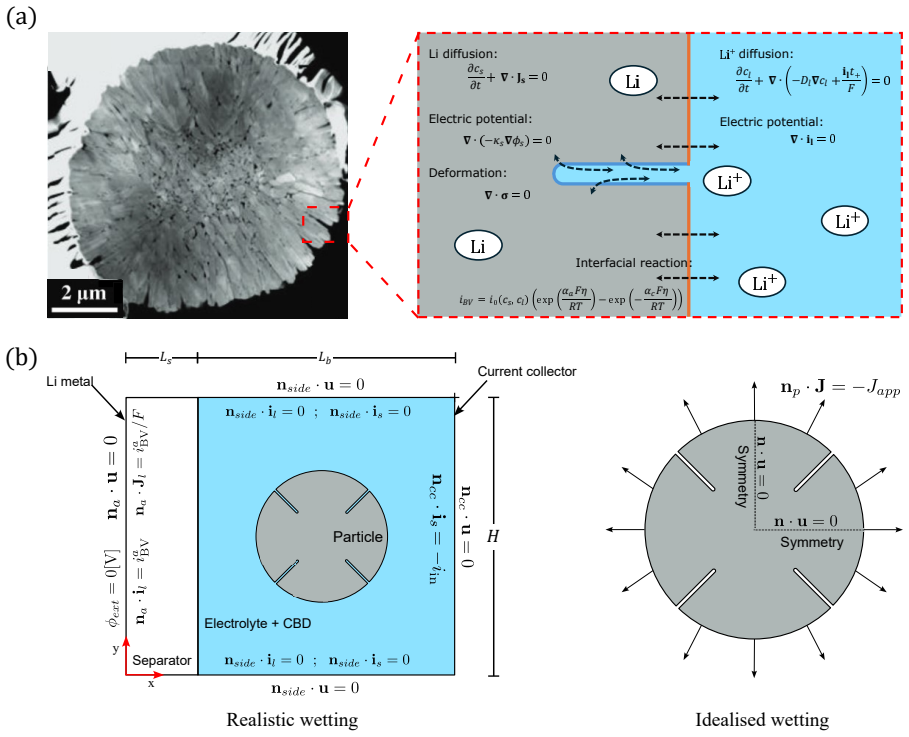
Cathode particle fracture is widely recognised as a major degradation mechanism in lithium-ion batteries, yet cracking also permits electrolyte wetting of newly exposed internal surfaces, modifying interfacial reaction pathways. The mechanistic role of electrolyte wetting in redistributing reactions within cracked particles remains unclear. Here, we isolate this effect through a controlled comparison between (i) a fully coupled electro-chemo-mechanical model resolving lithium concentration, electrostatic potential, and stress fields in both the active material and the electrolyte inside and outside cracks, and (ii) a single-particle chemo-mechanical model employing the conventional uniform flux assumption. The coupled model predicts strong spatial heterogeneity in interfacial reaction rates, with flux amplification approximately 8x relative to the imposed uniform flux at the crack tip. Reaction redistribution, and thus lithium flux, is governed predominantly by local solid-state lithium concentration and stress variations, while electrolyte potential gradients inside cracks remain secondary under the conditions considered. Uniform flux models can underpredict delivered capacity by 25% at 1C-rate; this discrepancy increases at higher rates. They also underestimate tensile stresses throughout the delithiation process by 10%, directly affecting crack driving conditions. These results demonstrate that neglecting crack-electrolyte coupling leads to systematic underestimation of both utilisation limits and fatigue-relevant stress histories.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper compares a fully coupled electro-chemo-mechanical model (resolving lithium concentration, electrostatic potential, and stress in active material and electrolyte inside/outside cracks) against a conventional single-particle chemo-mechanical model with uniform flux boundary conditions. It reports that the coupled model exhibits strong reaction-rate heterogeneity with ~8x flux amplification at crack tips, governed primarily by solid-state concentration and stress variations (electrolyte potential gradients inside cracks are secondary). Uniform-flux models are claimed to underpredict delivered capacity by 25% at 1C (increasing at higher rates) and tensile stresses by ~10% throughout delithiation, implying systematic underestimation of utilisation limits and fatigue-relevant stress histories when crack-electrolyte wetting is neglected.
Significance. If the reported discrepancies prove robust, the work isolates a previously under-appreciated mechanism by which crack wetting redistributes interfacial reactions and alters both capacity and stress evolution in fractured cathode particles. This provides a concrete, physics-based rationale for moving beyond uniform-flux assumptions in particle-scale degradation models and could improve predictions of rate capability and cycle life in systems where cracking is prevalent.
major comments (2)
- [Abstract] Abstract: The central quantitative claims (25% capacity underprediction at 1C and 10% tensile-stress underestimation) are presented as systematic outcomes of the coupled-versus-uniform comparison, yet they rest on a single fixed crack geometry, electrolyte conductivity, solid diffusivity, and particle size. No parameter-sensitivity study or mesh-convergence data are referenced to show that the discrepancy magnitude and the secondary role of electrolyte potential gradients persist across plausible ranges of crack aspect ratio or conductivity; without this, the 'systematic' label does not follow from the reported evidence.
- [Abstract] Abstract: The statement that 'electrolyte potential gradients inside cracks remain secondary under the conditions considered' is load-bearing for the interpretation that reaction redistribution is governed predominantly by solid-state concentration and stress. The manuscript provides no explicit test (e.g., a conductivity sweep or comparison of potential drop versus overpotential) demonstrating that this secondary status holds at the operating rates and crack widths examined.
minor comments (1)
- [Abstract] The abstract reports specific numerical discrepancies (8x flux, 25% capacity, 10% stress) without accompanying error bars, mesh-convergence metrics, or parameter ranges; adding these would strengthen the presentation even if the underlying comparison is sound.
Simulated Author's Rebuttal
We thank the referee for the constructive feedback on our manuscript. The comments highlight important aspects of generalizability and supporting evidence for our claims. We have revised the manuscript to include additional sensitivity analyses and explicit comparisons that address these points while preserving the core findings on reaction heterogeneity driven by solid-state effects.
read point-by-point responses
-
Referee: [Abstract] Abstract: The central quantitative claims (25% capacity underprediction at 1C and 10% tensile-stress underestimation) are presented as systematic outcomes of the coupled-versus-uniform comparison, yet they rest on a single fixed crack geometry, electrolyte conductivity, solid diffusivity, and particle size. No parameter-sensitivity study or mesh-convergence data are referenced to show that the discrepancy magnitude and the secondary role of electrolyte potential gradients persist across plausible ranges of crack aspect ratio or conductivity; without this, the 'systematic' label does not follow from the reported evidence.
Authors: We agree that the specific numerical values (25% capacity, 10% stress) are demonstrated for one representative crack geometry and parameter set, and that the abstract's use of 'systematic' requires qualification. The underlying mechanism—reaction-rate heterogeneity arising from local solid-state concentration and hydrostatic stress variations—is independent of the particular values chosen and follows directly from the coupled governing equations. To substantiate broader applicability, the revised manuscript now includes a dedicated sensitivity subsection. We varied crack aspect ratio (0.05–0.5), electrolyte conductivity (0.1–10 S m⁻¹), and particle radius (5–20 µm) while keeping other parameters fixed. Across this range the crack-tip flux amplification remains between 6× and 9×, capacity under-prediction stays within 20–30% at 1C (increasing with rate), and tensile-stress underestimation is 8–12%. Mesh-convergence data have been added to the Methods section, showing that key quantities (integrated capacity, maximum tensile stress, and tip flux) change by less than 3% upon doubling the element count. The abstract has been updated to state that the reported discrepancies are representative for the conditions examined and that the trends persist under moderate parameter variation. revision: yes
-
Referee: [Abstract] Abstract: The statement that 'electrolyte potential gradients inside cracks remain secondary under the conditions considered' is load-bearing for the interpretation that reaction redistribution is governed predominantly by solid-state concentration and stress. The manuscript provides no explicit test (e.g., a conductivity sweep or comparison of potential drop versus overpotential) demonstrating that this secondary status holds at the operating rates and crack widths examined.
Authors: We acknowledge that an explicit quantitative comparison was not presented in the original submission. In the revised manuscript we have added a direct decomposition of the local overpotential at the crack surfaces. We compute the electrolyte potential drop Δφ_e along the crack length and compare it to the solid-state contributions (chemical-potential term plus hydrostatic-stress term). For the base-case 1C discharge, the maximum |Δφ_e| inside the crack is 4–6 mV while the solid-state overpotential variation reaches 45–55 mV; thus the electrolyte contribution is <12% of the total driving-force heterogeneity. We further performed a conductivity sweep (0.1–10 S m⁻¹) at fixed crack width and rate. Even at the lowest conductivity the electrolyte term never exceeds 18% of the overpotential variation, and the reaction-rate amplification at the tip remains >6×. These results are now shown in a new figure and accompanying text, confirming that solid-state concentration and stress dominate under the examined conditions. revision: yes
Circularity Check
No circularity: simulation outputs from independent model comparison
full rationale
The paper's central results are numerical predictions obtained by solving two distinct physics-based models (fully coupled electro-chemo-mechanical vs. uniform-flux single-particle) under stated material parameters, crack geometries, and C-rates. The 25% capacity and 10% stress discrepancies are direct simulation outputs, not quantities that reduce to the inputs by definition, fitted-parameter renaming, or self-citation chains. No load-bearing self-citations, ansatz smuggling, or uniqueness theorems are invoked to force the reported heterogeneity or underpredictions. The derivation chain consists of standard continuum mechanics and electrochemistry equations solved numerically; the comparison is externally falsifiable by changing crack width, conductivity, or diffusivity.
Axiom & Free-Parameter Ledger
Reference graph
Works this paper leans on
-
[1]
Shahzad, I
K. Shahzad, I. I. Cheema, Low-carbon technologies in aut omotive industry and decarbonizing transport, Journal of Power Sources 591 (2024) 233888
2024
-
[2]
H.-J. Noh, S. Youn, C. S. Yoon, Y.-K. Sun, Comparison of th e structural and electrochemical properties of layered Li[NixCoyMnz]O2 (x = 1/3, 0.5, 0.6, 0. 7, 0.8 and 0.85) cathode material for lithium-ion batteries, Journal of Power Sources 233 (20 13) 121–130. 25
-
[3]
Han, A review on the key issues of the lithium ion batter y degradation among the whole life cycle, eTransportation (2019) 100005
X. Han, A review on the key issues of the lithium ion batter y degradation among the whole life cycle, eTransportation (2019) 100005
2019
-
[4]
Park, J.-Y
K.-J. Park, J.-Y. Hwang, H.-H. Ryu, F. Maglia, S.-J. Kim, P. Lamp, C. S. Yoon, Y.-K. Sun, Degradation Mechanism of Ni-Enriched NCA Cathode for Lithi um Batteries: Are Microcracks Really Critical?, ACS Energy Letters 4 (6) (2019) 1394–1400
2019
-
[5]
A. O. Kondrakov, A. Schmidt, J. Xu, H. Geßwein, R. Mönig, P . Hartmann, H. Sommer, T. Brezesinski, J. Janek, Anisotropic Lattice Strain and Me chanical Degradation of High- and Low-Nickel NCM Cathode Materials for Li-Ion Batteries, The Journal of Physical Chemistry C 121 (6) (2017) 3286–3294
2017
-
[6]
H. Liu, M. Wolfman, K. Karki, Y.-S. Yu, E. A. Stach, J. Caba na, K. W. Chapman, P. J. Chupas, Intergranular Cracking as a Major Cause of Long-Ter m Capacity Fading of Layered Cathodes, Nano Letters 17 (6) (2017) 3452–3457
2017
-
[7]
D. A. Santos, S. Rezaei, D. Zhang, Y. Luo, B. Lin, A. R. Bala krishna, B.-X. Xu, S. Banerjee, Chemistry–mechanics–geometry coupling in positive elect rode materials: a scale-bridging per- spective for mitigating degradation in lithium-ion batter ies through materials design, Chem- ical Science 14 (3) (2023) 458–484
2023
-
[8]
Ryu, K.-J
H.-H. Ryu, K.-J. Park, C. S. Yoon, Y.-K. Sun, Capacity Fad ing of Ni-Rich Li[Ni x Co y Mn 1– x – y ]O2 (0.6 ≤ x ≤ 0.95) Cathodes for High-Energy-Density Lithium-Ion Batte ries: Bulk or Surface Degradation?, Chemistry of Materials 30 (3) (2018) 1155–1163
2018
-
[9]
G. W. Nam, N.-Y. Park, K.-J. Park, J. Yang, J. Liu, C. S. Yoo n, Y.-K. Sun, Capacity Fading of Ni-Rich NCA Cathodes: Effect of Microcracking Extent, ACS Energy Letters 4 (12) (2019) 2995–3001
2019
-
[10]
Liao, S.-J
Q.-T. Liao, S.-J. Guo, M.-Y. Qi, S.-D. Zhang, P.-Z. Ma, J .-Y. Li, A.-M. Cao, L.-J. Wan, The genesis and control of microcracks in nickel-rich cathode m aterials for lithium-ion batteries, Sustainable Energy & Fuels 7 (19) (2023) 4805–4824
2023
-
[11]
Makimura, S
Y. Makimura, S. Zheng, Y. Ikuhara, Y. Ukyo, Microstruct ural Observation of LiNi 0.8 Co0.15 Al0.05 O2 after Charge and Discharge by Scanning Transmission Electr on Microscopy, Journal of The Electrochemical Society 159 (7) (2012) A1070–A1073. 26
2012
-
[12]
Grazioli, M
D. Grazioli, M. Magri, A. Salvadori, Computational mod eling of Li-ion batteries, Computa- tional Mechanics 58 (6) (2016) 889–909
2016
-
[13]
Y. Zhao, P. Stein, Y. Bai, M. Al-Siraj, Y. Yang, B.-X. Xu, A review on modeling of electro- chemo-mechanics in lithium-ion batteries, Journal of Powe r Sources 413 (2019) 259–283
2019
-
[14]
Pistorio, D
F. Pistorio, D. Clerici, F. Mocera, A. Somà, Review on th e numerical modeling of fracture in active materials for lithium ion batteries, Journal of Powe r Sources 566 (2023) 232875
2023
-
[15]
Klinsmann, D
M. Klinsmann, D. Rosato, M. Kamlah, R. M. McMeeking, Mod eling Crack Growth during Li Extraction in Storage Particles Using a Fracture Phase Fi eld Approach, Journal of The Electrochemical Society 163 (2) (2016) A102–A118
2016
-
[16]
Klinsmann, D
M. Klinsmann, D. Rosato, M. Kamlah, R. M. McMeeking, Mod eling crack growth during Li insertion in storage particles using a fracture phase field a pproach, Journal of the Mechanics and Physics of Solids 92 (2016) 313–344
2016
-
[17]
J. M. Allen, P. J. Weddle, A. Verma, A. Mallarapu, F. Usse glio-Viretta, D. P. Finegan, A. M. Colclasure, W. Mai, V. Schmidt, O. Furat, D. Diercks, T. Tani m, K. Smith, Quantifying the influence of charge rate and cathode-particle architect ures on degradation of Li-ion cells through 3D continuum-level damage models, Journal of Power Sources 512 (2021) 230415
2021
-
[18]
Y. Bai, D. A. Santos, S. Rezaei, P. Stein, S. Banerjee, B. -X. Xu, A chemo-mechanical damage model at large deformation: numerical and experimental stu dies on polycrystalline energy materials, International Journal of Solids and Structures 228 (2021) 111099
2021
-
[19]
Taghikhani, P
K. Taghikhani, P. J. Weddle, J. R. Berger, R. J. Kee, Mode ling Coupled Chemo-Mechanical Behavior of Randomly Oriented NMC811 Polycrystalline Li-I on Battery Cathodes, Journal of The Electrochemical Society 168 (8) (2021) 080511
2021
-
[20]
Singh, S
A. Singh, S. Pal, Chemo-mechanical modeling of inter- a nd intra-granular fracture in heteroge- neous cathode with polycrystalline particles for lithium- ion battery, Journal of the Mechanics and Physics of Solids 163 (2022) 104839
2022
-
[21]
W. Ai, B. Wu, E. Martínez-Pañeda, A coupled phase field fo rmulation for modelling fa- tigue cracking in lithium-ion battery electrode particles , Journal of Power Sources 544 (2022) 231805. 27
2022
-
[22]
Shishvan, N
S. Shishvan, N. Fleck, R. McMeeking, V. Deshpande, Crac king and associated volumetric expansion of NMC811 secondary particles, Journal of Power S ources 588 (2023) 233745
2023
-
[23]
Rezaei, J
S. Rezaei, J. N. Okoe-Amon, C. A. Varkey, A. Asheri, H. Ru an, B.-X. Xu, A cohesive phase- field fracture model for chemo-mechanical environments: St udies on degradation in battery materials, Theoretical and Applied Fracture Mechanics 124 (2023) 103758
2023
-
[24]
H. C. W. Parks, A. M. Boyce, A. Wade, T. M. M. Heenan, C. Tan , E. Martínez-Pañeda, P. R. Shearing, D. J. L. Brett, R. Jervis, Direct observation s of electrochemically induced intergranular cracking in polycrystalline NMC811 particl es, Journal of Materials Chemistry A 11 (39) (2023) 21322–21332
2023
-
[25]
Taghikhani, P
K. Taghikhani, P. J. Weddle, R. M. Hoffman, J. Berger, R. J . Kee, Electro-chemo-mechanical finite-element model of single-crystal and polycrystallin e NMC cathode particles embedded in an argyrodite solid electrolyte, Electrochimica Acta 46 0 (2023) 142585
2023
-
[26]
Asheri, S
A. Asheri, S. Rezaei, V. Glavas, B.-X. Xu, Microstructu re impact on chemo-mechanical frac- ture of polycrystalline lithium-ion battery cathode mater ials, Engineering Fracture Mechanics 309 (2024) 110370
2024
-
[27]
Bhowmick, X
A. Bhowmick, X. Gao, W. Lu, J. Chakraborty, J. Xu, Multis cale electro-chemo-mechanical model of high-capacity amorphous silicon anode material in sodium-ion batteries, Interna- tional Journal of Solids and Structures (2025) 113753
2025
-
[28]
S. Xia, L. Mu, Z. Xu, J. Wang, C. Wei, L. Liu, P. Pianetta, K . Zhao, X. Yu, F. Lin, Y. Liu, Chemomechanical interplay of layered cathode materials un dergoing fast charging in lithium batteries, Nano Energy 53 (2018) 753–762
2018
-
[29]
Ruess, S
R. Ruess, S. Schweidler, H. Hemmelmann, G. Conforto, A. Bielefeld, D. A. Weber, J. Sann, M. T. Elm, J. Janek, Influence of NCM Particle Cracking on Kine tics of Lithium-Ion Batteries with Liquid or Solid Electrolyte, Journal of The Electroche mical Society 167 (10) (2020) 100532
2020
-
[30]
Trevisanello, R
E. Trevisanello, R. Ruess, G. Conforto, F. H. Richter, J . Janek, Polycrystalline and Single Crystalline NCM Cathode Materials—Quantifying Particle C racking, Active Surface Area, and Lithium Diffusion, Advanced Energy Materials 11 (18) (20 21) 2003400. 28
-
[31]
W. Chen, L. J. Carrillo, A. Maji, X. Peng, J. Handy, S. Ban erjee, B. Xu, Deciphering the Interplay Between Wetting and Chemo-Mechanical Fracture i n Lithium-Ion Battery Cathode Materials, Advanced Functional Materials (2025) e17282
2025
-
[32]
J. Han, N. Sharma, K. Zhao, Computational modeling of co upled mechanical damage and electrochemistry in ternary oxide composite electrodes, J ournal of Power Sources 595 (2024) 234034
2024
-
[33]
Hageman, E
T. Hageman, E. Martínez-Pañeda, A phase field-based fra mework for electro-chemo- mechanical fracture: Crack-contained electrolytes, chem ical reactions and stabilisation, Com- puter Methods in Applied Mechanics and Engineering 415 (202 3) 116235
-
[34]
C. Cui, R. Ma, E. Martínez-Pañeda, Electro-chemo-mech anical phase field modeling of lo- calized corrosion: theory and COMSOL implementation, Engi neering with Computers 39 (6) (2023) 3877–3894
2023
-
[35]
Makuch, S
M. Makuch, S. Kovacevic, M. R. Wenman, E. Martínez-Pañe da, A microstructure-sensitive electro-chemo-mechanical phase-field model of pitting and stress corrosion cracking, Corrosion Science 232 (2024) 112031
2024
-
[36]
K. Park, H. Jung, L. Kuo, P. Kaghazchi, C. S. Yoon, Y. Sun, Improved Cycling Stability of Li[Ni0.90 Co0.05 Mn0.05 ]O2 Through Microstructure Modification by Boron Doping for Li- Ion Batteries, Advanced Energy Materials 8 (25) (2018) 1801202
2018
-
[37]
T. F. Fuller, M. Doyle, J. Newman, Simulation and Optimi zation of the Dual Lithium Ion Insertion Cell, Journal of The Electrochemical Society 141 (1) (1994) 1–10
1994
-
[38]
R. Xu, H. Sun, L. S. De Vasconcelos, K. Zhao, Mechanical a nd Structural Degradation of LiNi x Mn y Co z O 2 Cathode in Li-Ion Batteries: An Experimental Study, Journa l of The Electrochemical Society 164 (13) (2017) A3333–A3341
2017
-
[39]
R. Xu, Y. Yang, F. Yin, P. Liu, P. Cloetens, Y. Liu, F. Lin, K. Zhao, Heterogeneous damage in Li-ion batteries: Experimental analysis and theoretica l modeling, Journal of the Mechanics and Physics of Solids 129 (2019) 160–183. 29
2019
-
[40]
A. M. Boyce, E. Martínez-Pañeda, A. Wade, Y. S. Zhang, J. J. Bailey, T. M. Heenan, D. J. Brett, P. R. Shearing, Cracking predictions of lithium-ion battery electrodes by X-ray com- puted tomography and modelling, Journal of Power Sources 52 6 (2022) 231119
2022
-
[41]
P. Liu, R. Xu, Y. Liu, F. Lin, K. Zhao, Computational Mode ling of Heterogeneity of Stress, Charge, and Cyclic Damage in Composite Electrodes of Li-Ion Batteries, Journal of The Electrochemical Society 167 (4) (2020) 040527
2020
-
[42]
Ryu, N.-Y
H.-H. Ryu, N.-Y. Park, J. H. Seo, Y.-S. Yu, M. Sharma, R. M ücke, P. Kaghazchi, C. S. Yoon, Y.-K. Sun, A highly stabilized Ni-rich NCA cathode for high- energy lithium-ion batteries, Materials Today 36 (2020) 73–82
2020
-
[43]
Q. Lin, W. Guan, J. Zhou, J. Meng, W. Huang, T. Chen, Q. Gao , X. Wei, Y. Zeng, J. Li, Z. Zhang, Ni–Li anti-site defect induced intragranular cra cking in Ni-rich layer-structured cathode, Nano Energy 76 (2020) 105021
2020
-
[44]
H.-H. Ryu, B. Namkoong, J.-H. Kim, I. Belharouak, C. S. Y oon, Y.-K. Sun, Capacity Fading Mechanisms in Ni-Rich Single-Crystal NCM Cathodes, ACS Ene rgy Letters 6 (8) (2021) 2726–2734
2021
-
[45]
Y. Tu, B. Wu, W. Ai, E. Martínez-Pañeda, Influence of conc entration-dependent material properties on the fracture and debonding of electrode parti cles with core–shell structure, Journal of Power Sources 603 (2024) 234395
2024
-
[46]
T. E. Ashton, P. J. Baker, C. Sotelo-Vazquez, C. J. M. Foo ter, K. M. Kojima, T. Matsukawa, T. Kamiyama, J. A. Darr, Stoichiometrically driven disorde r and local diffusion in NMC cathodes, Journal of Materials Chemistry A 9 (16) (2021) 104 77–10486
2021
-
[47]
Ghosh, J
A. Ghosh, J. M. Foster, G. Offer, M. Marinescu, A Shrinkin g-Core Model for the Degrada- tion of High-Nickel Cathodes (NMC811) in Li-Ion Batteries: Passivation Layer Growth and Oxygen Evolution, Journal of The Electrochemical Society 1 68 (2) (2021) 020509
2021
-
[48]
L. O. Valøen, J. N. Reimers, Transport Properties of LiP F6-Based Li-Ion Battery Electrolytes, Journal of The Electrochemical Society (2005). 30
2005
-
[49]
Q. Gan, N. Qin, Z. Li, S. Gu, K. Liao, K. Zhang, L. Lu, Z. Xu, Z. Lu, Surface spinel recon- struction to suppress detrimental phase transition for sta ble LiNi0.8Co0.1Mn0.1O2 cathodes, Nano Research 16 (1) (2023) 513–520
2023
-
[50]
S. S. Pandurangi, D. S. Hall, C. P. Grey, V. S. Deshpande, N. A. Fleck, Chemo-Mechanical Analysis of Lithiation/Delithiation of Ni-rich Single Cry stals, Journal of The Electrochemical Society 170 (5) (2023) 050531
2023
-
[51]
Xu, Electrolytes and interphases in li-ion batterie s and beyond, Chemical reviews 114 (23) (2014) 11503–11618
K. Xu, Electrolytes and interphases in li-ion batterie s and beyond, Chemical reviews 114 (23) (2014) 11503–11618. 31
2014
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.