Recognition: unknown
Thermal-signature equivalence of breast tumors with heterogeneous perfusion in a modified Pennes bioheat model
Pith reviewed 2026-05-08 04:28 UTC · model grok-4.3
The pith
Different internal perfusion patterns in breast tumors produce nearly identical surface temperature signatures due to heat diffusion.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
In a modified Pennes bioheat model of multilayer breast tissue containing a finite tumor, uniform, rim-enhanced, necrotic-core, and anisotropic perfusion patterns generate clearly distinct internal temperature distributions. Heat diffusion and multilayer screening smooth these differences so that the surface temperature profiles become equivalent under a profile-distance metric for many combinations of patterns. Tumor depth decreases the distinguishability of surface signatures, whereas increasing tumor size enhances it. This establishes that a thermal anomaly at the surface does not guarantee unique identification of intratumoral perfusion heterogeneity.
What carries the argument
Profile-distance-based framework of thermal-signature equivalence, which measures when different intratumoral perfusion structures produce indistinguishable surface temperature profiles after diffusion.
If this is right
- Surface thermal anomalies detected by breast thermography do not uniquely identify specific intratumoral perfusion heterogeneities.
- Increasing tumor depth makes different perfusion patterns progressively harder to distinguish at the skin surface.
- Larger tumors improve the chance that internal perfusion differences remain visible in surface signatures.
- Heat diffusion through tissue layers acts as a strong low-pass filter that erases fine internal temperature variations.
- Static thermography has a fundamental limitation in resolving details of tumor blood-flow structure.
Where Pith is reading between the lines
- Diagnostic tools that rely solely on static surface temperature maps may need supplementary data from other modalities to resolve internal perfusion details.
- The equivalence framework could be tested by applying it to time-varying or dynamic thermography protocols.
- Extending the model to include metabolic heat generation changes or vascular remodeling would show whether the smoothing effect persists under more realistic conditions.
- The results suggest that inverse reconstruction of perfusion from surface data is likely ill-posed without strong additional constraints.
Load-bearing premise
The modified Pennes bioheat model with its chosen multilayer properties and perfusion values accurately represents real breast tissue heat transfer, and the profile-distance metric correctly identifies when surface signatures become indistinguishable for clinical purposes.
What would settle it
Direct measurement of surface temperature profiles on physical breast phantoms or patients with known tumor perfusion patterns, followed by comparison to the simulated profiles, would falsify the equivalence claim if statistically significant differences remain detectable beyond model predictions.
Figures





read the original abstract
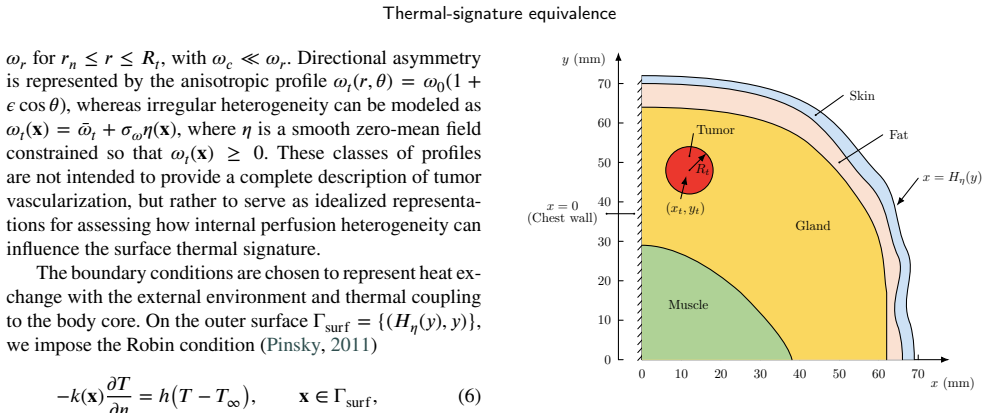
Breast thermography provides a noninvasive and contact-free method for observing tumor-associated thermal anomalies. However, the extent to which surface temperature patterns reflect the internal physiology of a tumor remains an open question. In this study, we investigate a modified Pennes bioheat model for multilayer breast tissue containing a finite-sized tumor with spatially heterogeneous intratumoral perfusion. Rather than focusing solely on the internal temperature field, we examine how different perfusion patterns are projected onto thermal signatures at the breast surface. We introduce a profile-distance-based framework of thermal-signature equivalence to quantify when different intratumoral perfusion structures remain distinguishable at the surface and when they become effectively indistinguishable. The results show that uniform, rim-enhanced, necrotic-core, and anisotropic perfusion patterns can produce clearly different internal temperature distributions, but these differences are strongly smoothed by heat diffusion and thermal screening before reaching the surface. Tumor depth reduces the distinguishability of surface signatures, whereas increasing tumor size enhances it. These findings highlight a fundamental limitation of static breast thermography: a thermal anomaly detected at the surface does not necessarily guarantee a unique identification of intratumoral perfusion heterogeneity.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper develops a modified Pennes bioheat model for multilayer breast tissue containing a finite tumor with heterogeneous intratumoral perfusion (uniform, rim-enhanced, necrotic-core, anisotropic patterns). Numerical solutions show that these patterns generate distinct internal temperature fields, but heat diffusion and thermal screening strongly smooth the differences at the breast surface. A profile-distance metric is introduced to quantify thermal-signature equivalence, with results indicating that distinguishability decreases with tumor depth and increases with tumor size. The central conclusion is that static breast thermography has a fundamental limitation: surface anomalies do not uniquely identify internal perfusion heterogeneity.
Significance. If the smoothing result is robust, the work supplies a clear physical explanation for why intratumoral perfusion details may be lost in surface thermography, which is useful for interpreting clinical data and setting realistic expectations for the modality. The profile-distance equivalence framework is a constructive addition that turns a qualitative observation into a quantifiable statement. The numerical demonstration of depth and size effects is a strength of the forward-modeling approach.
major comments (2)
- The specific multilayer thermal properties and perfusion magnitudes inserted into the modified Pennes equation are presented without accompanying sensitivity sweeps over physiological ranges. Because the attenuation of internal temperature contrasts at the surface is quantitatively controlled by conductivity, perfusion, and depth, modest changes in these parameters (well within literature values) can move surface differences above or below the equivalence threshold, directly affecting the claim that the patterns become indistinguishable.
- The numerical cutoff value in the profile-distance metric used to declare surface signatures equivalent is not justified by comparison to measured breast thermography data or clinical distinguishability criteria. The manuscript reports no direct validation of simulated surface maps against phantom or patient thermograms, so the reported equivalence may depend on the particular threshold chosen rather than a robust physical limit.
minor comments (2)
- The definition and normalization of the profile-distance metric should be stated explicitly in the methods section with an equation, rather than introduced only in the results when equivalence is concluded.
- Boundary conditions at the skin surface and the precise multilayer geometry (number of layers, thicknesses) are described only briefly; a table listing all parameter values with literature sources would improve reproducibility.
Simulated Author's Rebuttal
We thank the referee for the constructive comments on robustness and validation. We address each major point below with clarifications and revisions to strengthen the manuscript.
read point-by-point responses
-
Referee: The specific multilayer thermal properties and perfusion magnitudes inserted into the modified Pennes equation are presented without accompanying sensitivity sweeps over physiological ranges. Because the attenuation of internal temperature contrasts at the surface is quantitatively controlled by conductivity, perfusion, and depth, modest changes in these parameters (well within literature values) can move surface differences above or below the equivalence threshold, directly affecting the claim that the patterns become indistinguishable.
Authors: We selected representative parameter values from established literature on breast tissue (conductivity 0.4–0.5 W/m·K, perfusion 0.5–5 ml/min/100 g). To address the concern, the revised manuscript now includes sensitivity sweeps varying conductivity ±20%, perfusion rates by factors of 0.5–2.0, and tumor depth 1–4 cm. These confirm that the smoothing effect and the depth-dependent loss of distinguishability remain qualitatively robust, although absolute profile distances shift modestly. A new subsection and supplementary figure summarize the sweeps. revision: yes
-
Referee: The numerical cutoff value in the profile-distance metric used to declare surface signatures equivalent is not justified by comparison to measured breast thermography data or clinical distinguishability criteria. The manuscript reports no direct validation of simulated surface maps against phantom or patient thermograms, so the reported equivalence may depend on the particular threshold chosen rather than a robust physical limit.
Authors: The cutoff (normalized distance < 0.05) was chosen to lie below typical infrared camera resolution (~0.1 °C) and the model’s numerical precision, below which surface differences are unlikely to be detectable. We acknowledge that this study is a forward-modeling analysis without direct experimental validation. In revision we have clarified the threshold rationale in the methods, related it explicitly to measurement uncertainty, and added a limitations paragraph noting that phantom or clinical validation remains future work. The central physical result—that diffusion smooths internal perfusion contrasts—does not rely on the precise numerical value of the cutoff. revision: partial
Circularity Check
No significant circularity: forward bioheat simulations with post-processed equivalence metric
full rationale
The paper solves the modified Pennes bioheat PDE forward for prescribed intratumoral perfusion patterns (uniform, rim-enhanced, necrotic-core, anisotropic) as explicit inputs, computes the resulting 3D temperature fields, extracts surface profiles, and applies a newly defined profile-distance metric to classify signatures as equivalent or distinguishable. The smoothing conclusion follows directly from the diffusion term in the governing equation and the low-pass filtering effect of tissue layers; it is not obtained by fitting parameters to a target surface signature or by any self-referential closure. No load-bearing self-citations, ansatzes smuggled via prior work, or uniqueness theorems imported from the same authors are invoked to force the central result. The equivalence framework is a post-hoc definition on model outputs rather than a tautological re-expression of the inputs. This is standard numerical forward modeling without reduction of the claimed thermal-signature equivalence to its own construction.
Axiom & Free-Parameter Ledger
free parameters (2)
- intratumoral perfusion rates for each pattern
- tumor depth and size parameters
axioms (2)
- domain assumption Pennes bioheat equation governs steady-state heat transfer in perfused tissue
- domain assumption Multilayer breast geometry with finite tumor accurately represents anatomy
invented entities (1)
-
profile-distance-based thermal-signature equivalence framework
no independent evidence
Reference graph
Works this paper leans on
-
[1]
Journal of applied physiology , volume=
Analysis of tissue and arterial blood temperatures in the resting human forearm , author=. Journal of applied physiology , volume=
-
[2]
Journal of applied physiology , volume=
Pennes’ 1948 paper revisited , author=. Journal of applied physiology , volume=. 1998 , publisher=
1948
-
[3]
Thermal science and engineering progress , volume=
Early detection of the breast cancer using infrared technology--A comprehensive review , author=. Thermal science and engineering progress , volume=. 2022 , publisher=
2022
-
[4]
Applied Sciences , volume=
A review of techniques and bio-heat transfer models supporting infrared thermal imaging for diagnosis of malignancy , author=. Applied Sciences , volume=. 2024 , publisher=
2024
-
[5]
Scientific reports , volume=
Determining the thermal characteristics of breast cancer based on high-resolution infrared imaging, 3D breast scans, and magnetic resonance imaging , author=. Scientific reports , volume=. 2020 , publisher=
2020
-
[6]
International Journal of Thermal Sciences , volume=
Thermal modeling for breast tumor detection using thermography , author=. International Journal of Thermal Sciences , volume=. 2021 , publisher=
2021
-
[7]
Scientific reports , volume=
Breast cancer detection using enhanced IRI-numerical engine and inverse heat transfer modeling: model description and clinical validation , author=. Scientific reports , volume=. 2024 , publisher=
2024
-
[8]
Proceedings of the Institution of Mechanical Engineers, Part C: Journal of Mechanical Engineering Science , volume=
Inverse thermal modeling and experimental validation for breast tumor detection by using highly personalized surface thermal patterns and geometry of the breast , author=. Proceedings of the Institution of Mechanical Engineers, Part C: Journal of Mechanical Engineering Science , volume=. 2021 , publisher=
2021
-
[9]
International Journal of Heat and Mass Transfer , volume=
Modified Pennes bioheat equation with heterogeneous blood perfusion: A newer perspective , author=. International Journal of Heat and Mass Transfer , volume=. 2024 , publisher=
2024
-
[10]
Sensors , volume=
Dynamic Thermography-Based Early Breast Cancer Detection Using Multivariate Time Series , author=. Sensors , volume=. 2025 , publisher=
2025
-
[11]
Thermal Science and Engineering Progress , volume=
Influence of thermal contrast and limitations of a deep-learning based estimation of early-stage tumour parameters in different breast shapes using simulated passive and dynamic thermography , author=. Thermal Science and Engineering Progress , volume=. 2025 , publisher=
2025
-
[12]
Advances in heat transfer , volume=
Mathematical models of bioheat transfer , author=. Advances in heat transfer , volume=. 1992 , publisher=
1992
-
[13]
International Journal of Thermal Sciences , volume=
A review of thermography as promising non-invasive detection modality for breast tumor , author=. International Journal of Thermal Sciences , volume=. 2009 , publisher=
2009
-
[14]
Computational and mathematical methods in medicine , volume=
Evaluation of the diagnostic power of thermography in breast cancer using bayesian network classifiers , author=. Computational and mathematical methods in medicine , volume=. 2013 , publisher=
2013
-
[15]
Journal of Advanced Research , volume=
Parameter estimation of breast tumour using dynamic neural network from thermal pattern , author=. Journal of Advanced Research , volume=. 2016 , publisher=
2016
-
[16]
Systematic reviews , volume=
Breast thermography: a systematic review and meta-analysis , author=. Systematic reviews , volume=. 2024 , publisher=
2024
-
[17]
Infrared Physics & Technology , volume=
An inverse heat transfer approach for patient-specific breast cancer detection and tumor localization using surface thermal images in the prone position , author=. Infrared Physics & Technology , volume=. 2020 , publisher=
2020
-
[18]
Applied Sciences , volume=
Patient/breast-specific detection of breast tumor based on patients’ thermograms, 3d breast scans, and reverse thermal modelling , author=. Applied Sciences , volume=. 2021 , publisher=
2021
-
[19]
ASME Journal of Heat and Mass Transfer , volume=
Thermal modeling of patient-specific breast cancer with physics-based artificial intelligence , author=. ASME Journal of Heat and Mass Transfer , volume=. 2023 , publisher=
2023
-
[20]
Journal of Engineering and Science in Medical Diagnostics and Therapy , volume=
Detectability of breast cancer through inverse heat transfer modeling using patient-specific surface temperatures , author=. Journal of Engineering and Science in Medical Diagnostics and Therapy , volume=. 2025 , publisher=
2025
-
[21]
Cancers , volume=
Breast cancer screening using inverse modeling of surface temperatures and steady-state thermal imaging , author=. Cancers , volume=. 2024 , publisher=
2024
-
[22]
International Journal of Thermal Sciences , volume=
Simulation and clinical validation of the breast temperature field based on a multi-point heat source model , author=. International Journal of Thermal Sciences , volume=. 2025 , publisher=
2025
-
[23]
Scientific Reports , volume=
Early breast cancer detection via infrared thermography using a CNN enhanced with particle swarm optimization , author=. Scientific Reports , volume=. 2025 , publisher=
2025
-
[24]
International Journal of Surgery , volume=
Evaluation of digital infra--red thermal imaging as an adjunctive screening method for breast carcinoma: A pilot study , author=. International Journal of Surgery , volume=. 2014 , publisher=
2014
-
[25]
Bioengineering , volume=
Breast cancer detection using infrared thermography: A survey of texture analysis and machine learning approaches , author=. Bioengineering , volume=. 2025 , publisher=
2025
-
[26]
Thomas , title =
Shrivastava, Devashish and Vaughan, J. Thomas , title =. Journal of Biomechanical Engineering , volume =. 2009 , month =
2009
-
[27]
and Jiji, L
Weinbaum, S. and Jiji, L. M. , title =. Journal of Biomechanical Engineering , volume =. 1985 , month =
1985
-
[28]
2013 , publisher=
Physical properties of tissues: a comprehensive reference book , author=. 2013 , publisher=
2013
-
[29]
Computer methods in biomechanics and biomedical engineering , volume=
Surface temperature distribution of a breast with and without tumour , author=. Computer methods in biomechanics and biomedical engineering , volume=. 1999 , publisher=
1999
-
[30]
Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine , volume=
Effect of blood flow, tumour and cold stress in a female breast: A novel time-accurate computer simulation , author=. Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine , volume=. 2001 , publisher=
2001
-
[31]
International Conference on Medical Image Computing and Computer-Assisted Intervention , pages=
Dynamic thermal modeling of the normal and tumorous breast under elastic deformation , author=. International Conference on Medical Image Computing and Computer-Assisted Intervention , pages=. 2008 , organization=
2008
-
[32]
Physics in Medicine & Biology , volume=
Modeling static and dynamic thermography of the human breast under elastic deformation , author=. Physics in Medicine & Biology , volume=
-
[33]
ASME International Mechanical Engineering Congress and Exposition , volume=
Thermal analysis of cancerous breast model , author=. ASME International Mechanical Engineering Congress and Exposition , volume=. 2012 , organization=
2012
-
[34]
Journal of thermal biology , volume=
Simultaneous estimation of size, radial and angular locations of a malignant tumor in a 3-D human breast--A numerical study , author=. Journal of thermal biology , volume=. 2015 , publisher=
2015
-
[35]
International Journal of Hyperthermia , volume=
Inverse method for quantitative characterisation of breast tumours from surface temperature data , author=. International Journal of Hyperthermia , volume=. 2017 , publisher=
2017
-
[36]
Science , volume=
Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy , author=. Science , volume=. 2005 , publisher=
2005
-
[37]
Physiological reviews , volume=
Normalization of the vasculature for treatment of cancer and other diseases , author=. Physiological reviews , volume=. 2011 , publisher=
2011
-
[38]
Technology in cancer research & treatment , volume=
Hypoxia and the tumor microenvironment , author=. Technology in cancer research & treatment , volume=. 2021 , publisher=
2021
-
[39]
Scientific reports , volume=
Delineation of tumor habitats based on dynamic contrast enhanced MRI , author=. Scientific reports , volume=. 2017 , publisher=
2017
-
[40]
Scientific African , volume=
Deep learning-based approach in surface thermography for inverse estimation of breast tumor size , author=. Scientific African , volume=. 2024 , publisher=
2024
-
[41]
Applied Sciences , volume=
Influence of tissue thermophysical characteristics and situ-cooling on the detection of breast cancer , author=. Applied Sciences , volume=. 2023 , publisher=
2023
-
[42]
2011 , publisher=
Partial differential equations and boundary-value problems with applications , author=. 2011 , publisher=
2011
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.