Recognition: 1 theorem link
· Lean TheoremModeling the Impact of Exposed Cases in a Hantavirus Outbreak on a Cruise Ship
Pith reviewed 2026-05-11 01:48 UTC · model grok-4.3
The pith
Hantavirus model on a cruise ship estimates R0 at 2.76 and shows that hidden exposed cases create an undetected transmission reservoir.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
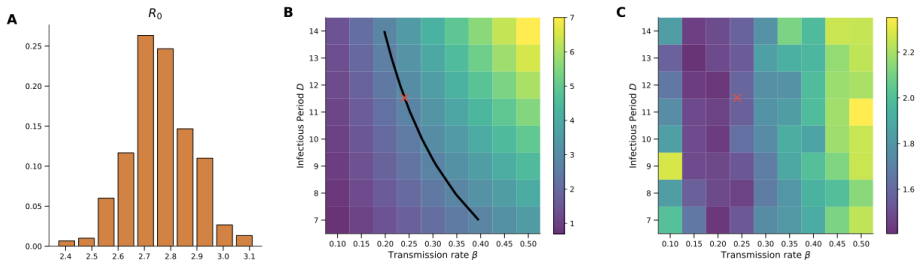
A discrete-time stochastic Susceptible-Exposed-Infectious-Recovered-Dead model, calibrated with an Ensemble Adjustment Kalman Filter to WHO and ECDC situation reports, produces an estimated basic reproduction number of 2.76 (95 % CI 2.52–2.99) and indicates that several exposed individuals remain unidentified during the early phase, forming a hidden reservoir that symptom-based surveillance alone cannot detect.
What carries the argument
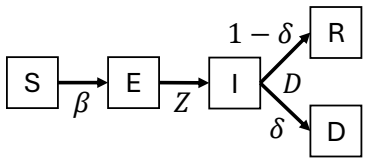
Discrete-time stochastic SEIRD model whose latent states and parameters are jointly inferred by the Ensemble Adjustment Kalman Filter from reported case counts, allowing estimation of both the reproduction number and the size of the undetected exposed compartment.
If this is right
- Sustained onboard transmission is likely without strict quarantine measures.
- Symptom-based surveillance alone leaves a hidden reservoir of exposed individuals.
- Rapid surveillance, widespread testing, and active monitoring of exposed people become necessary in confined travel settings.
- The same modeling framework can be applied to assess intervention needs in other dense, spatially constrained populations.
Where Pith is reading between the lines
- The same hidden-reservoir dynamic could appear in other confined environments such as dormitories, barracks, or long-haul flights.
- Real-time versions of the filter might enable earlier detection of hidden cases if partial data streams are available.
- Combining the model with genomic sequencing of cases could help distinguish imported versus onboard transmission chains.
Load-bearing premise
The reported case numbers from official sources accurately reflect the true incidence and that the filter can recover unbiased epidemiological parameters despite any unmodeled biases or data gaps.
What would settle it
After the outbreak, compare the model's predicted total number of exposed and infected individuals (including the hidden compartment) against the actual count obtained from comprehensive contact tracing or serological surveys.
Figures


read the original abstract
The emergence of a hantavirus variant aboard a commercial cruise ship presents a significant public health concern. This study develops a discrete-time stochastic Susceptible-Exposed-Infectious-Recovered-Dead model to estimate transmission dynamics, hidden exposed infections, and outbreak risk among passengers and crew. Epidemiological parameters and latent disease states were inferred using an Ensemble Adjustment Kalman Filter calibrated to reported case data from WHO and ECDC situation reports. The estimated basic reproduction number was 2.76, with a 95\% confidence interval of 2.52-2.99, indicating substantial potential for sustained onboard transmission before strict quarantine measures. Simulations further suggest that several exposed individuals may remain unidentified during the early outbreak phase, creating a hidden reservoir that symptom-based surveillance alone may fail to detect. These findings highlight the importance of rapid surveillance, widespread testing, targeted quarantine, and active monitoring of exposed individuals in confined travel settings. The proposed modeling framework can support timely outbreak assessment and intervention planning for infectious-disease events in similarly dense and spatially constrained populations.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper develops a discrete-time stochastic SEIRD model for a hantavirus outbreak on a cruise ship. Epidemiological parameters and latent states are inferred via Ensemble Adjustment Kalman Filter assimilation of reported case counts from WHO and ECDC situation reports. The central results are an estimated basic reproduction number of 2.76 (95% CI 2.52-2.99) and the inference of a sizable undetected exposed reservoir during the early phase.
Significance. If the modeling assumptions hold, the work demonstrates how data assimilation can quantify transmission potential and surveillance gaps in confined populations, supporting the value of rapid testing and targeted quarantine. The stochastic formulation and EAKF approach provide a reproducible framework for similar settings, though the quantitative claims rest on untested observation assumptions.
major comments (1)
- Methods (model calibration and observation model): The R0 estimate and the claim of a hidden exposed reservoir are obtained by treating the time series of reported cases as an unbiased observation of the true I(t) + E(t) process. No sensitivity analysis is reported that perturbs the observation model (e.g., time-varying reporting probability or explicit under-ascertainment error). Because under-counting is expected in an early-phase cruise-ship outbreak with symptom-based reporting, this assumption directly scales the inferred transmission rate and inflates posterior mass on the latent exposed compartment; it is therefore load-bearing for both headline results.
minor comments (2)
- The abstract and results section would benefit from an explicit statement of the observation equation used in the EAKF and the precise definition of the reported-case likelihood.
- Figure captions should clarify whether the simulated trajectories include parameter uncertainty or only state uncertainty.
Simulated Author's Rebuttal
We thank the referee for the constructive feedback and for recognizing the potential utility of the EAKF framework in confined settings. We address the single major comment below.
read point-by-point responses
-
Referee: Methods (model calibration and observation model): The R0 estimate and the claim of a hidden exposed reservoir are obtained by treating the time series of reported cases as an unbiased observation of the true I(t) + E(t) process. No sensitivity analysis is reported that perturbs the observation model (e.g., time-varying reporting probability or explicit under-ascertainment error). Because under-counting is expected in an early-phase cruise-ship outbreak with symptom-based reporting, this assumption directly scales the inferred transmission rate and inflates posterior mass on the latent exposed compartment; it is therefore load-bearing for both headline results.
Authors: We agree that the observation model is a central assumption and that the absence of sensitivity analysis to under-ascertainment is a limitation. The current formulation follows the standard EAKF observation operator used in prior epidemic assimilation studies, in which reported counts are treated as direct (noisy) observations of I(t) + E(t) given the symptom-based reporting in the WHO/ECDC situation reports. Nevertheless, we accept the referee's point that this choice affects the inferred transmission rate and exposed compartment size. In the revised manuscript we will add an explicit sensitivity analysis: we will introduce a constant reporting probability p (tested at values 0.5, 0.7, 0.9, and 1.0) into the observation model, re-run the EAKF ensemble, and report the resulting ranges for R0 and the early-phase exposed reservoir. We will also note that time-varying reporting is difficult to identify with the available daily aggregate counts but can be explored in future work with higher-resolution data. revision: yes
Circularity Check
No significant circularity; standard data-driven estimation
full rationale
The paper constructs a discrete-time stochastic SEIRD model and applies Ensemble Adjustment Kalman Filter assimilation to reported case counts to obtain parameter estimates (including R0) and latent state trajectories. This is a conventional inverse problem workflow: the model structure and observation model are specified first, then calibrated to external data. No equation reduces to its own input by construction, no parameter is fitted on a subset and then relabeled as an independent prediction of a closely related quantity, and no load-bearing premise rests on a self-citation chain. The reported R0 value and inferred exposed compartment are direct outputs of the assimilation step rather than tautological restatements of the inputs. The analysis is therefore self-contained given its stated assumptions and data source.
Axiom & Free-Parameter Ledger
free parameters (3)
- basic reproduction number =
2.76
- transmission and recovery rates
- latent state parameters
axioms (2)
- domain assumption The SEIRD model structure accurately represents hantavirus transmission dynamics
- domain assumption Reported case data from WHO and ECDC is sufficient and unbiased for calibration
Lean theorems connected to this paper
-
IndisputableMonolith/Foundation/RealityFromDistinction.leanreality_from_one_distinction uncleardiscrete-time stochastic SEIRD model... Ensemble Adjustment Kalman Filter calibrated to reported case data from WHO and ECDC
Reference graph
Works this paper leans on
-
[1]
https://www.who.int/news/item/07-05-2026-who-s-response-to-hantavirus-cases-linked-to-a- cruise-ship
“https://www.who.int/news/item/07-05-2026-who-s-response-to-hantavirus-cases-linked-to-a- cruise-ship.”
work page 2026
-
[2]
https://www.ecdc.europa.eu/sites/default/files/documents/tab-hantavirus-06052026.pdf
“https://www.ecdc.europa.eu/sites/default/files/documents/tab-hantavirus-06052026.pdf.”
-
[3]
Infections in confined spaces: cruise ships, military barracks, and college dormitories,
V. Kak, “Infections in confined spaces: cruise ships, military barracks, and college dormitories,” Infectious disease clinics of North America, vol. 21, no. 3, pp. 773–784, 2007
work page 2007
-
[4]
A. Pavli, H. C. Maltezou, A. Papadakis, P. Katerelos, G. Saroglou, A. Tsakris, and S. Tsiodras, “Respiratory infections and gastrointestinal illness on a cruise ship: a three-year prospective study,”Travel medicine and infectious disease, vol. 14, no. 4, pp. 389–397, 2016
work page 2016
-
[5]
The scaling of contact rates with population density for the infectious disease models,
H. Hu, K. Nigmatulina, and P. Eckhoff, “The scaling of contact rates with population density for the infectious disease models,”Mathematical biosciences, vol. 244, no. 2, pp. 125–134, 2013
work page 2013
-
[6]
Contact infection of infectious disease onboard a cruise ship,
N. Zhang, R. Miao, H. Huang, and E. Y. Chan, “Contact infection of infectious disease onboard a cruise ship,”Scientific reports, vol. 6, no. 1, p. 38790, 2016
work page 2016
-
[7]
J. Rocklöv, H. Sjödin, and A. Wilder-Smith, “Covid-19 outbreak on the diamond princess cruise ship: estimating the epidemic potential and effectiveness of public health countermeasures,” Journal of travel medicine, vol. 27, no. 3, p. taaa030, 2020
work page 2020
-
[8]
B. Batista, D. Dickenson, K. Gurski, M. Kebe, and N. Rankin, “Minimizing disease spread on a quarantined cruise ship: A model of covid-19 with asymptomatic infections,”Mathematical biosciences, vol. 329, p. 108442, 2020
work page 2020
-
[9]
J. C. Emery, T. W. Russell, Y. Liu, J. Hellewell, C. A. Pearson, G. M. Knight, R. M. Eggo, A. J. Kucharski, S. Funk,et al., “The contribution of asymptomatic sars-cov-2 infections to transmission on the diamond princess cruise ship,”Elife, vol. 9, p. e58699, 2020
work page 2020
-
[10]
Human hantavirus infections: epidemiology, clinical features, patho- genesis and immunology,
T. Manigold and P. Vial, “Human hantavirus infections: epidemiology, clinical features, patho- genesis and immunology,” 2014
work page 2014
-
[11]
The incubation period of hantavirus pulmonary syndrome.,
J. C. Young, G. R. Hansen, T. K. Graves, M. P. Deasy, J. G. Humphreys, C. L. Fritz, K. L. Gorham, A. S. Khan, T. G. Ksiazek, K. B. Metzger,et al., “The incubation period of hantavirus pulmonary syndrome.,”The American journal of tropical medicine and hygiene, vol. 62, no. 6, pp. 714–717, 2000
work page 2000
-
[12]
K. Mizumoto, K. Kagaya, A. Zarebski, and G. Chowell, “Estimating the asymptomatic pro- portion of coronavirus disease 2019 (covid-19) cases on board the diamond princess cruise ship, yokohama, japan, 2020,”Eurosurveillance, vol. 25, no. 10, p. 2000180, 2020
work page 2019
-
[13]
S. Zhang, M. Diao, W. Yu, L. Pei, Z. Lin, and D. Chen, “Estimation of the reproductive number of novel coronavirus (covid-19) and the probable outbreak size on the diamond princess cruise ship: A data-driven analysis,”International journal of infectious diseases, vol. 93, pp. 201–204, 2020
work page 2020
-
[14]
Incubation period of hantavirus cardiopulmonary syndrome,
P. A. Vial, F. Valdivieso, G. Mertz, C. Castillo, E. Belmar, I. Delgado, M. Tapia, and M. Ferrés, “Incubation period of hantavirus cardiopulmonary syndrome,”Emerging Infectious Diseases, vol. 12, no. 8, p. 1271, 2006. 7
work page 2006
-
[15]
O.Diekmann, J.A.P.Heesterbeek, andJ.A.J.Metz, “Onthedefinitionandthecomputationof the basic reproduction ratio r 0 in models for infectious diseases in heterogeneous populations,” Journal of mathematical biology, vol. 28, no. 4, pp. 365–382, 1990
work page 1990
-
[16]
P. Van den Driessche and J. Watmough, “Reproduction numbers and sub-threshold en- demic equilibria for compartmental models of disease transmission,”Mathematical biosciences, vol. 180, no. 1-2, pp. 29–48, 2002
work page 2002
-
[17]
The mathematics of infectious diseases,
H. W. Hethcote, “The mathematics of infectious diseases,”SIAM review, vol. 42, no. 4, pp. 599– 653, 2000
work page 2000
-
[18]
Differentiable agent-based epidemiology,
A. Chopra, A. Rodríguez, J. Subramanian, A. Quera-Bofarull, B. Krishnamurthy, B. A. Prakash, and R. Raskar, “Differentiable agent-based epidemiology,”arXiv preprint arXiv:2207.09714, 2022
-
[19]
V. P. Martínez, N. Di Paola, D. O. Alonso, U. Pérez-Sautu, C. M. Bellomo, A. A. Iglesias, R. M. Coelho, B. López, N. Periolo, P. A. Larson,et al., ““super-spreaders” and person-to- person transmission of andes virus in argentina,”New England Journal of Medicine, vol. 383, no. 23, pp. 2230–2241, 2020
work page 2020
-
[20]
Clusters of hantavirus infection, southern argentina,
M. E. Lázaro, G. E. Cantoni, L. M. Calanni, A. J. Resa, E. R. Herrero, M. A. Iacono, D. A. Enria, and S. M. G. Cappa, “Clusters of hantavirus infection, southern argentina,”Emerging Infectious Diseases, vol. 13, no. 1, p. 104, 2007
work page 2007
-
[21]
R.Li, S.Pei, B.Chen, Y.Song, T.Zhang, W.Yang, andJ.Shaman, “Substantialundocumented infection facilitates the rapid dissemination of novel coronavirus (sars-cov-2),”Science, vol. 368, no. 6490, pp. 489–493, 2020
work page 2020
-
[22]
Differential effects of intervention timing on covid-19 spread in the united states,
S. Pei, S. Kandula, and J. Shaman, “Differential effects of intervention timing on covid-19 spread in the united states,”Science advances, vol. 6, no. 49, p. eabd6370, 2020. 8
work page 2020
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.