Recognition: no theorem link
Travelling waves of invasion in microbial communities with phenotypic switching
Pith reviewed 2026-05-12 05:14 UTC · model grok-4.3
The pith
Phenotypic switching leaves competitor invasion speed unchanged but can speed up the switching population's invasion wave.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
In the minimal model of two competing species with one switching to a persister phenotype resilient to competition, the phenotypic switching has no effect on the speed of the travelling wave by which the competitors invade the first population. Conversely, phenotypic switching can speed up the wave by which this population invades their competitors. The results suggest that bacterial persistence can be an offensive rather than defensive ecological strategy.
What carries the argument
Travelling wave solutions in a reaction-diffusion system with stochastic and competitor-induced switching terms to a persister subpopulation that is resilient to competition.
If this is right
- Persistence confers an asymmetric advantage that accelerates expansion into occupied competitor territory.
- The invasion speed into competitors rises with the rate of switching to the persister state.
- The speed of competitor invasion remains insensitive to the presence of switching across the parameter ranges examined.
- Numerical solutions of the wave equations confirm the analytical predictions for both directions of invasion.
Where Pith is reading between the lines
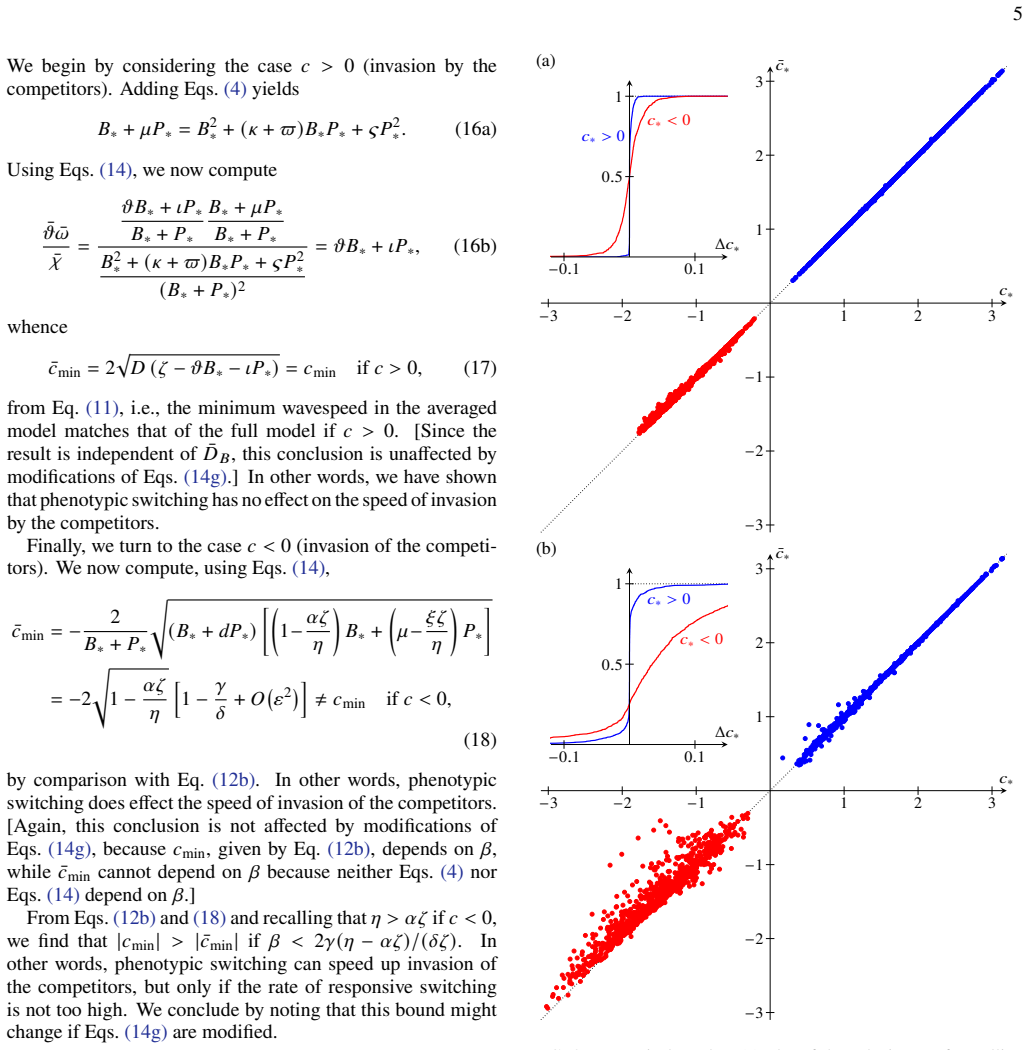
- The asymmetry could appear in other systems that use phenotypic switching, such as tumor invasion or invasive species fronts.
- Controlled lab assays in microfluidic channels could directly test whether measured wave speeds match the predicted directional difference.
- If persistence evolves under spatial competition, selection may favor offensive as well as survival benefits.
Load-bearing premise
The model assumes particular functional forms and rates for the switching to persisters together with the assumption that persisters are resilient specifically to competition.
What would settle it
A spatial competition experiment that measures invasion front speeds with and without the ability to switch to persisters would show unchanged speed in one direction and increased speed in the other if the claim holds.
Figures




read the original abstract
Complex microbial habitats see the spatial competition of different clonal bacterial populations that switch between different phenotypes. Here, we determine the effect of this subpopulation structure on the invasion of one species by another in a minimal model of two competing species: one species switches, both stochastically and in response to its competitor, to a persister phenotype resilient to competition. Surprisingly, our combined analytical and numerical results show that this phenotypic switching has no effect on the speed of the travelling wave by which the competitors invade the first population. Conversely, we discover that phenotypic switching can speed up the wave by which this population invades their competitors. Our results thus suggest, counterintuitively, that bacterial persistence can be an offensive, rather than defensive ecological strategy.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper analyzes a minimal two-species competition model in which one population switches stochastically and in response to competitor density to a persister phenotype that is completely resilient to competition. Using a combination of travelling-wave analysis (leading-edge linearization) and numerical simulations, the authors conclude that this switching leaves the speed of competitor invasion into the switching population unchanged, while it can increase the speed at which the switching population invades its competitors. The result is presented as counter-intuitive evidence that persistence can function as an offensive rather than purely defensive strategy.
Significance. If the central cancellation holds beyond the specific functional forms chosen, the work supplies an analytically tractable example in which subpopulation structure alters spatial invasion dynamics in a non-obvious, directional manner. The combination of closed-form wave-speed expressions with numerical confirmation of the pulled-front regime is a clear strength; it allows direct identification of the parameter regimes in which switching accelerates invasion.
major comments (2)
- [§3 (travelling-wave analysis)] The claim that competitor invasion speed is independent of switching rates (abstract and §3) is obtained from the leading-edge linearization under the exact assumptions of constant stochastic switching, linear competitor-induced switching, and zero competition effect on persister growth/death rates. These choices decouple the persister compartment from the marginal stability condition by construction; the paper provides no structural proof or numerical scan showing that the cancellation survives other biologically plausible choices (e.g., nonlinear induced-switching or partial persister resilience). This is load-bearing for the headline “no effect” result.
- [§4 (numerical results)] The numerical evidence that switching can accelerate invasion by the switching population is shown only for the same minimal closure. A brief robustness check against modest changes in the functional form of induced switching or persister mortality would strengthen the claim that the acceleration is a generic consequence of the two-phenotype structure rather than an artifact of the linear, absolute-resilience assumptions.
minor comments (2)
- [Abstract] The abstract states the result for “the competitors invade the first population” without reminding the reader that this independence is tied to the specific minimal-model closure; a single qualifying clause would prevent over-generalization by readers.
- [§2 (model definition)] Notation for the two switching rates (stochastic vs. competitor-induced) is introduced without an explicit table or equation block that lists all parameters and their biological interpretation; this makes it harder to track which parameters drop out of the wave-speed formula.
Simulated Author's Rebuttal
We thank the referee for the constructive comments on our manuscript. We address the major points below and outline the revisions we will make to clarify the scope of our results and strengthen the supporting evidence.
read point-by-point responses
-
Referee: [§3 (travelling-wave analysis)] The claim that competitor invasion speed is independent of switching rates (abstract and §3) is obtained from the leading-edge linearization under the exact assumptions of constant stochastic switching, linear competitor-induced switching, and zero competition effect on persister growth/death rates. These choices decouple the persister compartment from the marginal stability condition by construction; the paper provides no structural proof or numerical scan showing that the cancellation survives other biologically plausible choices (e.g., nonlinear induced-switching or partial persister resilience). This is load-bearing for the headline “no effect” result.
Authors: We agree that the exact independence of competitor invasion speed on switching rates follows directly from the leading-edge linearization under the stated assumptions of the minimal model. The persister compartment is decoupled from the marginal stability condition by construction in this setup, and we do not provide a general structural proof or claim that the cancellation holds for arbitrary functional forms. The manuscript presents this as an exact result for the chosen minimal model, which was selected for analytical tractability to illustrate a directional effect of phenotypic switching. We will revise the abstract, §3, and the discussion to explicitly highlight these assumptions and note that the 'no effect' result is specific to this closure. This addresses the load-bearing nature of the claim without overstating generality. revision: yes
-
Referee: [§4 (numerical results)] The numerical evidence that switching can accelerate invasion by the switching population is shown only for the same minimal closure. A brief robustness check against modest changes in the functional form of induced switching or persister mortality would strengthen the claim that the acceleration is a generic consequence of the two-phenotype structure rather than an artifact of the linear, absolute-resilience assumptions.
Authors: We concur that the numerical demonstration of accelerated invasion by the switching population is currently limited to the minimal model assumptions. To strengthen the interpretation that the acceleration arises from the two-phenotype structure, we will add a brief robustness analysis in the revised manuscript (as a new subsection or appendix). This will include numerical simulations with nonlinear forms of competitor-induced switching and small nonzero persister mortality rates, confirming that the increase in invasion speed persists under these modest perturbations. These checks will be presented alongside the existing results. revision: yes
Circularity Check
No circularity; derivation self-contained from model equations
full rationale
The paper constructs a minimal PDE model with explicit functional forms for stochastic and competitor-induced switching plus persister resilience, then obtains travelling-wave speeds via leading-edge linearization and numerical integration. The reported independence of competitor invasion speed from switching parameters is a direct algebraic consequence of those equations (effective growth rate of the invader decouples from the persister compartment under the stated linearity and zero-competition assumptions) rather than a definitional identity or self-citation chain. No step reduces the target result to its own inputs by construction, and the analysis does not rely on load-bearing self-citations or fitted parameters renamed as predictions. The result is therefore model-derived and self-contained.
Axiom & Free-Parameter Ledger
Reference graph
Works this paper leans on
-
[1]
There, near their equilibria𝑿 ±, Eqs
Necessary conditions for the existence of a travelling wave To obtain necessary conditions for the existence of a travelling wave, we consider the dynamics for𝑧→ ±∞. There, near their equilibria𝑿 ±, Eqs. (5) linearise to𝑿 ′ =J ± ·𝑿, where the JacobiansJ ± are given by J− = © « 0 1 0 0 0 0 𝛾−1+ (𝛼+𝛽) 𝜁 𝜂 −𝑐−𝛿0 0 0 0 0 0 1 0 0 − 1 𝑑 𝛽𝜁 𝜂 +𝛾 0 1 ...
-
[2]
Minimum wavespeed of travelling waves We can now obtain a lower bound,|𝑐|⩾|𝑐 min|, on the wavespeed of these travelling waves. The key observation is the following: In the(𝐵, 𝑅)or(𝑃, 𝑆)planes, the dynamics near𝑿 − are dominated by the stable eigenmode ofJ − that has the slowest decay as𝑧→ −∞, since the𝑁 − boundary conditions have removed the unstable eige...
-
[3]
Averaged model: effect of phenotypic variation To analyse the effect of phenotypic variation on these trav- elling waves of invasion, we need to compare our results to results for an averaged model [9, 10] without phenotypic vari- ation, with two effective populations ¯𝐵(𝑥, 𝑡), corresponding to bacteria or persisters, and ¯𝐴(𝑥, 𝑡), corresponding to compet...
-
[4]
Hence, as𝐵→𝛿/𝜅 − or𝐵→𝛿/𝜅 +,𝑃→ ∞in the first quadrant
Proof of Proposition 1 Equations (4) define(𝐵 ∗, 𝑃∗)as the intersection, in the pos- itive quadrant of the(𝐵, 𝑃)plane, of the hyperbolae H1 :𝐵 2 +𝜅𝐵𝑃+ (𝛾−1)𝐵−𝛿𝑃=0,(A1a) H2 :𝜛𝐵𝑃+𝜍 𝑃 2 −𝛾𝐵+ (𝛿−𝜇)𝑃=0.(A1b) We notice thatH 1 has a vertical asymptote𝐵=𝛿/𝜅. Hence, as𝐵→𝛿/𝜅 − or𝐵→𝛿/𝜅 +,𝑃→ ∞in the first quadrant. As𝐵→ ∞,𝑃∼ −𝜅𝐵→ −∞, so this branch must enter or lea...
-
[5]
Proof of Proposition 2 To prove Proposition 2, we analyse the eigenvalues of the JacobiansJ ±, given by Eqs. (6). Both Jacobians have a block matrix structure, J± = A± B± C± D± ! ,(A3) whereB − =C + =0. The eigenvalues ofJ ± are therefore the eigenvalues ofA ± andD ±, respectively. The eigenvalues ofD ± are determined by quadratic equations, and are given...
-
[6]
We take the initial conditions for Eqs
Numerical integration We integrate the partial differential equations (1) and (13) inMatlab(The MathWorks, Inc.) using thepdepefunction. We take the initial conditions for Eqs. (1) to be 𝐵(𝑥,0)= 𝐵∗ 2 (1+tanh𝑘𝑥),(C1a) 𝑃(𝑥,0)= 𝑃∗ 2 (1+tanh𝑘𝑥),(C1b) 𝐴(𝑥,0)= 𝐴∗ 2 (1−tanh𝑘𝑥),(C1c) 11 which defines the steepness parameter𝑘. To compute these ini- tial conditions...
-
[7]
Sampling of random systems To sample random systems, we sample the model parameters independently from uniform distributions. When sampling pa- rameters obeying the persister scalings (2), parameters that are of order𝑂(𝜀)are sampled from theU[0, 𝜀]distribution, while other parameters are sampled from theU[1/𝑓 , 𝑓]distribution for some𝑓 >1. This allows sam...
-
[8]
(We only integrate the dynamics for random systems satisfying these conditions.) a
Travelling-wave conditions For each random system sampled, we check whether the necessary conditions for the existence of a travelling wave of invasion are satisfied in the full or averaged models, and, if they are, compute the minimum wave velocities𝑐 min or ¯𝑐min. (We only integrate the dynamics for random systems satisfying these conditions.) a. Necess...
-
[9]
Additional discussion of the numerical results In Figs. 3 and 4 of the main text, we report wave speeds for those randomly sampled systems for which the necessary conditions for the existence of a travelling wave are satisfied in both the full model (1) and the averaged model (13) and for which the computations of both wave speeds converge. In this final ...
- [10]
-
[11]
Avery, Microbial cell individuality and the underlying sources of heterogeneity, Nat
S. Avery, Microbial cell individuality and the underlying sources of heterogeneity, Nat. Rev. Microbiol.4, 577 (2006)
work page 2006
-
[12]
D. Dubnau and R. Losick, Bistability in bacteria, Mol. Micro- biol.61, 564 (2006)
work page 2006
-
[13]
D. I. Bolnick, P. Amarasekare, M. S. Ara´ ujo, R. B¨ urger, J. M. Levine, M. Novak, V. H. Rudolf, S. J. Schreiber, M. C. Urban, and D. A. Vasseur, Why intraspecific trait variation matters in community ecology, Trends Ecol. Evol.26, 183 (2011)
work page 2011
-
[14]
A. Forsman and L. Wennersten, Inter-individual variation pro- motes ecological success of populations and species: evidence from experimental and comparative studies, Ecography39, 630 (2016)
work page 2016
-
[15]
M. M. Turcotte and J. M. Levine, Phenotypic plasticity and species coexistence, Trends Ecol. Evol.31, 803 (2016)
work page 2016
-
[16]
E. Kussell and S. Leibler, Phenotypic diversity, population growth, and information in fluctuating environments, Science 309, 2075 (2005)
work page 2075
-
[17]
D. S. Maynard, C. A. Serv ´an, J. A. Capit ´an, and S. Allesina, Phenotypic variability promotes diversity and stability in com- petitive communities, Ecol. Lett.22, 1776 (2019)
work page 2019
-
[18]
P. A. Haas, N. M. Oliveira, and R. E. Goldstein, Subpopula- tions and stability in microbial communities, Phys. Rev. Res.2, 022036(R) (2020)
work page 2020
-
[19]
P. A. Haas, M. A. Gutierrez, N. M. Oliveira, and R. E. Goldstein, Stabilization of microbial communities by responsive pheno- typic switching, Phys. Rev. Res.4, 033224 (2022)
work page 2022
-
[20]
E. M. Holdridge and D. A. Vasseur, Intraspecific variation pro- motes coexistence under competition for essential resources, Theor. Ecol.15, 225 (2022)
work page 2022
-
[21]
R. M. May, Will a large complex system be stable?, Nature (London)238, 413 (1972)
work page 1972
-
[22]
S. Allesina and S. Tang, The stability-complexity relationship at age 40: a random matrix perspective, Pop. Ecol.57, 63 (2015)
work page 2015
-
[23]
N. Q. Balaban, J. Merrin, R. Chait, L. Kowalik, and S. Leibler, Bacterial persistence as a phenotypic switch, Science305, 1622 (2004)
work page 2004
-
[24]
E. Maisonneuve and K. Gerdes, Molecular mechanisms under- lying bacterial persisters, Cell157, 539 (2014)
work page 2014
- [25]
-
[26]
J. L. Radzikowski, H. Schramke, and M. Heinemann, Bacte- rial persistence from a system-level perspective, Curr. Opin. Biotechnol.46, 98 (2017)
work page 2017
-
[27]
J. D. Murray,Mathematical Biology, 3rd ed., Vol. I (Springer, Berlin, Germany, 2002), Chap. 3, pp. 79–118 and Chap. 13, pp. 437–483
work page 2002
-
[28]
J. Hofbauer and K. Sigmund,Evolutionary Games and Popula- tion Dynamics(Cambridge University Press, Cambridge, Eng- land, 1998) Chap. 2–5, pp. 11–54
work page 1998
-
[29]
S. R. Dunbar, Traveling wave solutions of diffusive Lotka– Volterra equations: A heteroclinic connection inR 4, Trans. Am. Math. Soc.286, 557 (1984)
work page 1984
-
[30]
M. A. Lewis, B. Li, and H. F. Weinberger, Spreading speed and linear determinacy for two-species competition models, J. Math. Biol.45, 219 (2002)
work page 2002
- [31]
-
[32]
J. Fang and X.-Q. Zhao, Monotone wavefronts for partially de- generate reaction-diffusion systems, J. Dyn. Differ. Equ.21, 663 (2009)
work page 2009
-
[33]
Wang, Spreading speeds and traveling waves for non- cooperative reaction–diffusion systems, J
H. Wang, Spreading speeds and traveling waves for non- cooperative reaction–diffusion systems, J. Nonlinear Sci.21, 747 (2011)
work page 2011
-
[34]
van Saarloos, Front propagation into unstable states, Phys
W. van Saarloos, Front propagation into unstable states, Phys. Rep.386, 29 (2003)
work page 2003
-
[35]
A. Alhasanat and C. Ou, Minimal-speed selection of traveling waves to the Lotka–Volterra competition model, J. Diff. Equ. 266, 7357 (2019)
work page 2019
-
[36]
L. Tang and S. Chen, Traveling wave solutions for the diffusive Lotka–Volterra equations with boundary problems, Appl. Math. Comput.413, 126599 (2022)
work page 2022
-
[37]
Y. Wang, H. Li, and X. Li, Travelling wave fronts of Lotka– Volterra reaction-diffusion system in the weak competition case, Proc. R. Soc. Edinb.152, 912–938 (2022)
work page 2022
-
[38]
X. Ma, R. Liu, and L. Cai, Stability of traveling wave solutions for a nonlocal Lotka–Volterra model, Math. Biosci. Eng.21, 444 (2024)
work page 2024
-
[39]
X. Cao, Y. Wang, and M. Yao, Nonlinear stability of travel- ing wave fronts to the Lotka–Volterra competition system, Z. Angew. Math. Mech.105, e202300500 (2025)
work page 2025
-
[40]
C.-C. Chen, T.-Y. Hsiao, and S.-C. Wang, Non-monotone trav- eling waves of the weak competition Lotka–Volterra system, Discrete Contin. Dyn. Syst. B37, 206 (2026)
work page 2026
-
[41]
M. J. Simpson and S. W. McCue, Fisher–KPP-type models of biological invasion: open source computational tools, key con- cepts and analysis, Proc. R. Soc. A480, 20240186 (2024)
work page 2024
-
[42]
C.-K. Yap,Fundamental Problems in Algorithmic Algebra(Ox- ford University Press, Oxford, England, 2000) Chap. 6, pp. 141– 185
work page 2000
-
[43]
N. M. Oliveira, J. H. R. Wheeler, C. Deroy, S. C. Booth, E. J. Walsh, W. M. Durham, and K. R. Foster, Suicidal chemotaxis in bacteria, Nat. Commun.13, 7608 (2022)
work page 2022
-
[44]
Code is available atdoi.org/10.5281/zenodo.20072527
-
[45]
D. J. H. Garling,A Course in Mathematical Analysis, Vol. 1 (Cambridge University Press, Cambridge, England, 2013) Chap. 6–7, pp. 147–208
work page 2013
-
[46]
This is of course a mathematical assumption, but does not restrict the physical ar- gument
We neglect degenerate parameter values leading, e.g., to zero eigenvalues, tacitly throughout this paper. This is of course a mathematical assumption, but does not restrict the physical ar- gument
-
[47]
The confidence intervals are 95% Wilson intervals: see, e.g., L. D. Brown, T. T. Cai, and A. DasGupta, Interval estimation for a binomial proportion, Stat. Sci.16, 101 (2001)
work page 2001
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.