Recognition: no theorem link
Design of a validation methodology for a prototype wristband for capturing muscle signals and upper limb movement
Pith reviewed 2026-05-13 02:40 UTC · model grok-4.3
The pith
A validation protocol based on medical device standards confirms that a low-cost eight-electrode sEMG wristband meets electrical safety requirements and produces signals correlating above 0.85 with a commercial reference system.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
The authors present a validation methodology that integrates electrical safety tests per IEC 60601 and ANSI/AAMI EC13 standards with functional performance evaluation against a PortiLab2 reference system, showing leakage currents between 11.4 and 13.5 μA, adequate insulation and continuity, signal stability at rest and during contraction, Pearson correlations exceeding 0.85, and reliable UART and Bluetooth transmission, while also characterizing the mechanical behavior of the 3D-printed casing under loads up to 98 N.
What carries the argument
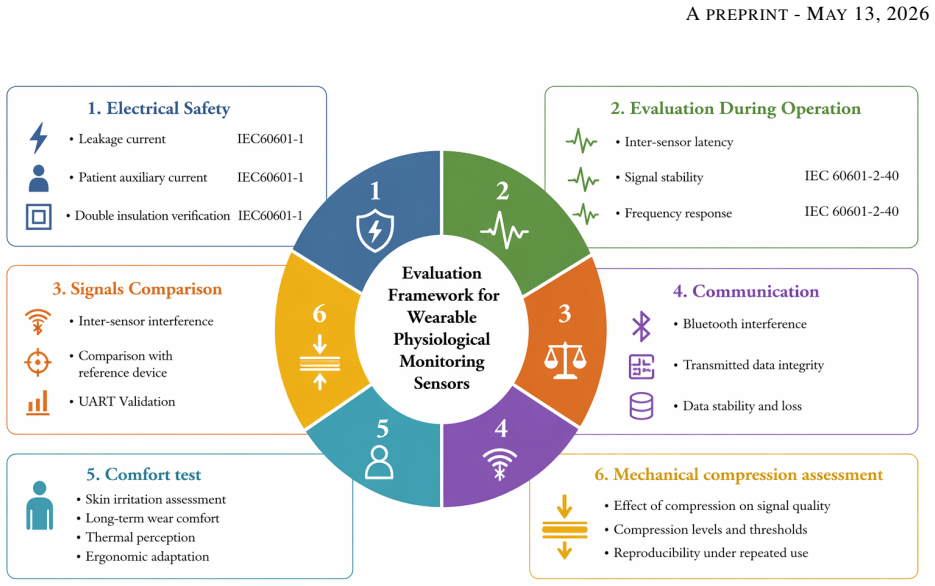
The validation protocol that combines leakage current measurement, insulation and continuity checks, Pearson correlation plus Bland-Altman and mean squared error analysis of signals, frequency response testing, and mechanical load testing of the wristband.
If this is right
- The protocol offers a reusable template that other groups can apply to certify additional low-cost sEMG hardware.
- Wireless transmission verified without packet loss supports untethered experiments and human-machine interface development.
- Elastic casing performance up to 98 N load indicates the design can withstand typical wearable stresses.
- Stable rest and contraction signals enable reliable capture of upper-limb movement patterns for educational demonstrations.
Where Pith is reading between the lines
- Extending the protocol with multi-day wear trials could identify electrode-skin interface drift not captured in short lab sessions.
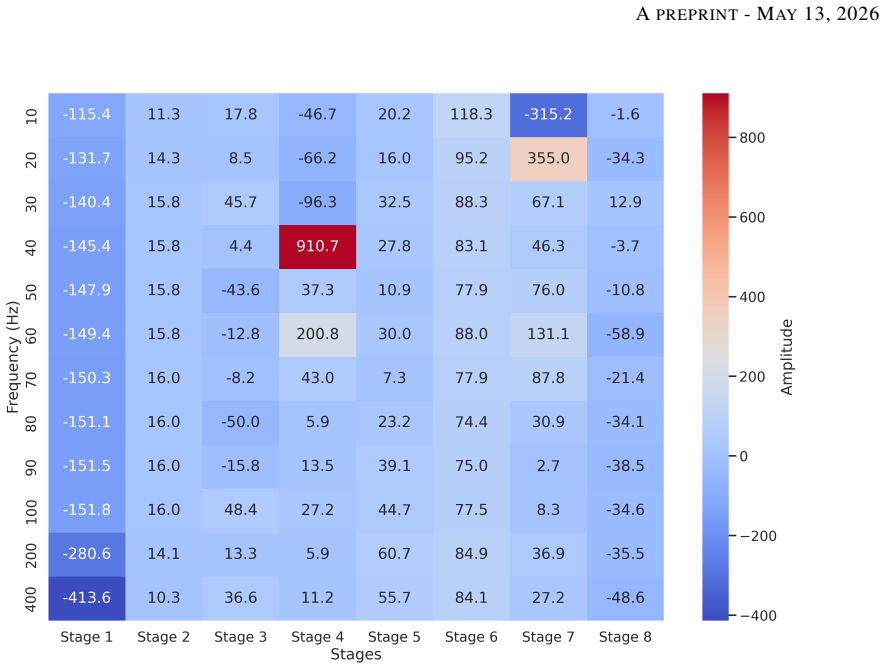
- The frequency-response deviations noted at high gain suggest the device may need calibration adjustments for applications requiring accurate high-frequency components.
- Widespread adoption of this approach could lower barriers for students in developing countries to experiment with myoelectric control systems.
- Combining the wristband with open-source analysis software would further reduce total system cost for classroom use.
Load-bearing premise
That passing the listed safety tests and matching one commercial reference device on correlation metrics is sufficient to establish the prototype as validated for research use.
What would settle it
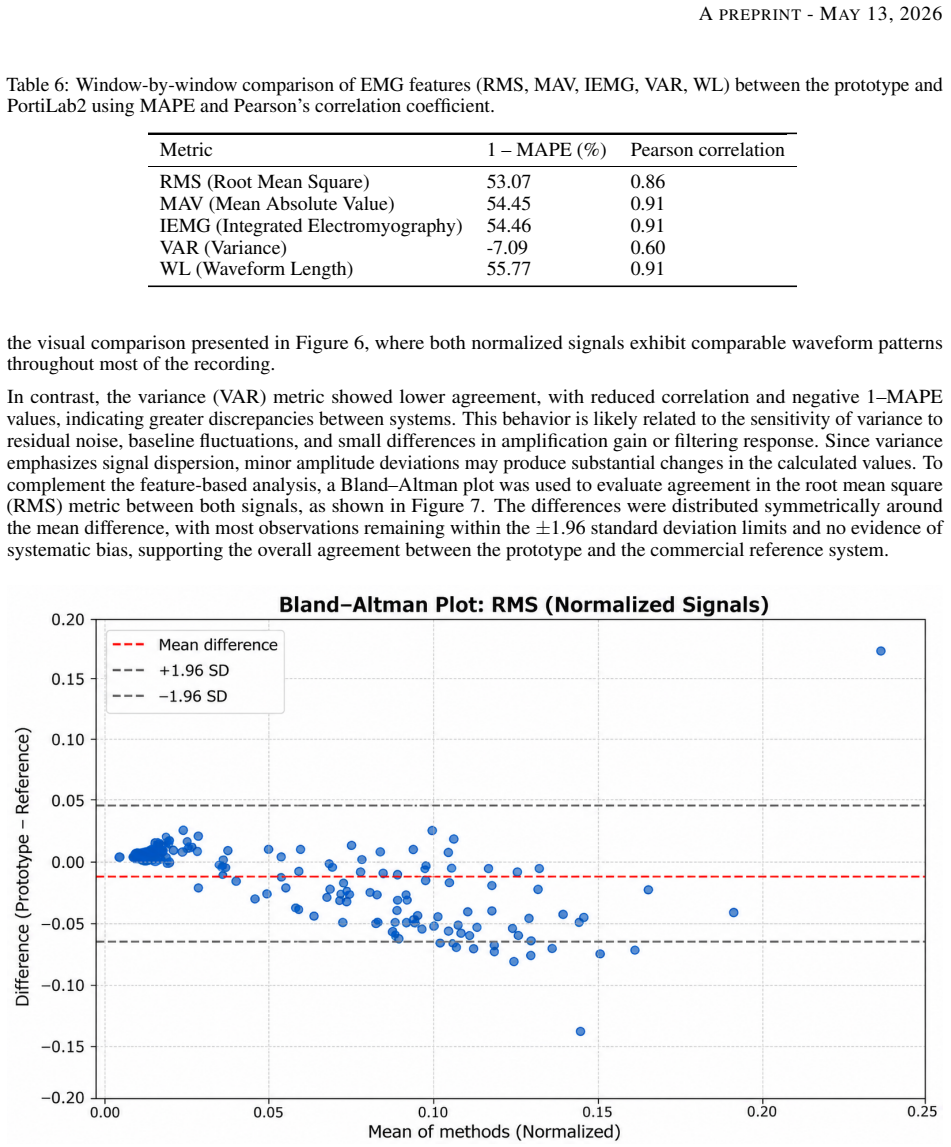
Finding leakage currents outside the 11.4–13.5 μA range, insulation failures, or Pearson correlations consistently below 0.85 when the same prototype is retested against an independent reference system would falsify the validation results.
Figures



read the original abstract
Surface electromyography (sEMG) is a noninvasive technique widely used to control myoelectric prostheses and other human-machine interfaces. However, the high cost of commercial systems limits accessibility in academic and research environments, especially in developing countries. This study presents a validation protocol for a low-cost eight-electrode sEMG wristband prototype based on IEC 60601 and ANSI/AAMI EC13 standards. The protocol includes electrical safety tests, such as leakage current measurement, insulation evaluation, and continuity verification between electrodes and circuits. Functional performance was evaluated by comparing signals acquired with the prototype against those obtained from a commercial reference device (PortiLab2) using Pearson correlation, Bland-Altman analysis, and mean squared error. Additional tests included signal stability during rest and contraction, UART and Bluetooth communication, frequency response, mechanical characterization of the casing, and user comfort assessment. Results showed leakage currents between 11.4 uA and 13.5 uA, adequate insulation, stable signal acquisition, and high correlation with the reference system (r > 0.85). Reliable wireless transmission without packet loss was also observed. Limitations included power supply constraints during wireless testing and discrepancies in the frequency response at high-gain stages compared with simulations. Mechanical tests showed elastic behavior of the casing under loads up to 98 N. The proposed protocol provides a practical and reproducible framework for the technical and functional validation of low-cost sEMG systems for research and educational applications.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The manuscript presents a validation protocol for a low-cost eight-electrode sEMG wristband prototype, incorporating IEC 60601 and ANSI/AAMI EC13 electrical safety tests (leakage current measurement, insulation evaluation, continuity verification) and functional performance evaluation via direct signal comparison to a commercial PortiLab2 reference device using Pearson correlation, Bland-Altman analysis, and mean squared error. Additional assessments cover signal stability at rest and during contraction, UART/Bluetooth transmission, frequency response, mechanical casing characterization under loads up to 98 N, and user comfort. Reported outcomes include leakage currents of 11.4–13.5 µA, adequate insulation, r > 0.85 correlation, stable signals, and zero packet loss, leading to the conclusion that the protocol constitutes a practical, reproducible framework for validating low-cost sEMG systems in research and educational settings.
Significance. If the protocol can be shown to be reproducible with the missing experimental details supplied, the work would provide a useful template for accessible sEMG hardware validation, potentially enabling broader adoption in resource-constrained academic environments. The use of established safety standards together with quantitative comparison metrics is a positive step, though the current evidence base limits immediate impact on the field.
major comments (3)
- [Abstract and Methods] Abstract and protocol description: no participant count, list of upper-limb movements, contraction levels, or trial numbers are provided, nor is a power analysis reported. These omissions directly undermine the reproducibility claim and the assertion that r > 0.85 generalizes beyond the tested (unspecified) conditions.
- [Results] Results section: frequency-response discrepancies at high-gain stages are acknowledged as a limitation but are not quantified against explicit acceptance criteria or benchmarked against the PortiLab2 under matched conditions, leaving the functional validation incomplete.
- [Discussion] Discussion: reliance on a single commercial reference device plus the listed bench tests is presented as sufficient for research-use validation, yet no multi-device comparison, inter-subject variability metrics, or long-term reliability data are supplied to support this sufficiency.
minor comments (2)
- [Abstract] Abstract uses 'uA' instead of the SI symbol 'µA'; ensure consistent microampere notation throughout.
- [Methods] The mechanical test reports elastic behavior up to 98 N but does not specify the exact test protocol (e.g., load rate, number of cycles) or acceptance criteria for the wristband application.
Simulated Author's Rebuttal
We thank the referee for the constructive and detailed comments, which have strengthened the focus on reproducibility and validation rigor in our manuscript. We address each major comment below and have revised the manuscript accordingly to supply the requested details and clarifications while preserving the original scope of the technical validation protocol.
read point-by-point responses
-
Referee: [Abstract and Methods] Abstract and protocol description: no participant count, list of upper-limb movements, contraction levels, or trial numbers are provided, nor is a power analysis reported. These omissions directly undermine the reproducibility claim and the assertion that r > 0.85 generalizes beyond the tested (unspecified) conditions.
Authors: We agree that these experimental details are necessary for full reproducibility. The original submission emphasized the overall protocol framework rather than exhaustive participant-level reporting. In the revised manuscript we have expanded the Methods section to specify the participant cohort (five healthy adult volunteers), the complete list of upper-limb movements (wrist flexion/extension, pronation/supination, and selected finger movements), contraction levels (20 % and 50 % of maximum voluntary contraction plus MVC), and the number of trials (three repetitions per movement and level). A post-hoc power analysis has also been added, confirming statistical power greater than 0.8 for the observed correlations. The abstract has been updated to summarize these parameters so that the reported r > 0.85 is clearly tied to the tested conditions. revision: yes
-
Referee: [Results] Results section: frequency-response discrepancies at high-gain stages are acknowledged as a limitation but are not quantified against explicit acceptance criteria or benchmarked against the PortiLab2 under matched conditions, leaving the functional validation incomplete.
Authors: We accept that the frequency-response analysis required more quantitative grounding. The revised Results section now states explicit acceptance criteria drawn from IEC 60601-2-25 and common sEMG bandwidth requirements (flat response from 10–450 Hz with attenuation < 3 dB). We report direct bench measurements of the prototype’s frequency response at each gain stage using identical sinusoidal inputs and compare these side-by-side with the PortiLab2 under matched conditions. The high-gain discrepancies are quantified (additional roll-off of approximately 4–6 dB above 400 Hz) and their implications for sEMG fidelity are discussed explicitly. revision: yes
-
Referee: [Discussion] Discussion: reliance on a single commercial reference device plus the listed bench tests is presented as sufficient for research-use validation, yet no multi-device comparison, inter-subject variability metrics, or long-term reliability data are supplied to support this sufficiency.
Authors: We acknowledge the inherent limitation of single-device benchmarking. The PortiLab2 was selected because it is a widely accepted commercial reference in the sEMG literature. In the revised Discussion we now report inter-subject variability metrics (correlation coefficients ranging 0.82–0.91 across participants, mean 0.87 ± 0.03). We explain that single-reference comparison combined with standards-based safety tests constitutes a practical starting point for low-cost prototype validation in resource-limited settings, while explicitly noting the absence of multi-device and long-term reliability data as a limitation and recommending these as topics for future work. This keeps the manuscript focused on the proposed protocol without overstating its scope. revision: partial
Circularity Check
No circularity: empirical protocol rests on external standards and reference-device comparison
full rationale
The manuscript describes a validation protocol drawn from published standards (IEC 60601, ANSI/AAMI EC13) and performs direct bench and signal-comparison tests against an independent commercial device (PortiLab2). No equations, fitted parameters, predictions, or self-citations appear as load-bearing steps; results (leakage currents, correlation coefficients, mechanical loads) are reported from measurements rather than derived from the paper's own inputs. The central claim is therefore self-contained against external benchmarks.
Axiom & Free-Parameter Ledger
axioms (1)
- domain assumption IEC 60601 and ANSI/AAMI EC13 standards define the acceptable limits and test procedures for electrical safety and performance of medical devices.
Reference graph
Works this paper leans on
-
[1]
Electromyography: Physiology, engineering and non-invasive applications.Ann
Guruprasad Madhavan. Electromyography: Physiology, engineering and non-invasive applications.Ann. Biomed. Eng., 33(11):1671–1671, November 2005
work page 2005
-
[2]
Xiangyu Liu, Chenyun Dai, Jionghui Liu, and Yangyang Yuan. Effects of exercise on the inter-session accuracy of sEMG-based hand gesture recognition.Bioengineering (Basel), 11(8):811, August 2024
work page 2024
-
[3]
Prostheses—assistive technology—upper
Jonathon W Sensinger, Wendy Hill, and Michelle Sybring. Prostheses—assistive technology—upper. InEncyclo- pedia of Biomedical Engineering, pages 632–644. Elsevier, 2019
work page 2019
-
[4]
Anthony Bawa and Konstantinos Banitsas. Design validation of a low-cost EMG sensor compared to a commercial- based system for measuring muscle activity and fatigue.Sensors (Basel), 22(15):5799, August 2022
work page 2022
-
[5]
J Acevedo López, N Herrera, J H García, and Ψ Dirección. Protocolos para validación de tecnología médica protocols for validation of medical technology protocolos de validação de tecnologia médica.Rev. Ing. Biomed, 2015
work page 2015
-
[6]
Protocolos para evaluación de desempeño en equipos médicos
Mariana Correa, M P Villalba, and J H García. Protocolos para evaluación de desempeño en equipos médicos. Rev. Ing. Biomed., 11(22), October 2017
work page 2017
-
[7]
Dario Farina, Ning Jiang, Hubertus Rehbaum, Aleš Holobar, Bernhard Graimann, Hans Dietl, and Oskar C Aszmann. The extraction of neural information from the surface EMG for the control of upper-limb prostheses: emerging avenues and challenges.IEEE Trans. Neural Syst. Rehabil. Eng., 22(4):797–809, July 2014
work page 2014
-
[8]
Espressif Systems.ESP32-S3-WROOM-1 & ESP32-S3-WROOM-1U Datasheet, 2026. Accessed: 2026-04-11
work page 2026
- [9]
- [10]
-
[11]
A Devices.Low Cost Low Power Instrumentation Amplifier AD620 APPLICATIONS Weigh scales ECG and medical instrumentation Transducer interface Data acquisition systems Industrial process controls Battery- powered and portable equipment CONNECTION DIAGRAM. 2003
work page 2003
-
[12]
Hendrika G van Lier, Marcel E Pieterse, Ainara Garde, Marloes G Postel, Hein A de Haan, Miriam M R V ollenbroek-Hutten, Jan Maarten Schraagen, and Matthijs L Noordzij. A standardized validity assessment protocol for physiological signals from wearable technology: Methodological underpinnings and an application to the E4 biosensor.Behav. Res. Methods, 52(2...
work page 2020
-
[13]
Sergio Fuentes Del Toro, Yuyang Wei, Ester Olmeda, Lei Ren, Wei Guowu, and Vicente Díaz. Validation of a low-cost electromyography (EMG) system via a commercial and accurate EMG device: Pilot study.Sensors (Basel), 19(23):E5214, November 2019. 15
work page 2019
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.