Recognition: 2 theorem links
· Lean TheoremBistability, Absolute Concentration Robustness, and Hysteresis in Dual-Site Futile Cycles with Bifunctional Enzymes
Pith reviewed 2026-05-13 01:49 UTC · model grok-4.3
The pith
In one dual-site futile cycle with bifunctional enzymes, the system exhibits bistability and absolute concentration robustness in the final product.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
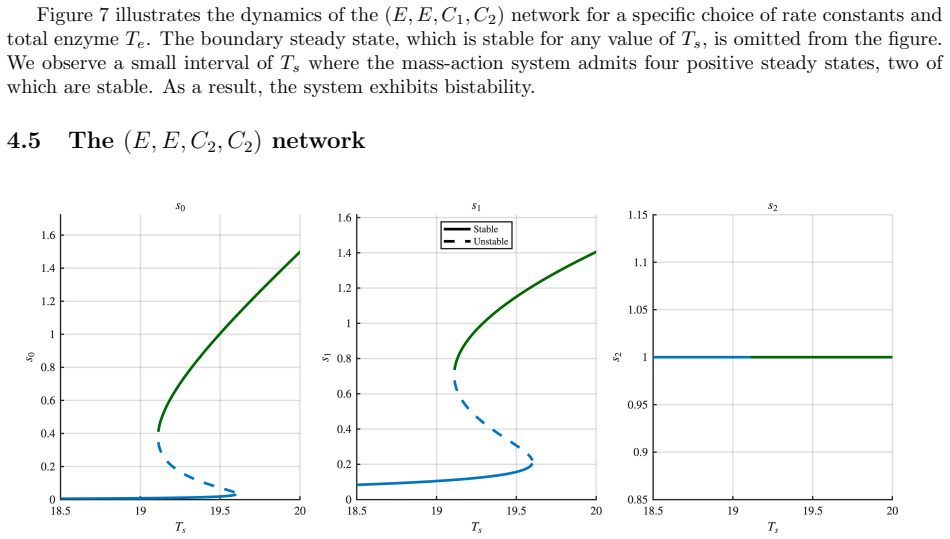
All four networks admit boundary steady states. The networks differ in the number and stability of boundary steady states, in the maximum number of positive steady states (ranging from two to four), and in whether bistability is present. In two networks, a transcritical bifurcation connects the boundary and positive steady state branches; in one case this is a backward bifurcation, producing hysteresis. One network simultaneously exhibits bistability and ACR in the final modification state, where the system can settle into either of two stable steady states with different intermediate concentrations yet identical final product concentration.
What carries the argument
Bifunctional enzyme-substrate compounds that perform the reverse modification steps, which shape the steady-state branches and enable both multiple stable states and absolute concentration robustness in the final product.
Load-bearing premise
The networks follow mass-action kinetics on the specific topologies where reverse steps occur through bifunctional enzyme-substrate compounds.
What would settle it
In a biological system matching one of these topologies, record the final product concentration at two different total substrate amounts that produce distinct stable intermediate levels; if the final concentrations differ, the simultaneous bistability and ACR claim is false.
Figures







read the original abstract
Bifunctional enzymes, which catalyze both the forward and reverse steps of a substrate modification reaction, arise naturally in bacterial two-component signaling systems and metabolic regulation. Beyond their well-known role in conferring absolute concentration robustness (ACR) on substrate species, bifunctional enzymes profoundly shape the dynamical landscape of the networks in which they appear. We study a class of dual-site futile cycles in which the reverse modification steps are carried out by bifunctional enzyme-substrate compounds, and provide a complete mathematical analysis of all four such networks, characterizing the existence, number, and stability of steady states, as well as the bifurcation structure as total substrate is varied. All four networks admit boundary steady states, in contrast to the non-bifunctional case. The networks differ in the number and stability of boundary steady states, in the maximum number of positive steady states (ranging from two to four), and in whether bistability is present. In two networks, a transcritical bifurcation connects the boundary and positive steady state branches; in one case this is a backward bifurcation, producing hysteresis. Perhaps the most striking phenomenon occurs in one of the four networks, which simultaneously exhibits bistability and ACR in the final modification state, where the system can settle into either of two stable steady states with different intermediate concentrations yet identical final product concentration.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper examines four dual-site futile cycle networks with bifunctional enzymes catalyzing reverse steps using mass-action ODEs. It delivers a full analysis of steady-state existence, multiplicity, stability, and bifurcations parameterized by total substrate. Boundary steady states exist in all cases, positive steady states range from 2 to 4, bistability occurs in some, and one network combines bistability (from backward transcritical bifurcation) with ACR in the terminal species, permitting two stable states with the same final product level but different intermediates.
Significance. This result is significant because it reveals how bifunctional enzymes can generate both robustness (ACR) and multistability in the same system, a combination not commonly analyzed in futile cycle models. The exhaustive treatment of all four topologies and the explicit bifurcation diagrams provide a solid foundation for understanding these motifs in two-component signaling systems. The mathematical rigor, including boundary state analysis contrasting with non-bifunctional cases, adds value to the field of mathematical systems biology.
major comments (1)
- [§4 (analysis of the network exhibiting bistability and ACR)] The central claim of simultaneous bistability and ACR in the final modification state depends on the two stable positive steady states having identical concentrations for the terminal species. The manuscript should explicitly derive or highlight the algebraic identity in the steady-state equations that makes the terminal species concentration independent of total substrate and the same for both equilibria. Without this explicit reduction, it is unclear if the invariance holds for generic parameter values or requires additional assumptions on the bifunctional complex equilibria.
minor comments (1)
- Clarify the labeling of the four networks throughout the text and figures to facilitate comparison of their differing behaviors.
Simulated Author's Rebuttal
We thank the referee for their thorough review, positive evaluation of the work's significance, and constructive suggestion. We address the major comment below and have incorporated the requested clarification into the revised manuscript.
read point-by-point responses
-
Referee: [§4 (analysis of the network exhibiting bistability and ACR)] The central claim of simultaneous bistability and ACR in the final modification state depends on the two stable positive steady states having identical concentrations for the terminal species. The manuscript should explicitly derive or highlight the algebraic identity in the steady-state equations that makes the terminal species concentration independent of total substrate and the same for both equilibria. Without this explicit reduction, it is unclear if the invariance holds for generic parameter values or requires additional assumptions on the bifunctional complex equilibria.
Authors: We agree that an explicit algebraic derivation strengthens the presentation. In the revised Section 4, we now derive the identity directly from the steady-state mass-action equations for the network in question. Setting the time derivatives to zero and using the conservation relations for the bifunctional enzyme-substrate complexes yields a cancellation in the equation for the terminal species, resulting in an algebraic relation that depends only on the rate constants and is independent of total substrate. This common value is attained by both positive steady states (including the two stable ones connected by the backward transcritical bifurcation), confirming ACR without requiring special assumptions on the complex equilibria beyond the standard mass-action framework and generic positivity of parameters. We have added a dedicated paragraph and a short appendix entry highlighting this reduction. revision: yes
Circularity Check
No circularity: direct algebraic analysis of mass-action steady states and bifurcations
full rationale
The paper derives the existence, multiplicity, and stability of steady states for the four dual-site futile cycle networks by solving the mass-action ODE steady-state equations directly and applying standard bifurcation techniques (transcritical, backward bifurcation) as total substrate varies. The simultaneous bistability-plus-ACR claim for one network follows from showing that the terminal species concentration satisfies an invariant algebraic relation independent of the two positive roots and of total substrate. No parameters are fitted, no predictions are renamed fits, and no load-bearing step reduces to a self-citation or ansatz imported from prior work by the same authors. The derivation is self-contained against the explicit polynomial systems.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption The reaction networks obey mass-action kinetics.
- domain assumption The four dual-site futile cycle topologies with bifunctional enzyme-substrate compounds are correctly specified.
Reference graph
Works this paper leans on
-
[1]
Robustness in glyoxylate bypass regulation.PLoS computational biology, 5(3):e1000297, 2009
Guy Shinar, Joshua D Rabinowitz, and Uri Alon. Robustness in glyoxylate bypass regulation.PLoS computational biology, 5(3):e1000297, 2009
work page 2009
-
[2]
Albert Goldbeter and Daniel E Koshland. An amplified sensitivity arising from covalent modification in biological systems.Proceedings of the National Academy of Sciences, 78(11):6840–6844, 1981
work page 1981
-
[3]
Structural sources of robustness in biochemical reaction networks
Guy Shinar and Martin Feinberg. Structural sources of robustness in biochemical reaction networks. Science, 327(5971):1389–1391, 2010
work page 2010
-
[4]
James E Ferrell and Rajani R Bhatt. Tripping the switch fantastic: how a protein kinase cascade can convert graded inputs into switch-like outputs.Trends in biochemical sciences, 22(8):288–289, 1997. 17
work page 1997
-
[5]
Martin Feinberg.Foundations of chemical reaction network theory, volume 10. Springer, 2019
work page 2019
-
[6]
Nikolai I Markevich, Jan B Hoek, and Boris N Kholodenko. Signaling switches and bistability arising from multisite phosphorylation in protein kinase cascades.Journal of Cell Biology, 164(3):353–359, 2004
work page 2004
-
[7]
Liming Wang and Eduardo D Sontag. On the number of steady states in a multiple futile cycle.Journal of Mathematical Biology, 57(1):29–52, 2008
work page 2008
-
[8]
Frank D Russo and Thomas J Silhavy. The essential tension: opposed reactions in bacterial two- component regulatory systems.Trends in microbiology, 1(8):306–310, 1993
work page 1993
-
[9]
Weihong Hsing, Frank D Russo, Karen K Bernd, and Thomas J Silhavy. Mutations that alter the kinase and phosphatase activities of the two-component sensor envz.Journal of bacteriology, 180(17):4538– 4546, 1998
work page 1998
-
[10]
Eric Batchelor and Mark Goulian. Robustness and the cycle of phosphorylation and dephosphorylation in a two-component regulatory system.Proceedings of the National Academy of Sciences, 100(2):691– 696, 2003
work page 2003
-
[11]
Joseph P Dexter and Jeremy Gunawardena. Dimerization and bifunctionality confer robustness to the isocitrate dehydrogenase regulatory system in escherichia coli.Journal of Biological Chemistry, 288(8):5770–5778, 2013
work page 2013
-
[12]
David F Anderson, Germ´ an A Enciso, and Matthew D Johnston. Stochastic analysis of biochemical reaction networks with absolute concentration robustness.Journal of The Royal Society Interface, 11(93):20130943, 2014
work page 2014
-
[13]
Badal Joshi and Gheorghe Craciun. Foundations of static and dynamic absolute concentration robust- ness.Journal of Mathematical Biology, 85(5):53, 2022
work page 2022
-
[14]
Badal Joshi and Gheorghe Craciun. Reaction network motifs for static and dynamic absolute concen- tration robustness.SIAM Journal on Applied Dynamical Systems, 22(2):501–526, 2023
work page 2023
-
[15]
Luis David Garc´ ıa Puente, Elizabeth Gross, Heather A Harrington, Matthew Johnston, Nicolette Meshkat, Mercedes P´ erez Mill´ an, and Anne Shiu. Absolute concentration robustness: Algebra and geometry.Journal of Symbolic Computation, 128:102398, 2025
work page 2025
-
[16]
Badal Joshi and Tung D Nguyen. Bifunctional enzyme action as a source of robustness in biochemical reaction networks: a novel hypergraph approach.Journal of The Royal Society Interface, 23(235), 2026
work page 2026
-
[17]
Pauline Van den Driessche and James Watmough. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission.Mathematical biosciences, 180(1-2):29–48, 2002
work page 2002
-
[18]
Further notes on the basic reproduction number
Pauline Van den Driessche and James Watmough. Further notes on the basic reproduction number. In Mathematical epidemiology, pages 159–178. Springer, 2008
work page 2008
-
[19]
Florin Avram, Rim Adenane, and Mircea Neagu. Advancing mathematical epidemiology and chemical reaction network theory via synergies between them.Entropy, 26(11):936, 2024
work page 2024
-
[20]
Florin Avram, Rim Adenane, Andrei D Halanay, and Matthew D Johnston. Stability in reaction network models via an extension of the next generation matrix method.arXiv preprint arXiv:2411.11867, 2024
-
[21]
Matthew Johnston and Florin Avram. The boundary reproduction number for determining boundary steady state stability in chemical reaction systems.Journal of Mathematical Biology, 92(2):29, 2026
work page 2026
-
[22]
Badal Joshi and Tung D Nguyen. Bifunctional enzyme provides absolute concentration robustness in multisite covalent modification networks.Journal of Mathematical Biology, 88(3):36, 2024. 18
work page 2024
-
[23]
Thapanar Suwanmajo and J Krishnan. Biphasic responses in multi-site phosphorylation systems.Jour- nal of The Royal Society Interface, 10(86):20130742, 2013
work page 2013
-
[24]
Abba B Gumel. Causes of backward bifurcations in some epidemiological models.Journal of Mathe- matical Analysis and Applications, 395(1):355–365, 2012
work page 2012
-
[25]
Steven H Strogatz.Nonlinear dynamics and chaos: with applications to physics, biology, chemistry, and engineering. Chapman and Hall/CRC, 2024
work page 2024
-
[26]
On the conditions under which an equation has only roots with negative real parts
Adolf Hurwitz et al. On the conditions under which an equation has only roots with negative real parts. Selected papers on mathematical trends in control theory, 65:273–284, 1964. Appendix This appendix contains detailed algebraic computations and proofs. Section A presents the full mass-action system, steady-state parameterization, proofs, and stability ...
work page 1964
-
[27]
As a result, there is a unique positive steady state in every compatibility class withT s > k 1/k∗ 2. To show the local stability of this unique positive steady state, we use the Routh-Hurwitz stability criterion [26]. Using the two conservation laws, we eliminate two specieseands 0. Using the parameterization in Proposition Theorem 3.1, the Jacobian matr...
-
[28]
Furthermore, since the living boundary steady state is locally asymptotically stable with respect to the intersection of the boundary face and compatibility class andρ(F V −1) =k ∗ 4s1/k1, it follows from Theorem 2.4 that it is stable ifs 1 < k 1/k∗ 4 and unstable ifs 1 > k 1/k∗ 4, wheres 1 is thes 1 entry of the living boundary steady state, and we are d...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.