Recognition: unknown
Modeling the impact of host diversity on the evolution of vector feeding preferences and implications for disease control
Pith reviewed 2026-05-07 10:16 UTC · model grok-4.3
The pith
Shortening the infectious period of the preferred host limits vector-borne disease persistence even as vectors adapt their feeding preferences.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
In the two-host model, protective actions on the vector's preferred host induce adaptive shifts in feeding preference that lower disease prevalence within that host; however, overall prevalence across both hosts can rise. Shortening the infectious period of the preferred host is shown to be an effective strategy for reducing disease persistence, and a threshold value governs whether a shift toward the non-preferred host amplifies or dampens the burden on the primary host.
What carries the argument
The coupled differential-equation model of transmission that incorporates adaptive rules for vector feeding preference, from which the basic reproduction number R0 and the preference-shift threshold are derived.
If this is right
- Shortening the infectious period of the preferred host reduces long-term disease persistence.
- A threshold value decides whether preference shifts toward the non-preferred host increase or decrease disease burden on the primary host.
- Protective measures on the preferred host produce preference shifts that cut prevalence there yet can raise combined prevalence in both hosts.
Where Pith is reading between the lines
- Interventions must track resulting preference changes, because ignoring adaptation can enlarge the total reservoir of infection.
- Similar dynamics in systems with more than two hosts could produce preference hierarchies that stabilize or expand disease maintenance.
- Empirical tests could compare observed preference shifts against the model's threshold after single-host interventions.
Load-bearing premise
Vector feeding preferences evolve according to specific adaptive rules under disease-control pressure, and the system contains exactly two hosts.
What would settle it
Field measurements of vector biting rates and host-specific prevalence before and after protective interventions on the preferred host, checking whether prevalence falls in the preferred host while total prevalence across hosts rises.
Figures







read the original abstract
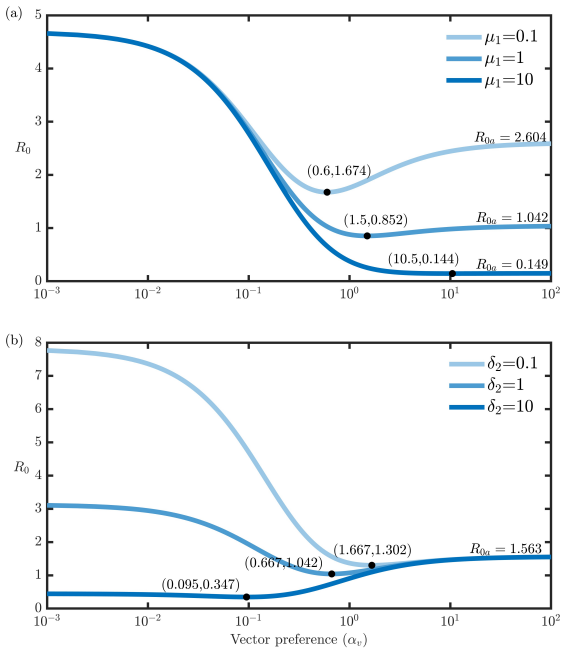
Vector-borne diseases often infect multiple host species, increasing the likelihood of disease persistence due to the presence of multiple reservoirs. Vector biting patterns and feeding preferences can shift in response to selective pressures introduced by disease control interventions, altering the dynamics of transmission. In this paper, we develop a mathematical model that couples host diversity and adaptive vector behavior with vector-borne disease transmission dynamics, focusing on a system with two hosts and a vector population exhibiting preference for one host. We derive the basic reproduction number, $R_0$, a threshold that determines the existence of two equilibria in our model, and obtain conditions that can lead to the long-term persistence of the disease. Our analysis suggests that shortening the infectious period of the vector's preferred host is an effective control strategy. We also identified a threshold that determines whether shifting vector preference toward a non-preferred host amplifies or reduces the disease burden on the primary preferred host. Our results show that protective measures for the preferred host can trigger adaptive shifts in vector preferences, reducing disease prevalence in that host. However, this shift may lead to an increase in overall host prevalence.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper develops a mathematical model for vector-borne disease transmission in a two-host system (one preferred) where vector feeding preferences evolve adaptively in response to selective pressures from disease control. It derives the basic reproduction number R0, identifies a threshold separating amplification versus reduction of disease burden on the preferred host under preference shifts, obtains conditions for disease persistence and equilibria, and concludes that shortening the infectious period of the preferred host is an effective control strategy while protective measures on the preferred host can reduce its prevalence but increase overall host prevalence.
Significance. If the central results hold under justified adaptive dynamics, the work would be significant for highlighting trade-offs in vector-borne disease control when vector behavior evolves, particularly the potential for preference shifts to offset interventions on preferred hosts. It contributes by coupling host diversity with evolutionary vector responses in an ODE framework, offering qualitative insights into persistence thresholds that could guide policy if validated against data.
major comments (2)
- [Model formulation] Model formulation section: the adaptive rules governing vector preference evolution are introduced as specific functional forms driven by selective pressure without derivation from first principles, empirical data on feeding behavior, or comparison to alternative kernels (e.g., frequency- vs. density-dependent updating). This is load-bearing because the sign of the identified preference-shift threshold and the net effect of shortening the preferred host's infectious period both depend on these rules; modest changes could reverse the amplification/reduction outcome or the control recommendation.
- [Results] Results on R0 and threshold: no sensitivity analysis is reported for the free parameters (preference adaptation rate, infectious period of preferred host) or for relaxing the strict two-host topology. The threshold separating amplification from reduction of burden on the primary host is obtained under the fixed model structure, so its robustness and the persistence conditions cannot be assessed without exploring alternative evolutionary dynamics or additional hosts.
minor comments (2)
- [Abstract] Abstract and introduction: equations for R0 and the preference threshold are referenced but not displayed, making it difficult to follow the derivation steps without immediately consulting the main text.
- [Discussion] Notation: the distinction between 'overall host prevalence' and prevalence in the preferred host should be clarified with explicit definitions or symbols when discussing the trade-off under preference shifts.
Simulated Author's Rebuttal
We thank the referee for their constructive comments on our manuscript. We address each major comment below and indicate the revisions that will be incorporated.
read point-by-point responses
-
Referee: [Model formulation] Model formulation section: the adaptive rules governing vector preference evolution are introduced as specific functional forms driven by selective pressure without derivation from first principles, empirical data on feeding behavior, or comparison to alternative kernels (e.g., frequency- vs. density-dependent updating). This is load-bearing because the sign of the identified preference-shift threshold and the net effect of shortening the preferred host's infectious period both depend on these rules; modest changes could reverse the amplification/reduction outcome or the control recommendation.
Authors: We agree that the specific functional forms for adaptive preference evolution are central to the results and that their justification merits expansion. These forms were chosen to represent selective pressure from control measures in a manner consistent with standard adaptive dynamics frameworks in evolutionary epidemiology. We did not derive them from first principles or empirical feeding data, nor compare them directly to alternatives such as frequency- versus density-dependent updating. In revision we will expand the model formulation section with additional justification, relevant citations on vector behavior, and a discussion of alternative kernels. We will also add a supplementary analysis using an alternative updating rule to confirm that the qualitative thresholds and control recommendations remain robust. A complete first-principles derivation would require new empirical studies outside the scope of this theoretical paper. revision: partial
-
Referee: [Results] Results on R0 and threshold: no sensitivity analysis is reported for the free parameters (preference adaptation rate, infectious period of preferred host) or for relaxing the strict two-host topology. The threshold separating amplification from reduction of burden on the primary host is obtained under the fixed model structure, so its robustness and the persistence conditions cannot be assessed without exploring alternative evolutionary dynamics or additional hosts.
Authors: The absence of sensitivity analysis is a valid observation. We will add a dedicated sensitivity analysis examining variation in the preference adaptation rate and the preferred host's infectious period, reporting effects on R0, the preference-shift threshold, and equilibrium prevalence. For the two-host assumption, we will extend the discussion section to outline how the core mechanisms extend to additional hosts while noting that quantitative thresholds would require case-specific parameterization. These additions will directly address robustness of the reported thresholds and persistence conditions. revision: yes
Circularity Check
No significant circularity; derivations are standard model analysis
full rationale
The paper constructs a coupled ODE system for two-host vector-borne transmission with an adaptive preference equation, then applies standard next-generation matrix methods to obtain R0 and bifurcation thresholds for equilibria and preference shifts. These quantities are explicit functions of the model parameters and rates; they do not reduce to the inputs by definition, nor are they obtained by fitting a subset of data and relabeling the fit as a prediction. No load-bearing self-citations or imported uniqueness theorems appear in the provided abstract or description. The adaptive rules are posited as modeling assumptions rather than derived results, so the subsequent threshold analysis remains a direct consequence of the stated equations rather than a tautology.
Axiom & Free-Parameter Ledger
free parameters (2)
- preference adaptation rate
- infectious period of preferred host
axioms (2)
- domain assumption Vector feeding preference evolves adaptively according to selective pressures from disease and control interventions
- standard math Disease persists or dies out based on a threshold R0 derived from the coupled transmission and preference dynamics
Reference graph
Works this paper leans on
-
[1]
Martinez, A
J. Martinez, A. Showering, C. Oke, R. T. Jones, J. G. Logan, Differential attraction in mosquito-human interactions and implications for disease control, Philosophical Transactions of the Royal Society B 376 (2021) 20190811
2021
-
[2]
N. M. Sene, B. Diouf, A. Gaye, A. Gueye, F. Seck, C. T. Diagne, I. Dia, D. Diallo, M. Diallo, et al., Blood feeding patterns of aedes aegypti populations in senegal, The American Journal of Tropical Medicine and Hygiene 106 (2022) 1402. 19
2022
-
[3]
J. Long, K. Maskell, R. Gries, S. Nayani, C. Gooding, G. Gries, Synergistic attraction of west- ern black-legged ticks, ixodes pacificus, to CO2 and odorant emissions from deer-associated microbes, Royal Society Open Science 10 (2023) 230084
2023
-
[4]
Ferraguti, A
M. Ferraguti, A. D. Martins, Y. Artzy-Randrup, Quantifying the invasion risk of west nile virus: Insights from a multi-vector and multi-host SEIR model, One Health 17 (2023) 100638
2023
-
[5]
M. M. de Wit, A. Dimas Martins, C. Delecroix, H. Heesterbeek, Q. A. Ten Bosch, Mech- anistic models for west nile virus transmission: a systematic review of features, aims and parametrization, Proceedings of the Royal Society B 291 (2024) 20232432
2024
-
[6]
Fiatsonu, A
E. Fiatsonu, A. Deka, M. L. Ndeffo-Mbah, Effectiveness of systemic insecticide dog treatment for the control of chagas disease in the tropics, Biology 12 (2023) 1235
2023
-
[7]
V. M. Schneider, J. E. Visone, C. T. Harris, F. Florini, E. Hadjimichael, X. Zhang, M. R. Gross, K. Y. Rhee, C. Ben Mamoun, B. F. Kafsack, et al., The human malaria parasite plasmodium falciparum can sense environmental changes and respond by antigenic switching, Proceedings of the National Academy of Sciences 120 (2023) e2302152120
2023
-
[8]
D. O. Escrivani, V. Scheidt, M. Tinti, J. Faria, D. Horn, Competition among variants is pre- dictable and contributes to the antigenic variation dynamics of african trypanosomes, PLOS Pathogens 19 (2023) 1–21
2023
-
[9]
Vanaerschot, S
M. Vanaerschot, S. Huijben, F. Van den Broeck, J.-C. Dujardin, Drug resistance in vector- borne parasites: multiple actors and scenarios for an evolutionary arms race, FEMS Microbi- ology Reviews 38 (2014) 41–55
2014
-
[10]
G. H. Palmer, T. Bankhead, H. S. Seifert, Antigenic variation in bacterial pathogens, in: Virulence Mechanisms of Bacterial Pathogens, John Wiley & Sons, Ltd, 2016, pp. 445–480
2016
-
[11]
Ross, The prevention of malaria, John Murray, 1911
R. Ross, The prevention of malaria, John Murray, 1911
1911
-
[12]
Macdonald, Epidemiological basis of malaria control, Bulletin of the World Health Orga- nization 15 (1956) 613–626
G. Macdonald, Epidemiological basis of malaria control, Bulletin of the World Health Orga- nization 15 (1956) 613–626
1956
-
[13]
Chitnis, J
N. Chitnis, J. M. Hyman, J. M. Cushing, Determining important parameters in the spread of malaria through the sensitivity analysis of a mathematical model, Bulletin of Mathematical Biology 70 (2008) 1272–1296
2008
-
[14]
Chamchod, N
F. Chamchod, N. F. Britton, Analysis of a vector-bias model on malaria transmission, Bulletin of Mathematical Biology 73 (2011) 639–657
2011
-
[15]
K. S. Rock, D. A. Wood, M. J. Keeling, Age-and bite-structured models for vector-borne diseases, Epidemics 12 (2015) 20–29
2015
-
[16]
M. N. Anwar, L. Smith, A. Devine, S. Mehra, C. R. Walker, E. Ivory, E. Conway, I. Mueller, J. M. McCaw, J. A. Flegg, et al., Mathematical models of plasmodium vivax transmission: A scoping review, PLOS Computational Biology 20 (3) (2024) e1011931
2024
-
[17]
S. L. Robertson, K. A. Caillouët, A host stage-structured model of enzootic west nile virus transmission to explore the effect of avian stage-dependent exposure to vectors, Journal of theoretical biology 399 (2016) 33–42. 20
2016
-
[18]
D. M. Bichara, Global analysis of multi-host and multi-vector epidemic models, Journal of Mathematical Analysis and Applications 475 (2019) 1532–1553
2019
-
[19]
M. H. Zahid, C. M. Kribs, Decoys and dilution: the impact of incompetent hosts on prevalence of chagas disease, Bulletin of Mathematical Biology 82 (2020) 41
2020
-
[20]
Yakob, M
L. Yakob, M. B. Bonsall, G. Yan, Modelling knowlesi malaria transmission in humans: vector preference and host competence, Malaria Journal 9 (2010) 1–7
2010
-
[21]
J. E. Simpson, P. J. Hurtado, J. Medlock, G. Molaei, T. G. Andreadis, A. P. Galvani, M. A. Diuk-Wasser, Vector host-feeding preferences drive transmission of multi-host pathogens: West nile virus as a model system, Proceedings of the Royal Society B: Biological Sciences 279 (2012) 925–933
2012
-
[22]
R. C. Rivera, S. Bilal, E. Michael, The relation between host competence and vector-feeding preference in a multi-host model: Chagas and cutaneous leishmaniasis, Mathematical Bio- sciences and Engineering 17 (2020) 5561–5583
2020
-
[23]
Bilal, R
S. Bilal, R. Caja Rivera, A. Mubayi, E. Michael, Complexity and critical thresholds in the dynamics of visceral leishmaniasis, Royal Society Open Science 7 (12) (2020) 200904
2020
-
[24]
L. Chen, S. Chen, P. Kong, L. Zhou, Host competence, interspecific competition and vector preference interact to determine the vector-borne infection ecology, Frontiers in Ecology and Evolution 10 (2022) 993844
2022
-
[25]
S. L. Wu, J. M. Henry, D. T. Citron, D. Mbabazi Ssebuliba, J. Nakakawa Nsumba, H. M. Sánchez C, O. J. Brady, C. A. Guerra, G. A. García, A. R. Carter, et al., Spatial dynamics of malaria transmission, PLOS Computational Biology 19 (6) (2023) e1010684
2023
-
[26]
Miller, A
E. Miller, A. Huppert, The effects of host diversity on vector-borne disease: The conditions under which diversity will amplify or dilute the disease risk, PLoS One 8 (2013) e80279
2013
-
[27]
Marini, R
G. Marini, R. Rosá, A. Pugliese, H. Heesterbeek, Exploring vector-borne infection ecology in multi-host communities: A case study of west nile virus, Journal of Theoretical Biology 415 (2017) 58–69
2017
-
[28]
S. A. Geritz, J. A. Metz, É. Kisdi, G. Meszéna, Dynamics of adaptation and evolutionary branching, Physical Review Letters 78 (1997) 2024
1997
-
[29]
Diekmann, J
O. Diekmann, J. Heesterbeek, M. G. Roberts, The construction of next-generation matrices for compartmental epidemic models, Journal of the Royal Society Interface 7 (2010) 873–885
2010
-
[30]
Wiggins, Introduction to applied nonlinear dynamical systems and chaos, Springer, 1990
S. Wiggins, Introduction to applied nonlinear dynamical systems and chaos, Springer, 1990
1990
-
[31]
Stone, K
C. Stone, K. Gross, Evolution of host preference in anthropophilic mosquitoes, Malaria journal 17 (2018) 1–11
2018
-
[32]
Ravigné, U
V. Ravigné, U. Dieckmann, I. Olivieri, Live where you thrive: joint evolution of habitat choice and local adaptation facilitates specialization and promotes diversity, The American Naturalist 174 (2009) E141–E169. 21
2009
-
[33]
Le Menach, S
A. Le Menach, S. Takala, F. E. McKenzie, A. Perisse, A. Harris, A. Flahault, D. L. Smith, An elaborated feeding cycle model for reductions in vectorial capacity of night-biting mosquitoes by insecticide-treated nets, Malaria journal 6 (2007) 1–12
2007
-
[34]
Gandy, E
S. Gandy, E. Kilbride, R. Biek, C. Millins, L. Gilbert, No net effect of host density on tick- borne disease hazard due to opposing roles of vector amplification and pathogen dilution, Ecology and Evolution 12 (2022) e9253
2022
-
[35]
Keesing, R
F. Keesing, R. D. Holt, R. S. Ostfeld, Effects of species diversity on disease risk, Ecology Letters 9 (2006) 485–498
2006
-
[36]
E. E. Kemibala, A. Mafra-Neto, T. Dekker, J. Saroli, R. Silva, A. Philbert, K. Nghabi, L. E. Mboera, A zooprophylaxis strategy using l-lactic acid (abate) to divert host-seeking malaria vectors from human host to treated non-host animals, Malaria Journal 19 (2020) 1–7
2020
-
[37]
Asale, L
A. Asale, L. Duchateau, B. Devleesschauwer, G. Huisman, D. Yewhalaw, Zooprophylaxis as a control strategy for malaria caused by the vector anopheles arabiensis (diptera: Culicidae): a systematic review, Infectious Diseases of Poverty 6 (2017) 1–14
2017
-
[38]
Keesing, R
F. Keesing, R. S. Ostfeld, Dilution effects in disease ecology, Ecology Letters 24 (2021) 2490– 2505
2021
-
[39]
R. S. Levine, D. L. Hedeen, M. W. Hedeen, G. L. Hamer, D. G. Mead, U. D. Kitron, Avian species diversity and transmission of west nile virus in atlanta, georgia, Parasites & Vectors 10 (2017) 1–12
2017
-
[40]
Y. A. Mwalugelo, W. P. Mponzi, L. L. Muyaga, H. H. Mahenge, G. C. Katusi, F. Muhonja, D. Omondi, A. O. Ochieng, E. W. Kaindoa, F. A. Amimo, Livestock keeping, mosquitoes and community viewpoints: a mixed methods assessment of relationships between livestock management, malariavectorbitingriskandcommunityperspectivesinruraltanzania, Malaria Journal 23 (1) ...
2024
-
[41]
+d3)Y 2 3 −w 1(d1 +δ 1)Z 2 1 . 23 Setw 1 = αvc1y∗ 3 µ1 , then we can derive that˙Wis negative definite if the following are true: 2α2 vc2 1(1−y ∗ 1 −z ∗ 1)2 d3(µ1 +d 1) < w 3 < d3(µ1 +d 1) 2c2 2α2 vN 2 h1(1−y ∗ 3)2 ,(B.1) w32c2 2N 2 h2(1−y ∗ 3)2 (δ2 +d 2)d3 < w 2 < (δ2 +d 2)d3w3 2c2 1(1−y ∗ 2)2 .(B.2) From here, it follows that a positivew3 can be chosen ...
-
[42]
+u3c2αvNh1(1−y ∗ 3)(y1 −y ∗ 1)(y3 −y ∗ 3) +u 2c1(1−y ∗ 2)(y3 −y ∗ 3)(y2 −y ∗
-
[43]
+u3c2Nh2(1−y ∗ 3)(y3 −y ∗ 3)(y2 −y ∗ 2)−(µ 1 +d 1)(y1 −y ∗ 1)2 −u 2(d2 +δ 2)(y2 −y ∗ 2)2 −u 3d3(y3 −y ∗ 3)2 −α vc1y3(y1 −y ∗ 1)2 −u 1(d1 +δ 1)(z1 −z ∗ 1)2 −u 2c1y3(y2 −y ∗ 2)2 −u 3c2αvNh1y1(y3 −y ∗ 3)2 −u 3c2Nh2y2(y3 −y ∗ 3)2. By selectingu 1 asu 1 = αvc1y∗ 3 µ1 , ˙Ubecomes negative definite if the following conditions hold true: α2 vc2 1(1−y ∗ 1 −z 1max)...
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.