Recognition: no theorem link
Glycemic Safety Tube: A Provably Safe Control Framework for Artificial Pancreas Systems under Parametric Uncertainty
Pith reviewed 2026-05-12 04:53 UTC · model grok-4.3
The pith
The Glycemic Safety Tube Control method keeps glucose levels inside safe bounds by design for artificial pancreas systems despite model uncertainty.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
The paper claims that by using a safety tube around the desired glucose trajectory, the controller can be designed such that the closed-loop glucose level is guaranteed to remain inside the tube for all time, provided the disturbances are bounded. This is achieved in a model-free manner, and explicit feasibility conditions are given for the tube parameters to also satisfy the insulin input bounds.
What carries the argument
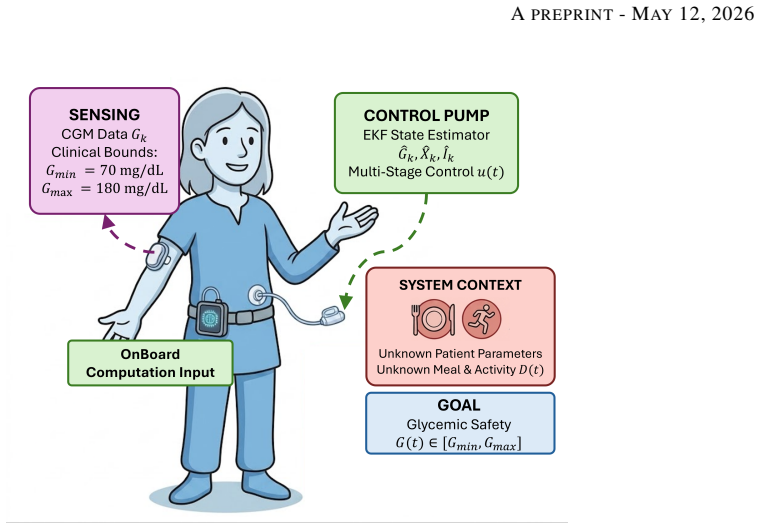
Glycemic Safety Tube: a prescribed time-varying safe envelope for glucose concentration that the control law is constructed to enforce.
Load-bearing premise
Meal disturbances and estimation errors stay inside the specific bounded sets used to prove the feasibility conditions.
What would settle it
An experiment or simulation in which glucose concentration leaves the prescribed safe range after a meal disturbance larger than the assumed bound is applied to the system under GSTC.
Figures




read the original abstract
Type 1 diabetes eliminates the body's ability to produce insulin, making glucose regulation entirely dependent on external insulin delivery and the control algorithm. Existing closed-loop methods either rely on accurate patient-specific models or do not provide formal safety guarantees, and are often computationally demanding for wearable devices. This paper proposes Glycemic Safety Tube Control (GSTC), a model-free and computationally efficient control framework for automated insulin delivery. The method enforces clinically relevant safety bounds on glucose levels by design, ensuring that glucose remains within a prescribed safe range. We also derive feasibility conditions that guarantee safety and input constraint satisfaction under bounded meal disturbances and estimation errors. The performance of GSTC is evaluated against state-of-the-art methods, including linear and nonlinear model predictive control and sliding mode control. The results demonstrate that GSTC maintains safety under varying meal patterns and patient conditions, highlighting its robustness and computational efficiency. Overall, GSTC provides a safe, efficient, and patient-independent approach for next-generation artificial pancreas systems.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper proposes Glycemic Safety Tube Control (GSTC), a model-free and computationally efficient framework for artificial pancreas systems in type 1 diabetes. It claims to enforce clinically relevant safety bounds on glucose levels by design, derive feasibility conditions that guarantee safety and input constraint satisfaction under bounded meal disturbances and estimation errors, and demonstrate robustness and efficiency in simulations against linear/nonlinear MPC and sliding mode control under varying meal patterns and patient conditions.
Significance. If the feasibility conditions and safety guarantees hold under realistic disturbance bounds, GSTC would provide a notable advance by offering a patient-independent, provably safe, and lightweight control method for automated insulin delivery. This addresses key limitations of existing approaches that either lack formal safety certificates or require accurate patient-specific models, with potential benefits for wearable device implementation and clinical translation in glycemic management.
major comments (1)
- Abstract: The central claim that GSTC 'derives feasibility conditions that guarantee safety and input constraint satisfaction under bounded meal disturbances and estimation errors' is load-bearing for the contribution, yet these conditions are conditional on a priori bounds whose selection, justification, or validation against clinical data distributions, worst-case patient variability, or sensor noise statistics is not addressed. Without such grounding, the provable safety reduces to a conditional guarantee whose practical relevance cannot be assessed.
Simulated Author's Rebuttal
We thank the referee for the constructive feedback on our manuscript. We address the major comment point by point below, proposing revisions where appropriate to clarify the practical grounding of our results.
read point-by-point responses
-
Referee: Abstract: The central claim that GSTC 'derives feasibility conditions that guarantee safety and input constraint satisfaction under bounded meal disturbances and estimation errors' is load-bearing for the contribution, yet these conditions are conditional on a priori bounds whose selection, justification, or validation against clinical data distributions, worst-case patient variability, or sensor noise statistics is not addressed. Without such grounding, the provable safety reduces to a conditional guarantee whose practical relevance cannot be assessed.
Authors: We agree that the feasibility conditions are conditional on user-specified bounds on disturbances and estimation errors, which is inherent to any robust control result providing formal guarantees under bounded uncertainty. The original manuscript derives these conditions rigorously but assumes the bounds are provided a priori, without extensive discussion of their selection. In the revised manuscript we will add a dedicated subsection (likely in Section III or IV) that provides guidance on choosing realistic bounds. This will include references to clinical literature on typical meal carbohydrate distributions in T1D patients, CGM sensor accuracy statistics (e.g., MARD values), and conservative worst-case variability estimates drawn from published studies. While a full empirical validation against large-scale patient datasets would require clinical trials outside the scope of this theoretical paper, the added discussion will explicitly link the bounds to available clinical data and explain how conservative choices preserve the safety guarantees. This revision will make the practical relevance of the conditional guarantees clearer without altering the core theoretical contributions. revision: yes
Circularity Check
No circularity: safety tube and feasibility conditions derived from external bounds
full rationale
The GSTC framework claims to enforce glucose safety bounds by design and to derive feasibility conditions that guarantee safety under a priori bounded meal disturbances and estimation errors. These bounds are treated as given external inputs to the derivation rather than quantities fitted or defined from the controller outputs themselves. No equations or steps in the provided text reduce the safety guarantees to self-referential definitions, fitted parameters renamed as predictions, or load-bearing self-citations. The derivation chain therefore remains logically independent of its target results and is self-contained against the stated assumptions.
Axiom & Free-Parameter Ledger
axioms (2)
- domain assumption Meal disturbances and estimation errors lie inside known compact sets
- standard math Standard Lyapunov or set-invariance arguments from control theory
Reference graph
Works this paper leans on
-
[1]
Mark A. Atkinson and George S. Eisenbarth. Type 1 diabetes: new perspectives on disease pathogenesis and treatment.The Lancet, 358(9277):221–229, 2001
work page 2001
-
[2]
International Diabetes Federation, Brussels, Belgium, 2024
International Diabetes Federation.IDF Diabetes Atlas, 11th Edition. International Diabetes Federation, Brussels, Belgium, 2024
work page 2024
-
[3]
Mark Peyrot et al. Insulin adherence behaviours and barriers in the multinational Global Attitudes of Patients and Physicians in Insulin Therapy study.Diabetic Medicine, 29:682–689, 2012
work page 2012
-
[4]
Diabetes Control and Complications Trial Research Group, David M. Nathan, Saul Genuth, John Lachin, Patricia Cleary, Obie Crofford, Michael Davis, Larry Rand, and Charles Siebert. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The New England Journal of Me...
work page 1993
-
[5]
Frances Cameron, B. Wayne Bequette, Darrell M. Wilson, Bruce A. Buckingham, Hyojin Lee, and Greg A. Niemeyer. Closed-loop artificial pancreas based on risk management.Journal of Diabetes Science and Technology, 5(2):368–379, 2011
work page 2011
-
[6]
Francis J. Doyle and Boris P. Kovatchev. Artificial pancreas: The first 20 years.Journal of Diabetes Science and Technology, 19(4):875–882, 2025
work page 2025
-
[7]
Tadej Battelino, Thomas Danne, Richard M. Bergenstal, Stephanie A. Amiel, Roy Beck, Torben Biester, Emanuele Bosi, Bruce A. Buckingham, William T. Cefalu, Kelly L. Close, Claudio Cobelli, Eyal Dassau, J. Hans DeVries, Kim C. Donaghue, Klemen Dovc, Francis J. Doyle, Satish Garg, George Grunberger, Richard Holt, Irl B. Hirsch, Nathalie Jeandidier, David Klo...
work page 2019
-
[8]
Annie Zhong, Pratik Choudhary, Claire McMahon, et al. Effectiveness of automated insulin management features of the MiniMed 640G sensor-augmented insulin pump.Diabetes Technology & Therapeutics, 18(10):657–663, 2016. 15 APREPRINT- MAY12, 2026
work page 2016
-
[9]
Pinsker, Samantha Leas, Lars Müller, and Shira Habif
Jordan E. Pinsker, Samantha Leas, Lars Müller, and Shira Habif. Real-world improvements in hypoglycemia in an insulin-dependent cohort with diabetes mellitus pre/post Tandem Basal-IQ technology remote software update. Endocrine Practice, 26(7):715–721, 2020
work page 2020
-
[10]
Emily G. Considine and Jennifer L. Sherr. Real-world evidence of automated insulin delivery system use.Diabetes Technology & Therapeutics, 26(S3):53–65, 2024
work page 2024
-
[11]
Steven J. Russell, Firas H. El-Khatib, Manasi Sinha, Katherine L. Magyar, Kendra McKeon, Laura G. Goergen, Courtney Balliro, Mallory A. Hillard, David M. Nathan, and Edward R. Damiano. Outpatient glycemic control with a bionic pancreas in type 1 diabetes.New England Journal of Medicine, 371(4):313–325, 2014
work page 2014
-
[12]
Boris P. Kovatchev, Daniel J. Cox, Linda A. Gonder-Frederick, and William Clarke. Symmetrization of the blood glucose measurement scale and its applications.Diabetes Care, 20(11):1655–1658, 1997
work page 1997
-
[13]
Chassin, Urs Haueter, Massimo Massi-Benedetti, Marina Orsini Federici, Thomas R
Roman Hovorka, Valeria Canonico, Ludovic J. Chassin, Urs Haueter, Massimo Massi-Benedetti, Marina Orsini Federici, Thomas R. Pieber, Hermann C. Schaller, Ludwig Schaupp, Thomas Vering, and Malgorzata E. Wil- inska. Nonlinear model predictive control of glucose concentration in subjects with type 1 diabetes.Physiological Measurement, 25(4):905–920, 2004
work page 2004
-
[14]
Garry M. Steil, Athanasios E. Panteleon, and Konstantin Rebrin. Closed-loop insulin delivery: The path to physiological glucose control.Advanced Drug Delivery Reviews, 56(2):125–144, 2004
work page 2004
-
[15]
Raimondo, Laura Bossi, Chiara D
Lalo Magni, Davide M. Raimondo, Laura Bossi, Chiara D. Man, Giancarlo De Nicolao, Boris Kovatchev, and Claudio Cobelli. Model predictive control of type 1 diabetes: An in silico trial.Journal of Diabetes Science and Technology, 1(6):804–812, 2007
work page 2007
-
[16]
Sahaj Saxena and Navin Kumar. Safe glucose regulation in artificial pancreas systems via robust control barrier functions.Transactions of the Institute of Measurement and Control, page 01423312251405018, 2025
work page 2025
-
[17]
M. E. Wilinska, R. Hovorka, et al. Simulation models for in silico testing of closed-loop glucose controllers in type 1 diabetes.Diabetes Technology & Therapeutics, 12(4):269–277, 2010
work page 2010
-
[18]
Anirudh Nath, Rajeeb Dey, and Carlos Aguilar-Avelar. Observer based nonlinear control design for glucose regulation in type 1 diabetic patients: An lmi approach.Biomedical Signal Processing and Control, 47:7–15, 2019
work page 2019
-
[19]
Boubekeur Targui, Juan-Francisco Castro-Gomez, Omar Hernández-González, Guillermo Valencia-Palomo, and María-Eusebia Guerrero-Sánchez. Observer-based control for plasma glucose regulation in type 1 diabetes mellitus patients with unknown input delay.International Journal for Numerical Methods in Biomedical Engineering, 40:e3826, 2024
work page 2024
- [20]
-
[21]
Elena Daskalaki, Athina Prountzou, Peter Diem, and Stavroula G Mougiakakou. Efficient adaptive control of the artificial pancreas using actor-critic learning.IEEE Transactions on Biomedical Engineering, 60(12):3300–3308, 2013
work page 2013
-
[22]
Deep reinforcement learning for closed-loop blood glucose control
Ian Fox, Jinsung Lee, Rodica Pop-Busui, and Jenna Wiens. Deep reinforcement learning for closed-loop blood glucose control. InProceedings of the Machine Learning for Healthcare Conference, pages 508–536. PMLR, 2020
work page 2020
-
[23]
Charalampos P Bechlioulis and George A Rovithakis. A low-complexity global approximation-free control scheme with prescribed performance for unknown pure feedback systems.Automatica, 50(4):1217–1226, 2014
work page 2014
-
[24]
Ratnangshu Das, Ahan Basu, and Pushpak Jagtap. Spatiotemporal tubes for temporal reach-avoid-stay tasks in unknown systems.IEEE Transactions on Automatic Control, 71(1):512–519, 2026
work page 2026
-
[25]
Ratnangshu Das and Pushpak Jagtap. Prescribed-time reach-avoid-stay specifications for unknown systems: A spatiotemporal tubes approach.IEEE Control Systems Letters, 8:946–951, 2024
work page 2024
-
[26]
Ratnangshu Das, Siddhartha Upadhyay, and Pushpak Jagtap. Real-time spatiotemporal tubes for dynamic unsafe sets.IEEE Robotics and Automation Letters, 11(2):2146–2153, 2025
work page 2025
-
[27]
Ratnangshu Das and Pushpak Jagtap. Approximation-free control of unknown euler-lagrangian systems under input constraints.arXiv preprint arXiv:2507.01426, 2025
-
[28]
Richard N. Bergman, Lynne S. Phillips, and Claudio Cobelli. Physiologic evaluation of factors controlling glucose tolerance in man: Measurement of insulin sensitivity and beta-cell glucose sensitivity from the response to intravenous glucose.The Journal of Clinical Investigation, 68(6):1456–1467, 1981. 16 APREPRINT- MAY12, 2026
work page 1981
-
[29]
M.E. Fisher. A semiclosed-loop algorithm for the control of blood glucose levels in diabetics.IEEE Transactions on Biomedical Engineering, 38(1):57–61, 1991
work page 1991
-
[30]
Stuart M Furler, M Biomed E, Edward W Kraegen, Robert H Smallwood, and Donald J Chisholm. Blood glucose control by intermittent loop closure in the basal mode: Computer simulation studies with a diabetic model. Diabetes Care, 8(6):553–561, 11 1985
work page 1985
-
[31]
Adaptive control in an artificial pancreas for people with type 1 diabetes
Dimitri Boiroux, Anne Katrine Duun-Henriksen, Signe Schmidt, Kirsten Nørgaard, Niels Kjølstad Poulsen, Henrik Madsen, and John Bagterp Jørgensen. Adaptive control in an artificial pancreas for people with type 1 diabetes. Control Engineering Practice, 58:332–342, 2017
work page 2017
-
[32]
Parker, III Doyle, Francis J., and Nicholas A
Robert S. Parker, III Doyle, Francis J., and Nicholas A. Peppas. A model-based algorithm for blood glucose control in type I diabetic patients.IEEE Transactions on Biomedical Engineering, 46(2):148–157, 1999
work page 1999
-
[33]
Dan Simon.Optimal State Estimation: Kalman, H Infinity, and Nonlinear Approaches. Wiley-Interscience, USA, 2006
work page 2006
-
[34]
Castañeda, Gema García-Sáez, M
Onofre Orozco-López, Carlos E. Castañeda, Gema García-Sáez, M. Elena Hernando, and Agustín Rodríguez- Herrero. Hybrid sliding mode control with input window constraints for exercise-aware glucose management in type 1 diabetes.Control Engineering Practice, 172:106931, 2026
work page 2026
-
[35]
P. Soru, G. De Nicolao, C. Toffanin, C. Dalla Man, C. Cobelli, and L. Magni. Mpc based artificial pancreas: Strategies for individualization and meal compensation.Annual Reviews in Control, 36(1):118–128, 2012
work page 2012
-
[36]
Jinyu Xie. simglucose v0.2.1. https://github.com/jxx123/simglucose, 2018. Accessed: MARCH-04- 2026
work page 2018
-
[37]
Chiara Dalla Man, Filippo Micheletto, Dayu Lv, Marc Breton, Boris Kovatchev, and Claudio Cobelli. The UV A/PADOV A type 1 diabetes simulator: New features.Journal of Diabetes Science and Technology, 8(1):26–34, 2014. A Appendix 0-meal → 3-meal CV% Dot size = BGRI CV = 36% threshold 10% 20% 30% 40% 36% P1 P2 P3 P4 P5 P6 Mean C ffi i t f i ti (%) +12.8pp +1...
work page 2014
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.