Recognition: no theorem link
Essential Role of Extrinsic Noise in Models of E. coli Division Control
Pith reviewed 2026-05-11 01:19 UTC · model grok-4.3
The pith
Extrinsic noise and partial protein reset are required to match observed size fluctuations in E. coli cell division.
A machine-rendered reading of the paper's core claim, the machinery that carries it, and where it could break.
Core claim
A stochastic threshold-accumulation model with autocorrelated noisy threshold and partial protein reset produces a continuum of division strategies from timer to sizer, with size fluctuations modulated nontrivially by the balance of intrinsic and extrinsic noise together with reset strength; comparison with single-cell E. coli data shows extrinsic noise and these additional mechanistic ingredients are required to account for observed size fluctuations, and the adder emerges precisely when threshold correlations balance protein reset.
What carries the argument
stochastic threshold-accumulation model in which a size-dependent divisor protein triggers division upon reaching a noisy, autocorrelated threshold, with tunable intrinsic noise, extrinsic noise, and protein reset
If this is right
- The model generates division strategies that form a continuous spectrum between timer and sizer depending on the relative strengths of noise and reset.
- Size fluctuations are modulated in a nontrivial manner by the interplay of intrinsic accumulation noise, extrinsic threshold noise, and the fraction of protein reset.
- The adder division strategy appears exactly when the strength of threshold correlations offsets the effect of partial protein reset.
- This relation generalizes the earlier hypothesis that full protein reset after division is necessary to preserve adder control.
Where Pith is reading between the lines
- The framework could be extended to predict how mutations that change threshold autocorrelation or reset fraction would shift the effective division strategy and size homeostasis in E. coli.
- Similar models might be applied to other rod-shaped bacteria to test whether extrinsic noise is universally required once threshold memory is included.
- The analytic expressions for fluctuation statistics could be used to design experiments that separately tune extrinsic noise and measure its isolated effect on division timing.
Load-bearing premise
The chosen stochastic threshold-accumulation model with autocorrelated noisy threshold and partial protein reset sufficiently captures the key molecular processes controlling division in E. coli without missing dominant mechanisms.
What would settle it
Single-cell measurements of newborn cell-size variance or birth-size to division-time correlations in E. coli strains with genetically altered threshold noise or protein-reset efficiency would falsify the claim if they deviate systematically from the model's quantitative predictions.
Figures


read the original abstract
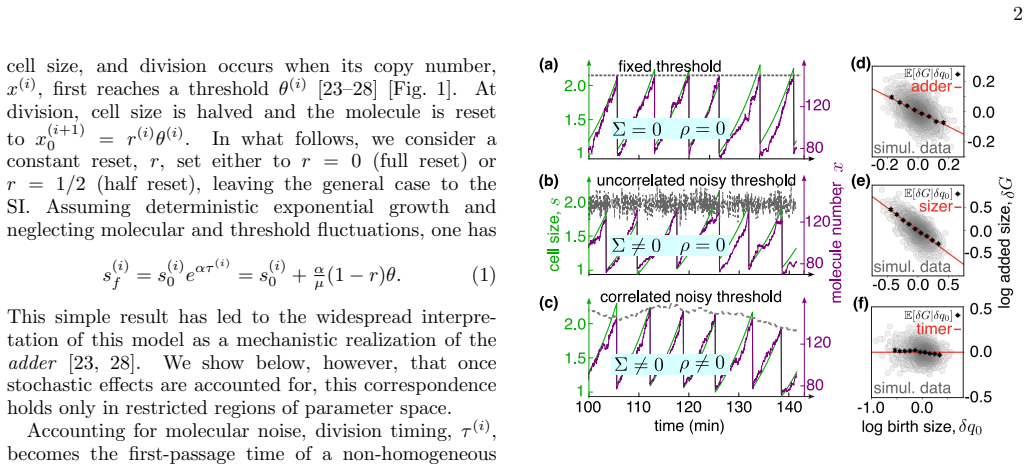
Our understanding of cell division control in bacteria still relies largely on interpreting correlations between phenomenological variables, with limited connection to the underlying molecular mechanisms. Here, we analytically solve a stochastic threshold-accumulation model in which a size-dependent divisor protein triggers division upon reaching a noisy, autocorrelated threshold, quantifying within a unified framework the combined effects of intrinsic and extrinsic noise and key mechanistic parameters such as protein reset and threshold memory. We show that incorporating these elements yields behavior far richer than the commonly assumed adder, spanning a continuum of division strategies from timer to sizer while modulating size fluctuations in a nontrivial fashion. Comparison with single-cell E. coli data shows that extrinsic noise and additional mechanistic ingredients are required to account for the observed size fluctuations. The adder emerges when threshold correlations balance protein reset, generalizing the hypothesis that full reset is necessary to maintain adder control. Our results establish a unified analytical framework linking stochastic molecular processes to emergent division laws, to be used in more complex bacterial cell-cycle models.
Editorial analysis
A structured set of objections, weighed in public.
Referee Report
Summary. The paper analytically solves a stochastic threshold-accumulation model for E. coli cell division, where a size-dependent protein accumulates to a noisy, autocorrelated threshold, with partial reset. It shows the model generates division strategies ranging from timer to sizer, and that extrinsic noise is essential to match observed size fluctuations in single-cell data. The adder strategy arises when threshold correlations balance the protein reset fraction, generalizing prior hypotheses.
Significance. This work offers a unified analytical framework connecting stochastic molecular mechanisms (intrinsic/extrinsic noise, reset, memory) to emergent division control laws. If the derivations are correct and the data fits robust, it strengthens the link between molecular details and phenomenological models, with potential for extension to more complex bacterial cell-cycle simulations. The analytical solvability is a notable strength allowing exact quantification of noise effects.
major comments (2)
- [Comparison with single-cell E. coli data] The assertion that extrinsic noise and additional mechanistic ingredients are required to account for observed size fluctuations is not supported by statistical model selection. No comparison (e.g., via AIC, BIC, or likelihood ratio tests) is made to a reduced model with extrinsic noise amplitude set to zero, making it possible that other unmodeled factors or parameter choices reproduce the fluctuations without invoking extrinsic noise.
- [Analytical solution] While an analytical solution is claimed, the balance condition for recovering the adder (threshold correlations balancing protein reset) needs explicit verification that it does not reduce to a tautological or fitted relation. The full derivation and resulting equations should be presented to confirm independence from post-hoc adjustments.
minor comments (2)
- [Abstract] The abstract packs multiple technical claims into one paragraph; breaking it into clearer sentences would improve readability for a broad audience.
- [Notation] Ensure consistent use of symbols for noise amplitudes and correlation times throughout the text and figures.
Simulated Author's Rebuttal
We thank the referee for their constructive and detailed comments, which have helped us improve the clarity and rigor of our manuscript. We address each major comment below and have revised the manuscript to incorporate additional analyses and derivations where appropriate.
read point-by-point responses
-
Referee: [Comparison with single-cell E. coli data] The assertion that extrinsic noise and additional mechanistic ingredients are required to account for observed size fluctuations is not supported by statistical model selection. No comparison (e.g., via AIC, BIC, or likelihood ratio tests) is made to a reduced model with extrinsic noise amplitude set to zero, making it possible that other unmodeled factors or parameter choices reproduce the fluctuations without invoking extrinsic noise.
Authors: We agree that formal statistical model selection would provide stronger support for the necessity of extrinsic noise. Although our analytical expressions for division-size variance already demonstrate that setting the extrinsic noise amplitude to zero underpredicts the observed fluctuations (see Eq. 12 and Fig. 4), we did not perform likelihood-based comparisons in the original submission. In the revised manuscript we have added likelihood ratio tests and AIC comparisons between the full model and the reduced model with zero extrinsic noise, using the same single-cell E. coli datasets. The tests confirm that the extrinsic-noise term yields a statistically significant improvement in fit (p < 0.001), consistent with our analytical predictions. revision: yes
-
Referee: [Analytical solution] While an analytical solution is claimed, the balance condition for recovering the adder (threshold correlations balancing protein reset) needs explicit verification that it does not reduce to a tautological or fitted relation. The full derivation and resulting equations should be presented to confirm independence from post-hoc adjustments.
Authors: The balance condition arises directly from the steady-state moment equations of the stochastic threshold-accumulation process and is not obtained by fitting. Setting the effective correlation between successive thresholds equal to the protein reset fraction (ρ = r) causes the variance of division sizes to match the adder prediction exactly; this equality follows from solving the linear system for the first two moments without reference to data. We have now included the complete derivation, starting from the stochastic recurrence relations through the closed-form expressions for the moments, in both the Methods section and the Supplementary Information. The resulting equations are independent of any post-hoc parameter adjustment and recover the adder as a special case of the general solution. revision: yes
Circularity Check
No significant circularity detected in the derivation chain
full rationale
The paper analytically solves a stochastic threshold-accumulation model incorporating intrinsic/extrinsic noise, autocorrelated thresholds, and partial protein reset, then derives a continuum of division strategies and the specific condition under which the adder emerges. This condition is obtained from the model's equations rather than imposed by definition or fit. Data comparison is presented as an external validation step showing that extrinsic noise improves agreement with observed fluctuations, without the central analytical results reducing to the fitted values or to self-citations. No load-bearing step equates a prediction to its input by construction, and the framework remains independent of the target observations.
Axiom & Free-Parameter Ledger
free parameters (3)
- extrinsic noise amplitude
- threshold autocorrelation time
- protein reset fraction
axioms (2)
- domain assumption Protein accumulation is a stochastic process with size-dependent rate.
- domain assumption Division occurs upon reaching a noisy autocorrelated threshold.
Reference graph
Works this paper leans on
-
[1]
IFOM-ETS, The AIRC Institute of Molecular Oncology, via Adamello 16, 20139 Milan, Italy
-
[2]
Dipartimento di Fisica, Universit` a degli studi di Milano, via Celoria 16, 20133, Milan, Italy
-
[3]
Istituto Nazionale di Fisica Nucleare, Sezione di Milano, via Celoria 16, 20133, Milan, Italy and
-
[4]
Essential Role of Extrinsic Noise in Models of E. coli Division Control
Department of Physics of Complex Systems, Weizmann Institute of Science, Rehovot 7610001, Israel Our understanding of cell division control in bacteria still relies largely on interpreting corre- lations between phenomenological variables, with limited connection to the underlying molecular mechanisms. Here, we analytically solve a stochastic threshold–ac...
work page internal anchor Pith review Pith/arXiv arXiv 2026
-
[5]
M. B. Ginzberg, R. Kafri, and M. Kirschner, Cell biol- ogy. on being the right (cell) size, Science348, 1245075 (2015)
work page 2015
-
[6]
G. E. Neurohr, R. L. Terry, J. Lengefeld, M. Bonney, G. P. Brittingham, F. Moretto, T. P. Miettinen, L. P. Vaites, L. M. Soares, J. A. Paulo, J. W. Harper, S. Bu- ratowski, S. Manalis, F. J. van Werven, L. J. Holt, and A. Amon, Excessive cell growth causes cytoplasm dilu- tion and contributes to senescence, Cell176, 1083 (2019)
work page 2019
-
[7]
G. E. Neurohr and A. Amon, Relevance and regulation of cell density, Trends Cell Biol.30, 213 (2020)
work page 2020
-
[8]
S. Xie, M. Swaffer, and J. M. Skotheim, Eukaryotic cell size control and its relation to biosynthesis and senes- cence, Annu. Rev. Cell Dev. Biol.38, 291 (2022)
work page 2022
-
[9]
J. Lengefeld and E. Zatulovskiy, Editorial: Cell size reg- ulation: molecular mechanisms and physiological impor- tance, Front. Cell Dev. Biol.11, 1219294 (2023)
work page 2023
- [10]
-
[11]
S. Taheri-Araghi, S. Bradde, J. T. Sauls, N. S. Hill, P. A. Levin, J. Paulsson, M. Vergassola, and S. Jun, Cell-size control and homeostasis in bacteria, Curr. Biol.25, 385 (2015)
work page 2015
- [12]
-
[13]
D. Chandler-Brown, K. M. Schmoller, Y. Winetraub, and J. M. Skotheim, The adder phenomenon emerges from independent control of pre- and post-start phases of the budding yeast cell cycle, Curr. Biol.27, 2774 (2017)
work page 2017
-
[14]
C. Garmendia-Torres, O. Tassy, A. Matifas, N. Molina, and G. Charvin, Multiple inputs ensure yeast cell size homeostasis during cell cycle progression, eLife7, e34025 (2018)
work page 2018
-
[15]
Y. J. Eun, P. Y. Ho, M. Kim, S. LaRussa, L. Robert, L. D. Renner, A. Schmid, E. Garner, and A. Amir, Ar- chaeal cells share common size control with bacteria de- spite noisier growth and division, Nat. Microbiol.3, 148 (2017)
work page 2017
- [16]
- [17]
-
[18]
Amir, Cell size regulation in bacteria, Phys
A. Amir, Cell size regulation in bacteria, Phys. Rev. Lett. 112(2014)
work page 2014
- [19]
-
[20]
P. Y. Ho, J. Lin, and A. Amir, Modeling cell size regula- tion: From single-cell-level statistics to molecular mech- anisms and population-level effects, Annu. Rev. Biophys. 47, 251 (2018)
work page 2018
- [21]
- [22]
- [23]
- [24]
-
[25]
S. Tiruvadi-Krishnan, J. M¨ annik, P. Kar, J. Lin, A. Amir, and J. M¨ annik, Coupling between DNA replication, seg- regation, and the onset of constriction in escherichia coli, Cell Rep.38, 110539 (2022)
work page 2022
-
[26]
P. Kar, S. Tiruvadi-Krishnan, J. M¨ annik, J. M¨ annik, and A. Amir, Using conditional independence tests to eluci- date causal links in cell cycle regulation in escherichia coli, Proc. Natl. Acad. Sci. U. S. A.120, e2214796120 (2023)
work page 2023
-
[27]
L. K. Harris and J. A. Theriot, Relative rates of surface and volume synthesis set bacterial cell size, Cell165, 1479 (2016)
work page 2016
- [28]
- [29]
-
[30]
F. Si, G. Le Treut, J. T. Sauls, S. Vadia, P. A. Levin, and S. Jun, Mechanistic origin of cell-size control and homeostasis in bacteria, Curr. Biol.29, 1760 (2019)
work page 2019
-
[31]
P. P. Pandey, H. Singh, and S. Jain, Exponential trajec- tories, cell size fluctuations, and the adder property in bacteria follow from simple chemical dynamics and divi- sion control, Phys. Rev. E.101, 062406 (2020)
work page 2020
-
[32]
M. Panlilio, J. Grilli, G. Tallarico, I. Iuliani, B. Sclavi, P. Cicuta, and M. Cosentino Lagomarsino, Threshold ac- cumulation of a constitutive protein explains e. coli cell- division behavior in nutrient upshifts, Proc. Natl. Acad. Sci. U. S. A.118(2021)
work page 2021
-
[33]
M. ElGamel and A. Mugler, Effects of molecular noise on cell size control, Phys. Rev. Lett.132, 098403 (2024)
work page 2024
-
[34]
L. Luo, Y. Bai, and X. Fu, Stochastic threshold in cell size control, Phys. Rev. Res.5(2023)
work page 2023
-
[35]
J. Pla, M. S´ anchez, P. Palacios, M. Vicente, and M. Aldea, Preferential cytoplasmic location of FtsZ, a protein essential for escherichia coli septation, Mol. Mi- crobiol.5, 1681 (1991). 6
work page 1991
- [36]
-
[37]
A. Ursinus, F. van den Ent, S. Brechtel, M. de Pedro, J.-V. H¨ oltje, J. L¨ owe, and W. Vollmer, Murein (pepti- doglycan) binding property of the essential cell division protein FtsN from escherichia coli, J. Bacteriol.186, 6728 (2004)
work page 2004
-
[38]
T. Mohammadi, G. E. J. Ploeger, J. Verheul, A. D. Com- valius, A. Martos, C. Alfonso, J. van Marle, G. Rivas, and T. den Blaauwen, The GTPase activity of escherichia coli FtsZ determines the magnitude of the FtsZ poly- mer bundling by ZapA in vitro, Biochemistry48, 11056 (2009)
work page 2009
-
[39]
H. P. Erickson, D. E. Anderson, and M. Osawa, FtsZ in bacterial cytokinesis: cytoskeleton and force generator all in one, Microbiol. Mol. Biol. Rev.74, 504 (2010)
work page 2010
-
[40]
G. W. Li, D. Burkhardt, C. Gross, and J. S. Weiss- man, Quantifying absolute protein synthesis rates reveals principles underlying allocation of cellular resources, Cell 157, 624 (2014)
work page 2014
-
[41]
B. Liu, L. Persons, L. Lee, and P. A. J. de Boer, Roles for both FtsA and the FtsBLQ subcomplex in FtsN- stimulated cell constriction in escherichia coli, Mol. Mi- crobiol.95, 945 (2015)
work page 2015
-
[42]
N. O. E. Vischer, J. Verheul, M. Postma, B. van den Berg van Saparoea, E. Galli, P. Natale, K. Gerdes, J. Luirink, W. Vollmer, M. Vicente, and T. den Blaauwen, Cell age dependent concentration of es- cherichia coli divisome proteins analyzed with ImageJ and ObjectJ, Front. Microbiol.6, 586 (2015)
work page 2015
-
[43]
A. J. F. Egan and W. Vollmer, The stoichiometric divi- some: a hypothesis, Front. Microbiol.6, 455 (2015)
work page 2015
-
[44]
W. D. Ramey and E. E. Ishiguro, Site of inhibition of peptidoglycan biosynthesis during the stringent response in escherichia coli, J. Bacteriol.135, 71 (1978)
work page 1978
-
[45]
Y. van Heijenoort, M. G´ omez, M. Derrien, J. Ayala, and J. van Heijenoort, Membrane intermediates in the pepti- doglycan metabolism of escherichia coli: possible roles of PBP 1b and PBP 3, J. Bacteriol.174, 3549 (1992)
work page 1992
-
[46]
van Heijenoort, Lipid intermediates in the biosynthesis of bacterial peptidoglycan, Microbiol
J. van Heijenoort, Lipid intermediates in the biosynthesis of bacterial peptidoglycan, Microbiol. Mol. Biol. Rev.71, 620 (2007)
work page 2007
-
[48]
C. Gardiner,Stochastic Methods. A Handbook for the Natural and Social Sciences, 5th ed., Springer Series in Synergetics (Springer Berlin Heidelberg, 2009)
work page 2009
-
[49]
A. Adiciptaningrum, M. Osella, M. C. Moolman, M. Cosentino Lagomarsino, and S. J. Tans, Stochastic- ity and homeostasis in the e. coli replication and division cycle, Sci. Rep.5, 18261 (2015)
work page 2015
-
[50]
A. S. Kennard, M. Osella, A. Javer, J. Grilli, P. Nghe, S. J. Tans, P. Cicuta, and M. Cosentino Lagomarsino, Individuality and universality in the growth-division laws of single e. coli cells, Phys. Rev. E.93, 012408 (2016)
work page 2016
-
[51]
M. Wallden, D. Fange, E. G. Lundius, ¨O. Baltekin, and J. Elf, The synchronization of replication and division cycles in individual e. coli cells, Cell166, 729 (2016)
work page 2016
-
[52]
G. Witz, E. van Nimwegen, and T. Julou, Initiation of chromosome replication controls both division and repli- cation cycles inE. colithrough a double-adder mecha- nism, eLife8, e48063 (2019)
work page 2019
-
[53]
P. Y. Ho and A. Amir, Simultaneous regulation of cell size and chromosome replication in bacteria, Front. Mi- crobiol.6, 662 (2015)
work page 2015
- [54]
- [55]
-
[56]
S. Hobson-Gutierrez and E. Kussell, Evolutionary advan- tage of cell size control, Phys. Rev. Lett.134, 118401 (2025)
work page 2025
-
[57]
Essential Role of Extrinsic Noise in Models ofE. coli Division Control
F. Proulx-Giraldeau, J. M. Skotheim, and P. Fran¸ cois, Evolution of cell size control is canalized towards adders or sizers by cell cycle structure and selective pressures, eLife11, e79919 (2022). Supplementary Information for “Essential Role of Extrinsic Noise in Models ofE. coli Division Control” A. PHENOMENOLOGICAL FRAMEWORK FOR CELL DIVISION CONTROL ...
work page 2022
-
[58]
=σ 2 s0 + α µ Cov(θi, si 0)−rCov(θ i−1, si 0) ,(S21) so that it remains to evaluate Cov(θ i, si
-
[59]
and Cov(θ i−1, si 0). Calculation ofCov(θ i, si 0).To compute this term, we use symmetric division,s i 0 = si−1 f /2, and Eq. (S20), to write Cov(θi, si
-
[60]
= 1 2 Cov θi, s i−1 f = 1 2 Cov θi, s i−1 0 + α µ θi−1 −rθ i−2 = 1 2 Cov(θi, si−1 0 ) + 1 2 α µ Cov(θi, θi−1)−rCov(θ i, θi−2) .(S22) Now, using the threshold statistics of Eq. (S19), we obtain Cov(θi, si
-
[61]
= 1 2 Cov(θi, si−1 0 ) + 1 2 α µ ρ−rρ 2 σ2 θ .(S23) Finally, using the discrete-time AR(1) threshold dynamics in Eq. (S18) and the fact thatη i is independent ofs i−1 0 , we can write Cov(θi, si−1 0 ) =ρCov(θ i−1, si−1 0 ),(S24) so that Eq. (S23) becomes the recursion Cov(θi, si
-
[62]
= 1 2 ρCov(θ i−1, si−1 0 ) + 1 2 α µ ρ−rρ 2 σ2 θ .(S25) In the stationary regime, Cov(θ i, si
- [63]
-
[64]
= 1 2 α µ ρ−rρ 2 1−ρ/2 σ2 θ .(S26) 4 Calculation ofCov(θ i−1, si 0):Following similar algebraic steps, and using the AR(1) relation Cov(θ i−1, si−1 0 ) = Cov(θi, si
-
[65]
in stationarity, we obtain Cov(θi−1, si
-
[66]
= 1 2 Cov(θi−1, si−1 0 ) + 1 2 α µ (1−rρ)σ 2 θ = 1 2 α µ 1−rρ 1−ρ/2 σ2 θ .(S27) Now substituting Eqs. (S26) and (S27) into Eq. (S21), we obtain Cov(si f , si
-
[67]
=σ 2 s0 + 1 2 α µ 2 σ2 θ (1−rρ)(ρ−r) 1−ρ/2 .(S28) Hence, in a stationary condition, the slope of the linear regression between birth and division size becomes 1−ζ ∆ = 1 + 1 2 α µ 2 σ2 θ σ2 s0 (1−rρ)(ρ−r) 1−ρ/2 .(S29) Size fluctuations As explained in the text, to computeσ 2 s0 in the presence of threshold fluctuations, we can use the law of total variance...
-
[68]
= Cov si 02−ϵ + α µ(1 +ϵ) θi −rθ i−12−ϵ , s i 0 , = 2−ϵσ2 s0 + α µ(1 +ϵ) Cov(θi, si 0)−r2 −ϵ Cov(θi−1, si 0) .(S46) Furthermore, Cov(θi, si
-
[69]
= 0 and Cov(θi−1, si
-
[70]
= 1 2 Cov θi−1, s i−1 f = 1 2 α µ(1 +ϵ)σ 2 θ , so that 1−ζ ∆ ≡ Cov(sf , s0) σ2 s0 = 1 2ϵ 1−(1 +ϵ) 2 r 2 α µ 2 σ2 θ σ2 s0 .(S47) From Eq. (S47), we can compute the division control parameterλ G as λG = 1− 1−ζ ∆ 2 = 1− 1 2ϵ+1 1−(1 +ϵ) 2 r 2 α µ 2 σ2 θ σ2 s0 .(S48) Size fluctuations We compute the variance of cell size at birth using the law of total varianc...
-
[71]
= Cov si 0 + α µ θi −rθ i−1 −ντ i , s i 0 , =σ 2 s0 + α µ Cov(θi, si 0)−rCov(θ i−1, si 0)−νCov(τ i, si 0) .(S53) Furthermore, Cov(θi, si
-
[72]
= 0. By considering small noise approximation around mean value si 0 =⟨s 0⟩+ ∆ i, the other two covariances will be given as below: Cov(si 0, τ i) = Cov si 0, 1 α ln si f si 0 !! (S54) = 1 α Cov si 0,ln 1 + ∆i+1 ⟨s0⟩ −Cov si 0,ln 1 + ∆i ⟨s0⟩ (S55) ≈ 1 α Cov si 0, ∆i+1 ⟨s0⟩ −Cov si 0, ∆i ⟨s0⟩ (S56) ≈ σ2 s0 α⟨s0⟩ 1−ζ ∆ 2 −1 ,[1−ζ ∆ = Cov(si f , si 0)/σ2 s0]...
-
[73]
Amir, Cell size regulation in bacteria, Phys
A. Amir, Cell size regulation in bacteria, Phys. Rev. Lett.112(2014)
work page 2014
-
[74]
P. Y. Ho, J. Lin, and A. Amir, Modeling cell size regulation: From single-cell-level statistics to molecular mechanisms and population-level effects, Annu. Rev. Biophys.47, 251 (2018)
work page 2018
- [75]
- [76]
-
[77]
M. ElGamel and A. Mugler, Effects of molecular noise on cell size control, Phys. Rev. Lett. 132, 098403 (2024)
work page 2024
-
[78]
N. G. Van Kampen,Stochastic processes in physics and chemistry, 3rd ed., North-Holland Personal Library (North-Holland, Oxford, England, 2007)
work page 2007
-
[79]
C. Gardiner,Stochastic Methods. A Handbook for the Natural and Social Sciences, 5th ed., Springer Series in Synergetics (Springer Berlin Heidelberg, 2009). 14
work page 2009
discussion (0)
Sign in with ORCID, Apple, or X to comment. Anyone can read and Pith papers without signing in.